Lupus Nephritis and Kidney Involvement
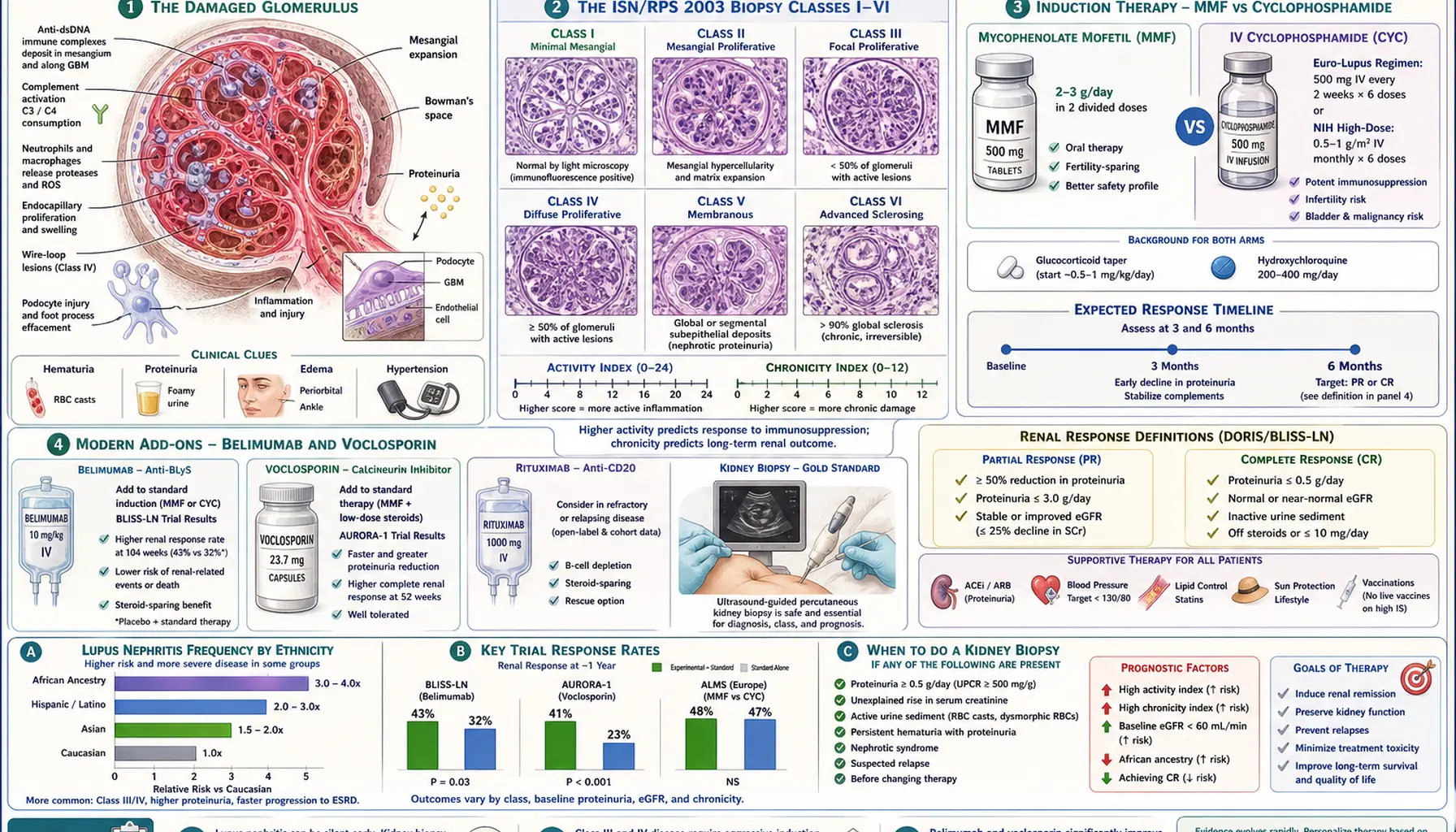
Table of Contents
- What Lupus Nephritis Is
- Why the Kidney Biopsy Is the Whole Game
- The ISN/RPS 2003 Classification (Class I–VI)
- Activity vs Chronicity Indices
- When to Biopsy — The Triggering Numbers
- Induction Therapy: MMF vs Cyclophosphamide
- Steroids — High-Dose vs Moderate
- Belimumab Add-On (BLISS-LN)
- Voclosporin (AURA-LV and AURORA)
- Rituximab in Refractory Disease
- Maintenance Therapy — The Long Haul
- Dialysis and Transplant Outcomes
- Pregnancy With Lupus Nephritis
- Day-to-Day Self-Care
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
What Lupus Nephritis Is
Lupus nephritis is what happens when systemic lupus erythematosus (SLE) attacks the glomeruli — the million tiny filters in each kidney that clean your blood. Immune complexes (antibody clumps, mostly containing anti-dsDNA) deposit in the glomerular basement membrane and mesangium. The body treats those deposits as wounds and pours in complement, neutrophils, and cytokines. The filters scar. Protein leaks into the urine. Waste products build up in the blood.
