Denosumab, Romosozumab, and Teriparatide
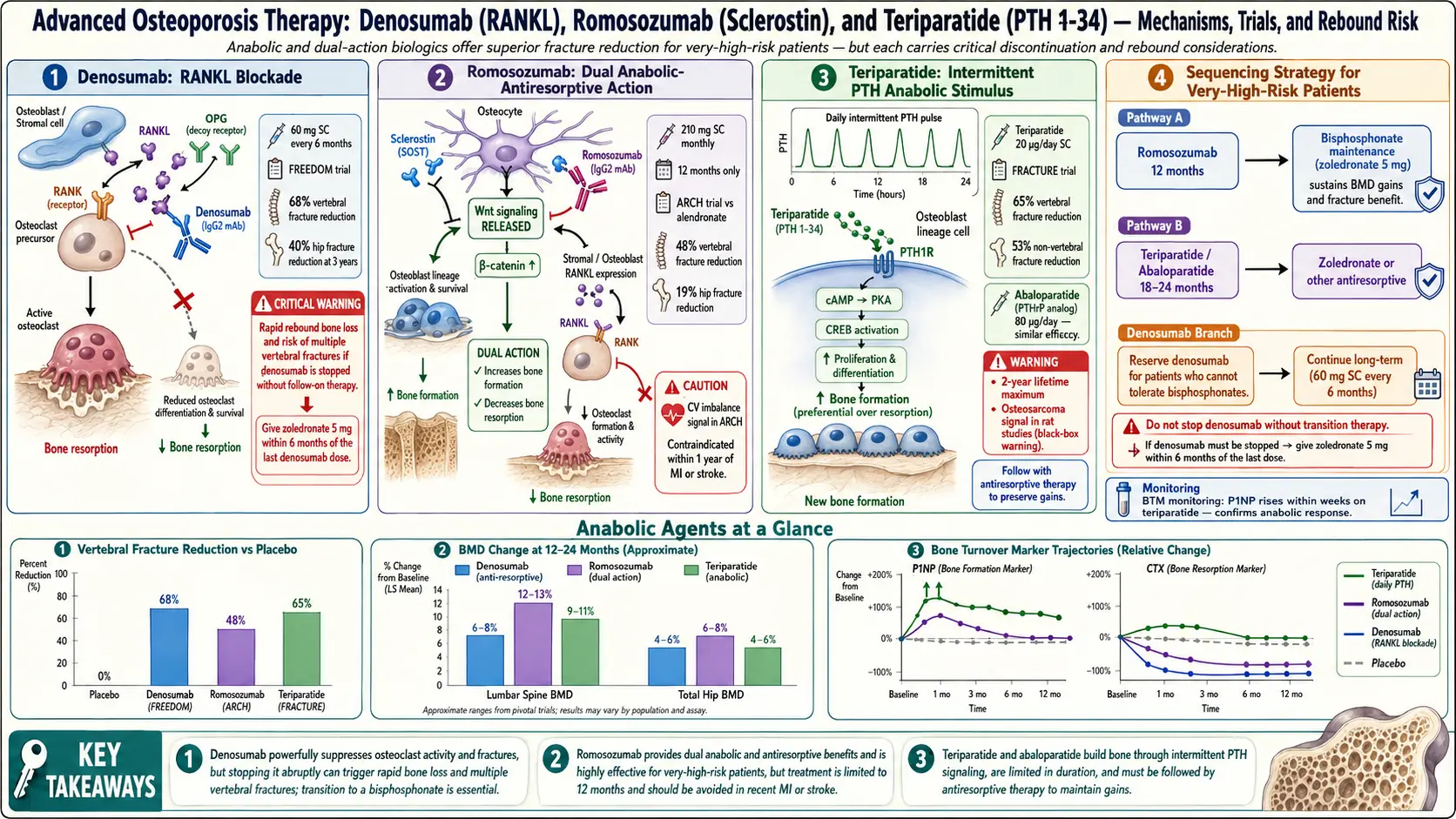
Table of Contents
- Who These Drugs Are For
- Two Categories: Antiresorptive vs. Anabolic
- Denosumab (Prolia) — How It Works
- Denosumab Evidence: The FREEDOM Trial
- The Rebound Fracture Problem
- Teriparatide (Forteo) — The First Anabolic
- Abaloparatide (Tymlos) — The PTHrP Cousin
- Romosozumab (Evenity) — The Dual-Action Drug
- The Romosozumab Cardiovascular Warning
- Sequencing: The Order Actually Matters
- Side Effects and Monitoring
- Cost, Insurance, and Getting Approved
- Questions to Ask Your Doctor
- Key Research Papers
- Connections
- Featured Videos
Who These Drugs Are For
If you are reading this page, your DEXA scan probably came back worse than expected, you broke a bone that should not have broken, or your doctor mentioned a drug that sounds expensive and complicated. The three drugs in the title — denosumab, teriparatide, and romosozumab — are the heavy hitters of osteoporosis pharmacology. They are not first-line for a mild bone-density dip. They are for people at high or very high fracture risk: T-score below −2.5 with a recent fragility fracture, multiple vertebral compression fractures, a hip fracture in the last year or two, or a FRAX 10-year major-fracture probability above roughly 20% (or hip-fracture probability above 3%).
These drugs exist because oral bisphosphonates — alendronate, risedronate, ibandronate — do not work for everyone. Some patients cannot tolerate them (reflux, esophagitis). Some absorb them poorly. Some have already been on them for five or ten years and continue to lose bone or sustain fractures. And some arrive at the rheumatologist's office with a T-score of −3.8 and two crushed vertebrae, in which case starting with an oral bisphosphonate is a bit like bringing a garden hose to a house fire. For those patients, the injectable biologics and the bone-building drugs change the trajectory of disease.
This page walks through what each drug does, the pivotal trials, the traps (especially the denosumab discontinuation rebound and the romosozumab cardiovascular signal), how they are sequenced, and what it all costs.
