Cutaneous Lupus and Photoprotection
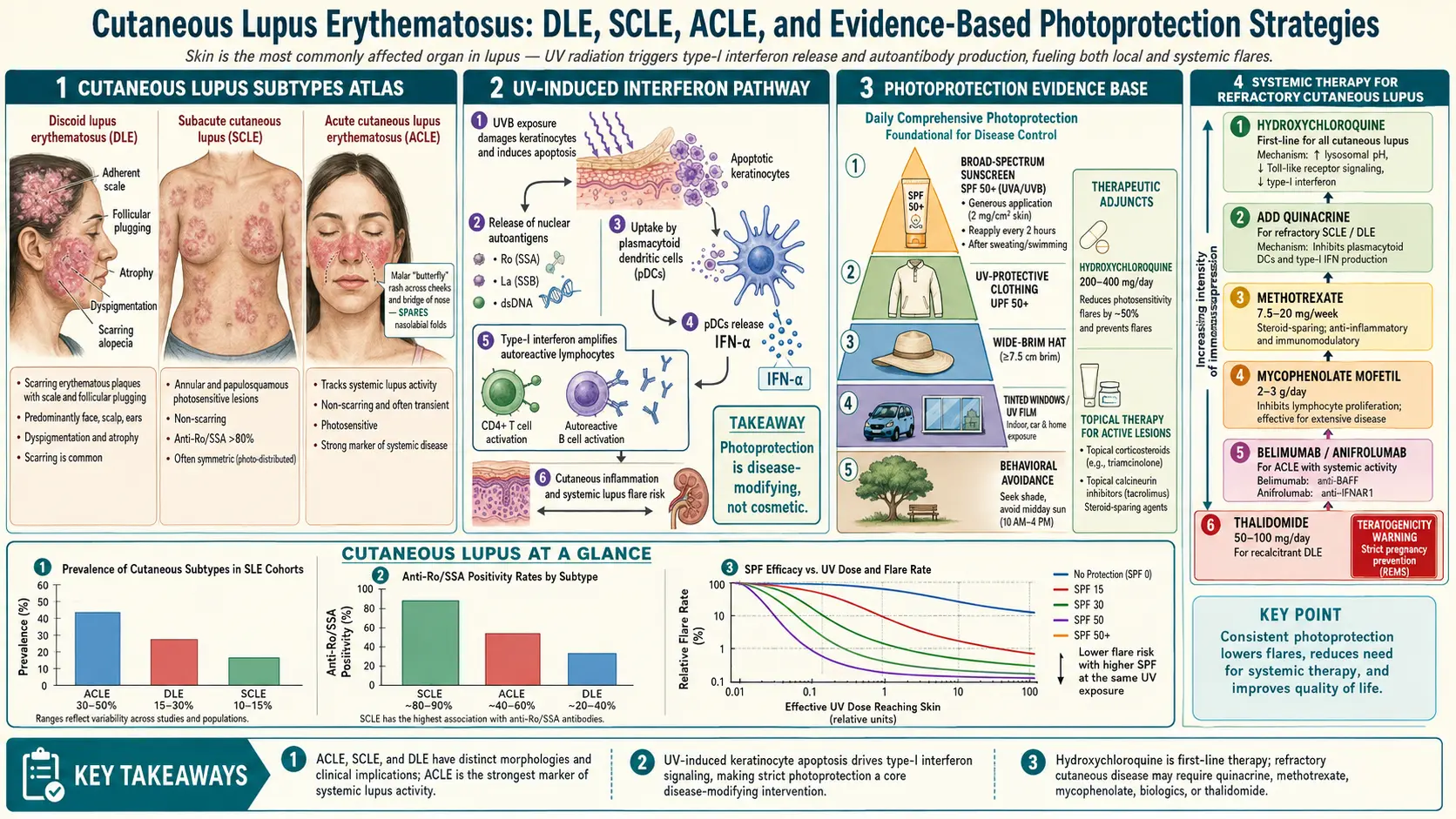
Table of Contents
- Why the Skin Is Ground Zero in Lupus
- Acute Cutaneous Lupus (ACLE) — The Butterfly Rash
- Subacute Cutaneous Lupus (SCLE)
- Chronic Cutaneous Lupus — Discoid and Beyond
- Non-Specific Lesions: Bullous, Panniculitis, Tumidus
- How UV Light Triggers Lupus Skin
- Sunscreen — Broad-Spectrum, Visible Light, Iron Oxides
- UPF Clothing, Tinted Window Film, Physical Measures
- Topical Steroids and Calcineurin Inhibitors
- Hydroxychloroquine and Quinacrine
- Thalidomide, Lenalidomide, and Refractory DLE
- Anifrolumab and the Interferon Pathway
- Managing Scars, Hair Loss, and Hyperpigmentation
- A Realistic Daily Routine
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
Why the Skin Is Ground Zero in Lupus
Roughly 70–80% of people with systemic lupus erythematosus (SLE) develop skin involvement at some point, and for many of them the rash was the first clue anything was wrong. A separate group has lupus that stays in the skin — cutaneous lupus erythematosus (CLE) without ever crossing the threshold into systemic disease. Understanding which form you have matters because the prognosis, workup, and treatment priorities are different.
Dermatologists split lupus skin disease into three buckets based on how long the lesions last and how deep the damage goes: acute (ACLE), subacute (SCLE), and chronic (CCLE). On top of those, there is a grab-bag of non-specific lesions — bullous lupus, lupus panniculitis (profundus), and lupus tumidus — that don't fit neatly into the three main categories but still belong to the lupus family. What unites almost all of them is photosensitivity: sunlight, fluorescent light, and even the visible light from a laptop screen can trigger flares that last weeks.
This page walks through each subtype, then spends serious time on photoprotection — the single most effective, under-used intervention in lupus skin care — followed by topical and systemic treatments that actually move the needle.
Acute Cutaneous Lupus (ACLE) — The Butterfly Rash