Roughly 35–60% of lupus patients develop clinically significant kidney involvement at some point, with higher rates in Black, Hispanic, and Asian patients than in white patients. Most cases appear in the first five years after SLE diagnosis — often in the first year — but new nephritis can show up a decade later, which is why every lupus follow-up visit includes a urine check.
Early lupus nephritis is usually silent. You will not feel your kidneys failing. The first sign is almost always a laboratory abnormality: protein in the urine, blood in the urine, a rising creatinine, or edema around the ankles or eyelids. By the time symptoms are obvious — fatigue, shortness of breath, uncontrolled blood pressure, foamy urine — damage may already be substantial. That is the single most important thing to know: lupus nephritis is diagnosed from labs, not from how you feel.
Why the Kidney Biopsy Is the Whole Game
Every serious decision in lupus nephritis — whether to use cyclophosphamide or mycophenolate, how long to treat, what prognosis to quote — depends on the kidney biopsy. Two patients with identical proteinuria and identical creatinine can have completely different biopsies and need completely different regimens. You cannot skip this step, and no blood test substitutes for it.
The procedure is outpatient. An interventional radiologist or nephrologist uses ultrasound guidance to pass a thin needle through the back and take two or three cores of kidney tissue. It takes about 30 minutes; you lie flat for four to six hours afterward to minimize bleeding. Major complications (significant bleeding, need for transfusion) occur in roughly 1–2% of biopsies. You go home the same day in most centers.
The pathologist then classifies the biopsy according to the ISN/RPS 2003 system (International Society of Nephrology / Renal Pathology Society), grading both what class of nephritis you have and how active and how scarred the tissue is. Those two findings determine everything that follows.
The ISN/RPS 2003 Classification (Class I–VI)
Six classes, each with a distinct prognosis and treatment path:
Class I — Minimal mesangial lupus nephritis. Immune deposits visible only by immunofluorescence or electron microscopy. Normal light microscopy. Usually no proteinuria, no hematuria. Excellent prognosis. No lupus-specific kidney treatment needed — manage systemic lupus as usual.
Class II — Mesangial proliferative lupus nephritis. Mesangial hypercellularity plus deposits on light microscopy. Mild proteinuria (usually under 1 g/day) and microscopic hematuria. Good prognosis. Again, no specific nephritis regimen — hydroxychloroquine, ACE inhibitor or ARB for blood-pressure and proteinuria control, and standard SLE therapy.
Class III — Focal lupus nephritis. Active or inactive endocapillary or extracapillary glomerulonephritis involving less than 50% of glomeruli. Proteinuria (often nephrotic), hematuria, sometimes an active urinary sediment with red-cell casts, rising creatinine. This is the first class that demands aggressive immunosuppression. Subdivided into III (A) active, III (A/C) active and chronic, and III (C) chronic sclerosing lesions only.
Class IV — Diffuse lupus nephritis. The same proliferative disease as Class III but involving 50% or more of glomeruli. The most common and the most dangerous class. Heavy proteinuria, red-cell casts, rising creatinine, often hypertension and edema. Untreated or undertreated Class IV progresses to end-stage kidney disease in the majority of patients. With modern induction therapy, ten-year renal survival is now around 80–90%. Further subdivided into IV-S (segmental, lesions affecting part of each involved glomerulus) and IV-G (global, lesions affecting the whole glomerulus).
Class V — Membranous lupus nephritis. Subepithelial immune deposits thicken the glomerular basement membrane. Nephrotic-range proteinuria (often >3.5 g/day), edema, hypoalbuminemia, hyperlipidemia — the nephrotic syndrome. Creatinine typically stays normal for years. Pure Class V has a more indolent course than proliferative disease but raises thrombosis risk and causes protein wasting. Class V often coexists with III or IV (reported as "III + V" or "IV + V"), and when it does, the proliferative component dictates treatment.