Two Categories: Antiresorptive vs. Anabolic
Bone is constantly remodeled. Osteoclasts chew up old bone (resorption). Osteoblasts lay down new bone (formation). In a healthy young adult the two processes balance. In osteoporosis, resorption outpaces formation and net bone mass falls.
Osteoporosis drugs split into two mechanistic camps:
- Antiresorptive drugs slow the osteoclasts. Less bone is chewed away, so net density rises. Bisphosphonates and denosumab work this way. Think of it as cutting spending rather than earning more.
- Anabolic drugs stimulate the osteoblasts to build fresh bone. Teriparatide and abaloparatide are pure anabolics. Romosozumab is the hybrid: it both stimulates bone formation and suppresses resorption, a dual action that produces the largest short-term density gains of any approved therapy.
This distinction is not academic. It drives two practical rules. First, anabolics have caps on how long you can use them — they are sprints, not marathons. Second, whatever gains you build during an anabolic course will evaporate within a year or two unless you follow with an antiresorptive. The drugs are tools in a sequence, not stand-alone solutions.
Denosumab (Prolia) — How It Works
Denosumab is a fully human monoclonal antibody that binds RANKL (receptor activator of nuclear factor kappa-B ligand). RANKL is the signal osteoblasts and other cells send to osteoclast precursors saying "grow up and start dissolving bone." When denosumab binds RANKL, that signal never reaches the receptor (RANK) on the osteoclast precursor. No signal, no mature osteoclast, no resorption. Within days of a dose, markers of bone turnover (CTX, P1NP) fall to near-undetectable levels.
The dosing is one of its selling points: 60 mg subcutaneous injection every six months. Your rheumatologist, primary care doctor, or infusion nurse gives it — most often in the back of the upper arm, thigh, or abdomen. There is no requirement to stand upright for thirty minutes, no esophageal irritation, no fasting. For patients who failed oral bisphosphonates for GI reasons, denosumab solved a real problem.