ACLE is the classic lupus rash most people recognize from medical textbooks: the malar or “butterfly” rash that splays across the cheeks and bridge of the nose while sparing the nasolabial folds (the creases running from nose to mouth corners). The rash is flat or slightly raised, pink to violaceous red, and often feels warm. It does not usually itch or scale, which helps distinguish it from rosacea or seborrheic dermatitis.
A key feature is that ACLE tracks the sun. Most flares follow a sunny weekend, a beach day, or even a long drive with an unprotected left arm and face. Lesions appear within hours to a few days and typically resolve within weeks — without scarring — once the trigger stops and treatment begins.
There is also a generalized form of ACLE: a widespread maculopapular rash on the trunk, arms, and V of the neck. It looks like a drug reaction or a viral exanthem and is sometimes mistaken for one.
The clinical bottom line: ACLE almost always means you have systemic lupus. Unlike SCLE and discoid lupus, ACLE rarely occurs as a purely skin-limited disease. When a butterfly rash is confirmed on biopsy, a full SLE workup (ANA, anti-dsDNA, complement, CBC, urinalysis) is mandatory. See the lupus autoantibodies guide for what those numbers mean.
Subacute Cutaneous Lupus (SCLE)
SCLE sits in the middle of the spectrum. About 50% of SCLE patients meet criteria for SLE, but the systemic disease tends to be milder — joints, skin, and fatigue rather than kidneys or brain. The hallmark is extreme photosensitivity, usually on the upper back, chest, shoulders, and outer arms (the classic sun-exposed “shawl” distribution). The face can be involved but is often spared.
SCLE comes in two morphologies that can occur in the same patient:
- Annular SCLE — ring-shaped lesions with a raised red border and a pale, often slightly scaly center. They can merge into arcs and polycyclic patterns.
- Papulosquamous SCLE — red, scaly plaques that look a lot like psoriasis or eczema. Dermatologists who aren't thinking about lupus frequently misdiagnose this form for years.