Class VI — Advanced sclerosing lupus nephritis. More than 90% of glomeruli globally sclerosed. This is burnt-out disease. Immunosuppression no longer helps the kidneys — it only exposes you to infection risk. The focus shifts to dialysis planning, transplant workup, and controlling lupus activity elsewhere in the body.
Activity vs Chronicity Indices
Classification alone is not enough. The pathologist also scores two indices that tell you how reversible the damage is:
Activity index (0–24). Scores features that respond to immunosuppression: endocapillary hypercellularity, karyorrhexis/fibrinoid necrosis, cellular crescents, subendothelial hyaline deposits (wire loops), interstitial inflammation. A high activity index means "there is inflammation left to quench" — worth treating hard.
Chronicity index (0–12). Scores irreversible damage: glomerular sclerosis, fibrous crescents, tubular atrophy, interstitial fibrosis. A high chronicity index means "much of what is left is scar, not fire." Treatment is less helpful, toxicity risk weighs more, and prognosis is guarded regardless of therapy.
Two Class IV biopsies tell very different stories: Class IV with activity 15, chronicity 2 is the patient you throw the kitchen sink at — this kidney will likely recover. Class IV with activity 4, chronicity 10 is a patient in whom aggressive therapy may do more harm than good. Ask your nephrologist for both numbers when you review the biopsy report.
When to Biopsy — The Triggering Numbers
Any of the following in a lupus patient is a standard indication to biopsy:
- Proteinuria >500 mg/g on a spot urine protein-to-creatinine ratio (equivalent to roughly >500 mg/day on a 24-hour collection), confirmed on a repeat sample.
- Rising serum creatinine without another explanation (not dehydration, not NSAID exposure, not contrast).
- Active urinary sediment — dysmorphic red blood cells, red-cell casts, white-cell casts on a fresh microscopy.
- Unexplained decline in estimated GFR.
- Nephrotic syndrome (edema, albumin <3.5 g/dL, proteinuria >3.5 g/day).
The 2019 EULAR/ERA-EDTA and 2024 KDIGO recommendations all agree: you cannot treat lupus nephritis blind. Even if you are certain clinically, the class and the chronicity index will shape how you treat and how long you treat. The only reasons to defer biopsy are absolute contraindications — uncontrolled bleeding risk, uncontrollable blood pressure, a single functioning kidney in certain situations, or pregnancy beyond the second trimester — and those are negotiable with an experienced operator.
Induction Therapy: MMF vs Cyclophosphamide
Induction means "quench the fire" — get the active inflammation under control in the first 3–6 months. Two first-line regimens have dominated the evidence for twenty years:
Mycophenolate mofetil (MMF). Oral tablet, dosed 2–3 grams per day in divided doses. The Aspreva Lupus Management Study (ALMS) and earlier trials showed MMF is at least as effective as IV cyclophosphamide for induction, with a cleaner side-effect profile — no bladder toxicity, less ovarian suppression, less hair loss. MMF has become the default induction drug for most Class III and Class IV patients, especially women of childbearing age and patients of African or Hispanic ancestry, where MMF may actually outperform cyclophosphamide.
IV cyclophosphamide — Euro-Lupus regimen. The landmark Euro-Lupus Nephritis Trial (Houssiau et al., 2002) showed that low-dose IV cyclophosphamide 500 mg every two weeks for six doses (total 3 grams) worked as well as the older, much more toxic NIH regimen of monthly high-dose pulses for six months followed by quarterly pulses. Ten-year follow-up confirmed durable benefit. Euro-Lupus is the default cyclophosphamide regimen in European populations today, with MMF preferred for non-European ancestries.
Practical comparison your rheumatologist is thinking about:
- Fertility. MMF is teratogenic but not gonadotoxic — fertility returns after stopping. Cyclophosphamide causes dose-dependent ovarian and testicular damage; the Euro-Lupus low dose is far safer than historical regimens but still a consideration in young patients. Egg or sperm banking is reasonable before cyclophosphamide.
- Cost and logistics. MMF is a pill, taken twice daily at home. Cyclophosphamide is six half-day infusion visits.
- Ancestry. Subgroup analyses of ALMS suggest MMF works better than cyclophosphamide in Black and Hispanic patients. This is now a common tiebreaker.
- Severity. Very aggressive disease — rapidly rising creatinine, crescentic glomerulonephritis on biopsy — may still warrant cyclophosphamide (and sometimes plasma exchange) rather than MMF.