Prolia is the 60 mg formulation for osteoporosis. Xgeva, the same molecule at 120 mg monthly, is used in cancer-related skeletal disease — different dose, different indication. If anyone ever writes you a prescription for Xgeva for osteoporosis, that is a mistake.
Denosumab Evidence: The FREEDOM Trial
The pivotal study was FREEDOM (Fracture Reduction Evaluation of Denosumab in Osteoporosis Every 6 Months), published by Cummings and colleagues in the New England Journal of Medicine in 2009. Nearly 8,000 postmenopausal women with T-scores between −2.5 and −4.0 were randomized to denosumab or placebo for three years. The results were definitive:
- 68% reduction in new vertebral fractures.
- 40% reduction in hip fractures.
- 20% reduction in non-vertebral fractures.
- Lumbar-spine BMD increased roughly 9% and total-hip BMD roughly 6% over three years.
The FREEDOM Extension trial followed a subset for an additional seven years — ten years of denosumab in total. Unlike bisphosphonates, where BMD tends to plateau, denosumab produced continuous BMD gains year after year. By year ten, lumbar-spine BMD was up roughly 21% from baseline and hip BMD roughly 9%. Fracture rates stayed low throughout. For a drug given twice a year with few side effects, these numbers are genuinely impressive.
The Rebound Fracture Problem
Here is the catch that makes denosumab both loved and feared by endocrinologists. When you stop taking it, the effect does not just fade — it reverses violently. Within six to twelve months of a missed dose, osteoclast activity rebounds to levels higher than baseline, bone turnover markers spike, BMD falls rapidly back toward pre-treatment values, and patients experience a wave of multiple vertebral compression fractures. Cases of patients sustaining four, five, or even eight simultaneous new vertebral fractures have been reported in the literature.
This is not theoretical. Cummings et al. re-analyzed FREEDOM data in 2018 and showed that patients who discontinued denosumab had vertebral fracture rates that rose to and then exceeded the placebo group within two years of stopping. The mechanism is biological: denosumab never actually kills osteoclast precursors, it just silences their signaling. When the antibody washes out, a large population of un-stimulated precursors floods into action all at once.
The practical rules that follow from this are absolute:
- Never miss a dose by more than four weeks. If your injection is due in May, do not let it drift to August. Put it in your calendar the day you get each shot.
- Do not stop denosumab without a follow-on plan. If you and your doctor decide to stop — because of side effects, cost, or preference — you must transition to a potent antiresorptive, almost always zoledronic acid (one IV infusion), timed roughly six months after what would have been your next Prolia dose.
- Do not treat denosumab as a drug holiday candidate. Bisphosphonates have drug holidays (see the bisphosphonates article). Denosumab does not.
If you ever find out your insurance changed and Prolia needs prior auth again, do not wait passively. Call your rheumatologist that day. The rebound clock is already ticking.
Teriparatide (Forteo) — The First Anabolic
Teriparatide is recombinant parathyroid hormone 1-34 — the first 34 amino acids of your own PTH, the same hormone your parathyroid glands secrete. Chronic elevation of PTH (as in hyperparathyroidism) dissolves bone. But pulsed PTH exposure — a once-daily injection that peaks and falls within hours — does the opposite. It preferentially activates osteoblasts, which build new bone faster than osteoclasts can remove it. For the first time in osteoporosis treatment, patients were not just preserving bone but making new trabecular microarchitecture.
Dosing: 20 micrograms subcutaneously once daily, from a prefilled multi-dose pen that looks like an insulin injector. You give it to yourself in the thigh or abdomen, usually in the evening. Most patients get comfortable with the self-injection within a few days.