About 70–90% of SCLE patients are anti-Ro/SSA positive, and many are also anti-La/SSB positive — the same antibodies central to Sjögren's syndrome. This matters for two reasons: (1) SCLE patients often develop dry eyes and dry mouth, and (2) pregnant SCLE patients with anti-Ro are at risk for neonatal lupus and congenital heart block, so the pregnancy plan needs an anti-Ro-aware obstetrician (see lupus and pregnancy).
Drug-induced SCLE is real and common. More than 100 medications have been reported to trigger it; the top offenders are hydrochlorothiazide (a very common blood-pressure drug), terbinafine (antifungal), proton-pump inhibitors, ACE inhibitors, calcium channel blockers, and TNF inhibitors. Onset is weeks to months after starting the drug. If your SCLE flare coincides with a new prescription, bring the full medication list to your rheumatologist — stopping the offender usually clears the rash over 6–12 weeks.
SCLE lesions generally heal without scars but can leave long-lasting hypopigmentation — pale patches that last months to years and stand out against tanned skin. Patients with darker skin tones may find these cosmetically distressing; camouflage makeup and time are usually the answer, because the pigment eventually returns.
Chronic Cutaneous Lupus — Discoid and Beyond
The chronic cutaneous category is dominated by discoid lupus erythematosus (DLE), the most common scarring form of cutaneous lupus. Discoid lesions start as red, scaly, coin-shaped plaques, usually on the face, scalp, and ears. Over months they develop three signature features:
- Follicular plugging — small keratin plugs in hair follicles that feel like a bed of nails when you run a finger across the plaque.
- Atrophy and scarring — the central plaque sinks, thins, and loses its normal skin markings.
- Dyspigmentation — the center becomes pale (hypopigmented) while the border darkens (hyperpigmented). In darker skin tones this contrast is dramatic and emotionally hard.
When DLE hits the scalp, the result is permanent, scarring alopecia — the hair follicle itself is destroyed, so no amount of minoxidil or transplant will bring it back. Early treatment is the only way to save the follicles, which is why a new scaly scalp patch in a lupus patient is a dermatology emergency, not a cosmetic concern.
DLE comes in two distributions: localized (head and neck only) and generalized (below the neck, usually on arms and upper trunk). Generalized DLE is more likely to be associated with systemic disease.
The SLE progression question. Patients with isolated DLE ask whether their skin disease will turn into systemic lupus. The data are reassuring but not zero: about 5–10% of DLE patients eventually progress to SLE, usually within the first few years after diagnosis. Risk factors include generalized (below-the-neck) lesions, a positive ANA at high titer, low complement, arthralgias, and cytopenias. Isolated, localized DLE with a negative ANA almost never progresses. Annual rheumatology follow-up with bloodwork is still worth it.
Other chronic forms include hypertrophic (verrucous) DLE, which looks warty and can mimic squamous cell carcinoma, and chilblain lupus, which produces painful, cold-induced purple nodules on fingers and toes — overlapping clinically with Raynaud's but with biopsy-proven lupus pathology.
Non-Specific Lesions: Bullous, Panniculitis, Tumidus
A handful of lupus skin manifestations don't fit the acute/subacute/chronic grid cleanly:
- Bullous lupus erythematosus — tense fluid-filled blisters on sun-exposed skin, driven by autoantibodies against type VII collagen (the same target as epidermolysis bullosa acquisita). Rare but dramatic; usually responds beautifully to dapsone.
- Lupus panniculitis (lupus profundus) — deep, painful, firm nodules in the subcutaneous fat, most often on the upper arms, thighs, buttocks, and face. When they heal they leave depressed, saucer-shaped scars that can be cosmetically severe. Hydroxychloroquine is first-line.
- Lupus tumidus (lupus erythematosus tumidus) — juicy, smooth, pink-to-violet plaques on sun-exposed skin without scale, follicular plugging, or scarring. It's the most exquisitely photosensitive form of lupus skin — some patients flare after a single lunch outside — but it also has the best prognosis, heals without scarring, and rarely associates with systemic disease.