Steroids — High-Dose vs Moderate
For decades, induction started with three days of IV methylprednisolone pulses (500–1000 mg daily) followed by oral prednisone 1 mg/kg/day — often 60 mg or more — tapered slowly over a year. The toll was catastrophic: weight gain, diabetes, cataracts, osteoporosis, avascular necrosis of the hips, cushingoid features, mood changes, infection.
The Lupus Nephritis Trial (Rovin et al.) and subsequent real-world studies have changed practice. Current recommendations (EULAR 2023, KDIGO 2024) favor:
- IV methylprednisolone pulses 250–500 mg daily for 3 days (not 1000 mg), which preserves efficacy and reduces infection risk.
- Oral prednisone starting at 0.3–0.5 mg/kg/day (typically 20–30 mg for most adults, not 60), taper to 7.5 mg/day by three months and ideally to 5 mg or less by six months.
- Steroid-sparing add-ons — belimumab or voclosporin — so that the cumulative steroid dose over the first year is meaningfully lower than the historical standard.
The data behind this shift are consistent: lower steroid doses combined with modern steroid-sparing immunosuppression achieve the same renal response rates as the old high-dose regimens with fewer infections and far less long-term damage. If your nephrologist is still quoting 1 mg/kg/day as a starting dose, ask directly whether a lower dose with a belimumab or voclosporin add-on would be appropriate for your biopsy.
Belimumab Add-On (BLISS-LN)
Belimumab is a monoclonal antibody against BLyS (B-lymphocyte stimulator). By depriving autoreactive B cells of a key survival signal, it reduces autoantibody production. It was first approved for non-renal lupus in 2011.
The BLISS-LN trial (Furie et al., NEJM 2020) randomized 448 patients with active Class III, IV, or V lupus nephritis to receive either standard induction (MMF or cyclophosphamide plus steroids) alone or the same regimen plus IV belimumab 10 mg/kg monthly. At two years, 43% of belimumab patients achieved the primary efficacy renal response vs 32% on standard therapy alone, and the risk of renal events or death was significantly lower. The FDA approved belimumab for lupus nephritis in December 2020.
In practice, belimumab is now added to induction and continued through maintenance for most Class III, IV, and V patients who can tolerate it and whose insurance will cover it. It is given IV monthly or as a weekly subcutaneous injection (200 mg). Infection risk is modestly increased, and depression has been reported; serious adverse events are uncommon.
Voclosporin (AURA-LV and AURORA)
Voclosporin is a next-generation calcineurin inhibitor, structurally related to cyclosporine but with more predictable pharmacokinetics (no trough monitoring needed) and a better metabolic profile. It reduces proteinuria both by immunosuppression and by a direct effect on the podocyte cytoskeleton that stabilizes the glomerular filtration barrier.
Two trials drove approval. AURA-LV (phase 2) showed that adding voclosporin 23.7 mg twice daily to MMF plus low-dose steroids roughly doubled the complete renal response rate at 24 weeks. AURORA-1 (phase 3, Rovin et al., Lancet 2021) confirmed the benefit: 41% complete renal response at one year with voclosporin vs 23% with MMF plus steroids alone, with proteinuria dropping faster in the voclosporin arm. AURORA-2 (two-year extension) showed the benefit was durable without a decline in GFR.
FDA approved voclosporin for adult lupus nephritis in January 2021. It is oral, taken twice daily, and does not require drug-level monitoring. Side effects include a small reversible decrease in eGFR early on (reflecting intrarenal hemodynamics, not true damage), hypertension, and occasional hyperkalemia. Voclosporin and belimumab are now the two main "add-on" pillars alongside MMF or cyclophosphamide; which you get often depends on insurance formulary and clinician preference rather than head-to-head evidence, since they have not been directly compared.
Rituximab in Refractory Disease
Rituximab depletes CD20+ B cells. The LUNAR trial did not meet its primary endpoint for lupus nephritis — meaning rituximab is not first-line. But for patients who fail standard induction (persistent heavy proteinuria or worsening creatinine despite MMF or cyclophosphamide plus steroids plus a belimumab or voclosporin add-on), rituximab is a widely used off-label rescue option with reasonable real-world evidence of benefit. Typical dosing: 1 gram IV at day 0 and day 14, sometimes repeated at six months.