The pivotal Fracture Prevention Trial (FPT), published by Neer and colleagues in NEJM in 2001, randomized 1,637 postmenopausal women with prior vertebral fractures to teriparatide or placebo. At a median of 21 months of follow-up, teriparatide cut new vertebral fractures by 65% and non-vertebral fragility fractures by 53%. The trial was stopped early when rat studies showed osteosarcomas at very high lifetime doses. Follow-up data in humans has not shown an osteosarcoma signal at therapeutic exposure, and the FDA removed the black-box warning in 2020.
Two hard caps: the lifetime maximum is two years, and you only get one two-year course. The two-year limit is partly regulatory history and partly biological — after 18–24 months the anabolic window starts to close as osteoclasts catch up. At the end of the course, BMD gains are real but will vanish without a follow-on antiresorptive. Zoledronic acid or denosumab is the standard choice.
Abaloparatide (Tymlos) — The PTHrP Cousin
Abaloparatide is a synthetic analog of PTH-related protein (PTHrP), a paracrine signaling molecule involved in skeletal development. It binds the same receptor (PTH1R) as teriparatide but with a different conformational preference that biases the receptor toward the anabolic signaling branch. In theory, this means more bone formation for less resorption per unit of dose.
Dosing: 80 micrograms subcutaneously daily, also via prefilled pen, also capped at 18–24 months.
The ACTIVE trial (Miller et al., JAMA 2016) randomized 2,463 postmenopausal women with osteoporosis to abaloparatide, teriparatide, or placebo for 18 months. Abaloparatide reduced new vertebral fractures by 86% and non-vertebral fractures by 43% versus placebo. Compared head-to-head with teriparatide, abaloparatide produced numerically greater hip BMD gains and less hypercalcemia, though the two drugs have never been compared with fracture endpoints in a powered trial. In practice, abaloparatide and teriparatide are interchangeable in most patients; the choice often comes down to insurance formulary and co-pay.
Romosozumab (Evenity) — The Dual-Action Drug
Romosozumab is a humanized monoclonal antibody against sclerostin, a protein secreted by osteocytes (the mature bone cells embedded in mineralized bone) that normally suppresses the Wnt signaling pathway in osteoblasts. Sclerostin is, in effect, bone's built-in brake on construction. Remove the brake and osteoblasts proliferate and lay down matrix. But the biology has a second dimension: the same Wnt signaling that drives osteoblasts also, indirectly, reduces RANKL and therefore slows osteoclasts. Romosozumab is simultaneously anabolic and antiresorptive — the only approved drug that does both.
Dosing: 210 mg subcutaneously once monthly for 12 months, delivered as two 105 mg injections back-to-back (your nurse will give them in adjacent sites). After the 12-month course you must transition to an antiresorptive — alendronate, zoledronic acid, or denosumab — or nearly all the BMD gains will be lost within a year.
Two pivotal trials:
- FRAME (Cosman et al., NEJM 2016). 7,180 postmenopausal women, romosozumab vs. placebo for 12 months, then both groups on denosumab for 12 months. At 12 months, new vertebral fractures were reduced 73%. Lumbar-spine BMD rose 13% in one year — a number no other osteoporosis drug has matched in that timeframe. The effect held through year two on denosumab.
- ARCH (Saag et al., NEJM 2017). 4,093 postmenopausal women with a prior fragility fracture, romosozumab for 12 months vs. alendronate for 12 months, then both groups on alendronate for another year. Head-to-head, romosozumab-then-alendronate beat alendronate-then-alendronate: 48% fewer new vertebral fractures and 38% fewer hip fractures at 24 months.
These are, on paper, the best fracture numbers any osteoporosis drug has ever produced. Romosozumab is the drug of choice for patients with very high fracture risk — recent hip fracture, multiple vertebral fractures, T-score below −3.5 — who need the biggest possible early density gain.