How UV Light Triggers Lupus Skin
Sunlight triggers lupus through at least three overlapping mechanisms. Understanding them makes photoprotection choices easier.
1. UV induces keratinocyte apoptosis. When ultraviolet light hits skin cells, it damages DNA and triggers programmed cell death. Normally, dying cells are swept up quietly by macrophages. In lupus, clearance is slow, and dying cells spill their contents — including nuclear proteins like Ro/SSA — onto the cell surface and into the local tissue. Autoantibodies grab these exposed antigens and call in inflammation.
2. UV drives a type I interferon response. Damaged skin cells release interferon-alpha and -beta, cytokines that are elevated in lupus blood at baseline and that orchestrate most of the downstream skin inflammation. This is the pathway that anifrolumab blocks.
3. Visible light and near-infrared also matter. For decades, photoprotection was designed around UVB (sunburn wavelengths, 290–320 nm) and UVA (320–400 nm). Newer studies show that visible light (400–700 nm) also triggers lupus flares, especially in patients with darker skin and in SCLE. This is why standard clear sunscreens — which block UV but let visible light through — sometimes fail to protect lupus skin, and why tinted sunscreens with iron oxides are a game-changer (next section).
Sunscreen — Broad-Spectrum, Visible Light, Iron Oxides
Sunscreen in lupus is not a cosmetic; it is medicine. A realistic product and a realistic routine:
- Choose broad-spectrum SPF 50+. “Broad-spectrum” means UVA and UVB. SPF refers mostly to UVB protection; a high SPF with strong UVA coverage (look for the PA++++ rating or UVA logo in a circle on European products) is the baseline.
- Look for mineral filters. Zinc oxide and titanium dioxide block a broader range of wavelengths than many chemical filters and are less irritating on inflamed lupus skin. A combination of mineral plus modern chemical filters (tinosorb, avobenzone well stabilized) is often the most elegant.
- Insist on a tinted formulation with iron oxides. This is the single most important upgrade in the last decade. Iron oxides are the pigments that make a sunscreen look beige or brown; they are the only common sunscreen ingredient that blocks visible light. For SCLE, tumidus, melasma-like hyperpigmentation, and anyone with skin of color, tinted mineral sunscreen dramatically outperforms clear sunscreen. Brands that explicitly include iron oxides include ISDIN Eryfotona Ageless/Actinica, La Roche-Posay Anthelios Tinted, EltaMD UV Elements, and Avène Mineral Tinted.
- Apply enough. Sunscreen studies use 2 mg/cm², which works out to a full teaspoon for the face and neck alone. Most people apply a quarter of that and get a quarter of the labeled SPF.
- Reapply every 2 hours outdoors, and after swimming or sweating. Set a phone alarm if you need one.
- Use sunscreen indoors near windows. Ordinary window glass blocks UVB but lets UVA and visible light through. A sunlit desk, car commute, or kitchen window can drive a flare.
UPF Clothing, Tinted Window Film, Physical Measures
Sunscreen alone is never enough for active cutaneous lupus. The skin you can cover with fabric is protected essentially 24/7 and without reapplication. A practical wardrobe:
- UPF 50+ long-sleeve shirts. Coolibar, Solbari, UV Skinz, Uniqlo UV Cut, and Columbia Omni-Shade all sell breathable, lightweight options. A UPF rating of 50 blocks 98% of UV.
- Wide-brim hats. At least a 3-inch (7.5 cm) brim all the way around — baseball caps leave the ears and neck exposed. Consider a neck drape for the scalp-DLE patient.
- Wraparound UV-blocking sunglasses. Protects periocular skin and reduces photosensitive headache.
- UV gloves for driving. The left arm and left cheek of drivers in right-hand-traffic countries (and the right arm in the UK/Japan/Australia) take a disproportionate UV dose through the driver's window.