Rituximab's main risks are infections (especially if repeated) and hypogammaglobulinemia with chronic use. Check IgG levels annually if you are on long-term B-cell depletion. Your covering rheumatologist will also add Pneumocystis jirovecii prophylaxis (typically trimethoprim-sulfamethoxazole) during periods of intensive immunosuppression.
For a deeper look at all three biologic options, see the biologics page.
Maintenance Therapy — The Long Haul
Once induction has quieted the inflammation (proteinuria falling toward 500 mg/g, creatinine stable, inactive sediment), treatment shifts to maintenance — lower-intensity therapy to prevent relapse. Lupus nephritis relapses in roughly 30–40% of patients, and each relapse adds scar. Maintenance is not optional.
Typical maintenance regimen:
- MMF 1–2 g/day (or azathioprine 2 mg/kg/day if MMF is not tolerated or pregnancy is planned).
- Hydroxychloroquine 5 mg/kg/day actual body weight, max 400 mg/day — everyone with lupus, including nephritis, should be on this unless truly intolerant. See the hydroxychloroquine page for why.
- Low-dose prednisone — 5 mg or less per day, with the goal of eventually stopping.
- Continued belimumab or voclosporin if started in induction.
- ACE inhibitor or ARB for blood pressure and proteinuria control, targeting BP <130/80 mmHg.
- SGLT2 inhibitor (dapagliflozin or empagliflozin) — increasingly added in patients with residual proteinuria or declining eGFR, based on the DAPA-CKD and EMPA-KIDNEY trials, which included patients with non-diabetic proteinuric kidney disease.
Maintenance duration is at least 3–5 years after complete renal response. Some patients stay on low-dose maintenance indefinitely; others taper off with close monitoring. Stopping too soon predicts relapse. Your nephrologist and rheumatologist will watch urine protein, sediment, creatinine, complement (C3, C4), and anti-dsDNA every 1–3 months through induction and every 3–6 months in maintenance.
Dialysis and Transplant Outcomes
Despite modern therapy, roughly 10–20% of lupus nephritis patients progress to end-stage kidney disease within ten years, with higher rates in Black and Hispanic patients and in those whose disease was caught late. If your kidney function declines toward end-stage, the news is better than it used to be:
- Dialysis. Lupus patients on hemodialysis or peritoneal dialysis have similar survival to non-lupus dialysis patients. Lupus activity often decreases on dialysis (the "burned-out lupus" phenomenon), and many patients can taper immunosuppression substantially. Flares still occur, so monitoring continues.
- Transplant. Kidney transplant is the best long-term option. Lupus recurs in the transplanted kidney in only 2–10% of cases, and graft survival is similar to transplants for other causes of end-stage disease. Most centers wait 3–6 months of clinical quiescence on dialysis before listing. Living-donor transplant is strongly preferred when available — better outcomes, shorter wait, and no dialysis time.
- Antiphospholipid antibodies complicate transplant because of thrombosis risk in the graft vessels. Testing for lupus anticoagulant, anti-cardiolipin, and anti-beta-2-glycoprotein is standard during transplant workup.
For the broader picture of what kidney disease means day-to-day — dialysis choices, transplant workup, diet — see kidney disease.
Pregnancy With Lupus Nephritis
Pregnancy and lupus nephritis need careful choreography. The rules:
- Wait for quiescence. Conception should happen after at least 6 months of stable disease with proteinuria <500 mg/g, normal creatinine, and no active sediment. Pregnancy conceived during active nephritis has much higher rates of preeclampsia, preterm birth, fetal loss, and permanent maternal kidney damage.
- Switch medications first. MMF and cyclophosphamide are teratogenic and must be stopped at least 3 months before conception. The safe substitutes for maintenance in pregnancy are azathioprine, hydroxychloroquine, and tacrolimus. Low-dose prednisone is acceptable.
- Hydroxychloroquine continues through pregnancy — it reduces flare risk, lowers preeclampsia risk, and may protect against congenital heart block in anti-Ro/SSA-positive mothers.
- Low-dose aspirin 81–150 mg daily from weeks 12–16 onward reduces preeclampsia risk, which is already elevated in lupus nephritis.
- Monitoring is monthly in the first two trimesters and every two weeks in the third, with urine protein-to-creatinine ratios, creatinine, complement, and anti-dsDNA. Distinguishing lupus flare from preeclampsia late in pregnancy is tricky — both cause hypertension and proteinuria — and sometimes requires consultation between maternal-fetal medicine, nephrology, and rheumatology.