The Romosozumab Cardiovascular Warning
ARCH also produced a signal that made regulators nervous. Over 12 months, romosozumab patients had more major adverse cardiovascular events (cardiac death, non-fatal myocardial infarction, non-fatal stroke) than alendronate patients: 2.5% vs. 1.9%. The absolute difference was small, the numbers were not statistically overwhelming, and FRAME (which compared romosozumab to placebo rather than alendronate) did not show the same imbalance. But the FDA took the signal seriously and added a boxed warning when Evenity was approved in 2019.
Practical rules:
- Do not use romosozumab within one year of a heart attack or stroke. This is a hard contraindication.
- In patients with known coronary artery disease, prior stroke, or high cardiovascular risk factors (uncontrolled hypertension, diabetes with end-organ damage), the benefit/risk calculation shifts — many of those patients should get a different drug.
- If romosozumab is the right choice despite CV risk, the decision needs to be a joint one between you, your bone specialist, and your cardiologist, documented clearly.
For a 72-year-old woman with a recent hip fracture and no CV history, the fracture-prevention benefit clearly outweighs the CV risk. For a 78-year-old diabetic with three prior stents and ongoing angina, it does not. Know which patient you are.
Sequencing: The Order Actually Matters
For decades, osteoporosis was treated as "pick one drug, stay on it." The modern understanding is that drugs should be deployed in sequences designed to maximize lifetime BMD and minimize fracture risk. The single most important principle is this:
Anabolic first, then antiresorptive. Not the reverse.
Evidence from several crossover studies (including the DATA-Switch trial) showed that patients who got teriparatide or romosozumab after long prior bisphosphonate exposure had blunted anabolic responses. Prolonged osteoclast suppression before giving an anabolic agent partially closes the window of opportunity — osteoblasts need their osteoclast partners to cycle normally before they can be maximally stimulated. Going the other direction — anabolic first, then locking in the gains with an antiresorptive — produces substantially larger BMD increases.
A few concrete sequences used in practice:
- Very high risk, treatment-naive: Romosozumab 12 months → zoledronic acid (annual IV) or denosumab (twice yearly). Front-loads the largest possible BMD gain, then protects it.
- High risk, treatment-naive, no CV concern: Teriparatide or abaloparatide 18–24 months → denosumab or zoledronic acid.
- Already on oral bisphosphonate but still fracturing: Stop the oral, start teriparatide, abaloparatide, or romosozumab; be prepared for a somewhat smaller anabolic response than a naive patient would get. Follow with an antiresorptive.
- On denosumab and planning to stop: Never stop into nothing. Give zoledronic acid roughly six months after the last Prolia dose, then monitor CTX.
The take-home: if your doctor starts you on alendronate now and then, five years from now, thinks about teriparatide, the teriparatide will work less well than it would have if used first. This is a worth-pushing-on conversation at diagnosis.
Side Effects and Monitoring
Denosumab. Common: mild injection-site reactions, back pain, muscle ache, eczema. Uncommon but serious: hypocalcemia (especially in patients with chronic kidney disease — always correct low calcium and vitamin D before dosing), skin infections including cellulitis, atypical femoral fractures (rare, shared with bisphosphonates), and osteonecrosis of the jaw (ONJ, roughly 0.05% annual risk; see dental guidance below).
Teriparatide and abaloparatide. Common: transient nausea (first injections), leg cramps, mild dizziness on standing after early doses, headache. Monitor serum calcium — mild hypercalcemia is common and usually benign. Contraindicated in Paget disease of bone, unexplained elevations of alkaline phosphatase, prior skeletal radiation, and active or prior bone cancer.
Romosozumab. Common: arthralgia, headache, injection-site reactions. Serious: the cardiovascular signal above, plus ONJ and atypical femoral fractures (very rare). Like denosumab, hypocalcemia needs to be corrected before starting.
Osteonecrosis of the jaw (ONJ) is rare with all three drugs at osteoporosis doses but real. Get a dental cleaning and any needed extractions or implants done before starting if possible, maintain good oral hygiene throughout, and tell your dentist which drug you are on before any invasive procedure. Most rheumatologists do not require stopping Prolia before routine dental work; for major surgery the timing can be coordinated.
Monitoring. DEXA every 1–2 years, bone turnover markers (CTX, P1NP) in selected cases, serum calcium and 25(OH)D at baseline and periodically, and calcium 1000–1200 mg/day plus vitamin D 800–1000 IU/day throughout. See the calcium, vitamin D, and protein article for details.
Cost, Insurance, and Getting Approved
List prices (U.S., 2025–2026 range; your actual cost depends on insurance, Medicare Part B vs. Part D, and manufacturer copay cards):
- Prolia (denosumab): roughly $1,500–$1,800 per 60 mg injection, so about $3,000–$3,600/year. Usually billed through Medicare Part B as a provider-administered drug, which means 20% coinsurance unless you have a supplement. Amgen's FIRSTSTEP copay program can reduce commercial-insurance out-of-pocket to $25 per dose.
- Forteo (teriparatide): brand pen roughly $3,500–$4,000/month. A generic teriparatide (Bonsity) is now available and cheaper. Usually Medicare Part D, so subject to the deductible and donut-hole math until out-of-pocket hits the 2025 cap.
- Tymlos (abaloparatide): roughly $2,000–$2,700/month list. Manufacturer copay card (commercial insurance) brings it near $0 for many patients.
- Evenity (romosozumab): roughly $1,800–$2,200 per monthly dose (two 105 mg injections), about $22,000–$26,000 for the full 12-month course. Usually Medicare Part B (provider-administered).
Prior authorization is the rule, not the exception. Every payer wants documentation of (1) a qualifying DEXA T-score or a fragility fracture, (2) failure, intolerance, or contraindication to oral bisphosphonates for the anabolics, (3) adequate calcium and vitamin D, and (4) a plan for the follow-on antiresorptive with romosozumab and teriparatide. Appeals succeed more often than people assume — do not take a first denial as final. A well-written appeal citing FREEDOM, ACTIVE, FRAME, or ARCH and documenting why the patient needs this specific drug (recent hip fracture, stacked vertebral fractures, failed oral therapy) wins a majority of the time.
Manufacturer patient-assistance programs (Amgen Assist, Radius Health Patient Support, Lilly Cares) can cover the drug entirely for uninsured patients who meet income thresholds. Ask your prescriber's office for the forms — most rheumatology practices do these weekly.
Questions to Ask Your Doctor
- Given my T-score, FRAX, and fracture history, am I in the "high" or "very high" risk category?
- Should we start with an anabolic (teriparatide, abaloparatide, or romosozumab) rather than an oral bisphosphonate?
- What is the full sequence you envision — not just the first drug but the follow-on?
- If romosozumab is on the table, what is my cardiovascular risk, and do we need a cardiology opinion first?
- If denosumab is on the table, do we have a contingency plan if insurance ever lapses so I do not miss a dose?
- Do I need a dental clearance before starting?
- What bone turnover markers (if any) will we check, and how often?
- What is the realistic out-of-pocket cost, and is there a copay or assistance program I should apply to now?
Key Research Papers
Pivotal trials, long-term extensions, and discontinuation studies for denosumab, teriparatide, abaloparatide, and romosozumab, together with the sequencing and combination evidence that determines how these agents are used in practice.
- Cummings SR, San Martin J, McClung MR, et al. Denosumab for Prevention of Fractures in Postmenopausal Women with Osteoporosis (FREEDOM). New England Journal of Medicine. 2009;361(8):756–765.
- Bone HG, Wagman RB, Brandi ML, et al. 10 Years of Denosumab Treatment in Postmenopausal Women with Osteoporosis: Results from the Phase 3 Randomised FREEDOM Trial and Open-Label Extension. Lancet Diabetes & Endocrinology. 2017;5(7):513–523.