- Delivery planning. Most pregnancies in quiescent lupus nephritis deliver near term. Active nephritis or preeclampsia often forces preterm delivery. Cesarean rates are higher, but vaginal delivery is possible.
See the pregnancy and lupus page for the full medication swap list, antiphospholipid considerations, and neonatal lupus screening.
Day-to-Day Self-Care
Medication is most of the battle, but a handful of daily habits genuinely move the needle:
- Blood pressure discipline. Buy a home cuff. Target <130/80 mmHg. Every 10 mmHg of systolic reduction slows kidney decline measurably.
- Sodium restriction. Aim for <2 g/day. This improves both blood pressure and the effectiveness of your ACE inhibitor or ARB.
- Protein intake — moderate, not low. Roughly 0.8 g/kg/day for most patients with reduced kidney function; do not go lower unless a dietitian specifically advises.
- NSAIDs are off-limits once you have active or chronic kidney disease. Ibuprofen, naproxen, celecoxib, and diclofenac all cut renal blood flow. Use acetaminophen, heat/ice, or physical therapy instead.
- Sun protection. UV drives lupus flares, which drive nephritis flares. See the photoprotection page.
- Vaccinations. Annual influenza, pneumococcal (PCV20 or PCV15 plus PPSV23), shingles (recombinant, two doses), and HPV if age-eligible. Avoid live vaccines while on high-dose immunosuppression.
- Bone protection. Vitamin D 1000–2000 IU daily, calcium 1000 mg daily from food where possible, a DEXA scan at baseline and every 1–2 years on steroids. A bisphosphonate is reasonable after about three months on ≥7.5 mg/day of prednisone.
- Infection vigilance. Fever on immunosuppression is not a "watch and wait" problem. Call your team the same day.
Key Research Papers
- Weening JJ, et al. The classification of glomerulonephritis in systemic lupus erythematosus revisited. J Am Soc Nephrol. 2004. (ISN/RPS 2003 classification.)
- Houssiau FA, et al. Immunosuppressive therapy in lupus nephritis: the Euro-Lupus Nephritis Trial, a randomized trial of low-dose versus high-dose intravenous cyclophosphamide. Arthritis Rheum. 2002.
- Appel GB, et al. Mycophenolate mofetil versus cyclophosphamide for induction treatment of lupus nephritis (ALMS). J Am Soc Nephrol. 2009.
- Furie R, et al. Two-year, randomized, controlled trial of belimumab in lupus nephritis (BLISS-LN). N Engl J Med. 2020.
- Rovin BH, et al. Efficacy and safety of voclosporin versus placebo for lupus nephritis (AURORA-1): a double-blind, randomised, multicentre, placebo-controlled, phase 3 trial. Lancet. 2021.
- Rovin BH, et al. A randomized, controlled double-blind study comparing the efficacy and safety of dose-ranging voclosporin with placebo in achieving remission in patients with active lupus nephritis (AURA-LV). Kidney Int. 2019.
- Fanouriakis A, et al. 2019 update of the joint European League Against Rheumatism and European Renal Association-European Dialysis and Transplant Association (EULAR/ERA-EDTA) recommendations for the management of lupus nephritis. Ann Rheum Dis. 2020.
- KDIGO 2024 Clinical Practice Guideline for the Management of Lupus Nephritis. Kidney Int. 2024.
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on lupus nephritis classification, therapy, and outcomes:
- Lupus nephritis ISN/RPS classification
- MMF vs cyclophosphamide induction for lupus nephritis
- Euro-Lupus Nephritis Trial long-term outcomes
- Belimumab in lupus nephritis (BLISS-LN)
- Voclosporin AURORA trials in lupus nephritis
- Rituximab in refractory lupus nephritis
- Kidney transplant outcomes in lupus nephritis
- Pregnancy outcomes in lupus nephritis
- Activity and chronicity indices in lupus nephritis
- SGLT2 inhibitors in proteinuric kidney disease
Connections
- Kidney Disease
- Lupus Overview
- Lupus and Pregnancy
- Biologics: Belimumab, Anifrolumab and Rituximab
- Hydroxychloroquine and Antimalarials
- Neuropsychiatric Lupus
- ACR-EULAR 2019 Classification Criteria
- Lupus-Sjogren's Overlap
- ANA, dsDNA and Lupus Autoantibodies
- Cutaneous Lupus and Photoprotection
- Sjogren's Syndrome
- Glomerulonephritis
- Preeclampsia
- Creatinine
- eGFR
- Hypertension