- Cummings SR, Ferrari S, Eastell R, et al. Vertebral Fractures After Discontinuation of Denosumab: A Post Hoc Analysis of the Randomized Placebo-Controlled FREEDOM Trial and Its Extension. Journal of Bone and Mineral Research. 2018;33(2):190–198.
- Anastasilakis AD, Polyzos SA, Makras P, Aubry-Rozier B, Kaouri S, Lamy O. Clinical Features of 24 Patients with Rebound-Associated Vertebral Fractures After Denosumab Discontinuation: Systematic Review and Additional Cases. Journal of Bone and Mineral Research. 2017;32(6):1291–1296.
- Neer RM, Arnaud CD, Zanchetta JR, et al. Effect of Parathyroid Hormone (1-34) on Fractures and Bone Mineral Density in Postmenopausal Women with Osteoporosis (Fracture Prevention Trial). New England Journal of Medicine. 2001;344(19):1434–1441.
- Miller PD, Hattersley G, Riis BJ, et al. Effect of Abaloparatide vs Placebo on New Vertebral Fractures in Postmenopausal Women with Osteoporosis: The ACTIVE Randomized Clinical Trial. JAMA. 2016;316(7):722–733.
- Cosman F, Crittenden DB, Adachi JD, et al. Romosozumab Treatment in Postmenopausal Women with Osteoporosis (FRAME). New England Journal of Medicine. 2016;375(16):1532–1543.
- Saag KG, Petersen J, Brandi ML, et al. Romosozumab or Alendronate for Fracture Prevention in Women with Osteoporosis (ARCH). New England Journal of Medicine. 2017;377(15):1417–1427.
- Leder BZ, Tsai JN, Uihlein AV, et al. Denosumab and Teriparatide Transitions in Postmenopausal Osteoporosis (the DATA-Switch Study): Extension of a Randomised Controlled Trial. Lancet. 2015;386(9999):1147–1155.
- Tsai JN, Uihlein AV, Lee H, et al. Teriparatide and Denosumab, Alone or Combined, in Women with Postmenopausal Osteoporosis: The DATA Study Randomised Trial. Lancet. 2013;382(9886):50–56.
- Eastell R, Rosen CJ, Black DM, Cheung AM, Murad MH, Shoback D. Pharmacological Management of Osteoporosis in Postmenopausal Women: An Endocrine Society Clinical Practice Guideline. Journal of Clinical Endocrinology & Metabolism. 2019;104(5):1595–1622.
- Khan AA, Morrison A, Hanley DA, et al. Diagnosis and Management of Osteonecrosis of the Jaw: A Systematic Review and International Consensus. Journal of Bone and Mineral Research. 2015;30(1):3–23.
- Shoback D, Rosen CJ, Black DM, Cheung AM, Murad MH, Eastell R. Pharmacological Management of Osteoporosis in Postmenopausal Women: An Endocrine Society Guideline Update. Journal of Clinical Endocrinology & Metabolism. 2020;105(3):587–594.
Live PubMed Searches
Live PubMed queries that update as new papers are indexed.
- PubMed: denosumab FREEDOM
- PubMed: denosumab rebound fractures
- PubMed: teriparatide fracture prevention
- PubMed: abaloparatide ACTIVE
- PubMed: romosozumab FRAME ARCH
- PubMed: romosozumab cardiovascular
- PubMed: anabolic to antiresorptive sequencing
- PubMed: DATA-Switch trial
- PubMed: sclerostin and Wnt signaling
- PubMed: ONJ antiresorptives
Connections
- Osteoporosis
- Bisphosphonates
- Calcium, Vitamin D and Protein Intake
- DEXA Scan: T-Score and Z-Score
- FRAX Risk Assessment
- Secondary Osteoporosis Causes
- Fracture Prevention and Fall Risk
- Postmenopausal Osteoporosis and Hormone Therapy
- Weight-Bearing and Resistance Exercise
- Calcium
- Vitamin D3
- Arthritis
- Stroke
- Hyperparathyroidism
- Kidney Disease