Tinted window film is underused and under-prescribed. High-quality ceramic or nano-carbon films block 99% of UVA/UVB and reduce visible light by 20–70% depending on shade. Applied to home windows (especially south- and west-facing), car side windows, and office glass, film turns high-risk environments into safe ones. U.S. state laws regulate car-window tint darkness, but a medical exemption for lupus is available in most states — your rheumatologist or dermatologist can write a letter specifying the reason. Professional installation runs $200–$800 for a car and $5–$15 per square foot for home windows. For patients with severe photosensitivity, this is often the best dollar they spend.
Don't forget indoor fluorescent and some LED lighting emit low-level UV. If work involves long hours under harsh fluorescents and flares track the work week, swap bulbs for filtered LEDs or add UV-blocking sleeves to tubes.
Topical Steroids and Calcineurin Inhibitors
For active lesions, topical anti-inflammatories are first-line. The decision is which potency and where.
- Low-potency topical steroids (hydrocortisone 2.5%, desonide) — face, neck, skin folds. Safe for short courses (2–4 weeks) to calm ACLE flares.
- Mid-potency steroids (triamcinolone 0.1%, mometasone) — trunk, arms, legs.
- High-potency steroids (clobetasol 0.05%, halobetasol) — scalp DLE, thick discoid plaques, palms/soles. Potent steroids thin facial skin quickly; avoid on the face unless a dermatologist specifically pulses them.
- Intralesional triamcinolone — injected directly into stubborn discoid plaques every 4–6 weeks. Dramatic for isolated lesions that resist topicals.
- Topical calcineurin inhibitors (tacrolimus 0.1% ointment, pimecrolimus 1% cream) — these non-steroid immunomodulators are a huge win for facial and intertriginous lupus because they don't cause skin atrophy. Used long-term on the face without the thinning, telangiectasia, or perioral dermatitis that steroids cause. Apply twice daily; a mild burning sensation in the first few days is common and subsides.
Hydroxychloroquine and Quinacrine
For any cutaneous lupus that isn't controlled by topicals — and certainly for any patient with SLE — hydroxychloroquine (HCQ, brand name Plaquenil) is first-line systemic therapy. It is the single most important drug in lupus, with evidence for reducing flares, protecting the kidneys, reducing thrombosis risk, and improving survival. See the hydroxychloroquine guide for dosing, monitoring, and the retinal toxicity question.
For cutaneous disease specifically, dosing is the same (5 mg/kg of actual body weight per day, capped around 400 mg). It takes 6–12 weeks for skin response, which is frustrating when lesions are actively spreading; bridge with topicals and, if severe, a short course of prednisone.
Roughly one-third of cutaneous lupus patients don't respond adequately to HCQ alone. The next step, classically, is adding quinacrine (mepacrine) — another antimalarial from the same era as HCQ and chloroquine. Quinacrine is not sold by standard U.S. pharmacies; it has to be obtained from a compounding pharmacy (common dose 100 mg daily). It does not carry the retinal toxicity risk of HCQ, so the ophthalmology monitoring burden doesn't double. The main side effect is yellow-orange skin discoloration that reverses when the drug is stopped. The HCQ-plus-quinacrine combination rescues many patients who had been considered antimalarial failures.
If hydroxychloroquine plus quinacrine still fails, the options are to switch HCQ to chloroquine (slightly more potent, higher retinal risk), or to move up to immunosuppressants (methotrexate, mycophenolate, azathioprine) or to biologics.
Thalidomide, Lenalidomide, and Refractory DLE
For scarring discoid lupus that has resisted antimalarials, topicals, and standard immunosuppressants, thalidomide produces near-miraculous clearance in 70–80% of patients, often within weeks. Its mechanism involves inhibition of TNF-alpha and modulation of T-cell responses. The catches are serious:
- Severe teratogen. Thalidomide caused one of the worst pharmaceutical disasters of the 20th century. In the U.S. it is prescribed only through the THALOMID REMS program with mandatory monthly pregnancy testing for patients of childbearing potential and strict contraception requirements.
- Peripheral neuropathy. Up to 25–50% of long-term users develop dose-dependent sensory neuropathy — numbness, tingling, and burning in the feet — that may not fully reverse after stopping the drug. Regular neurological exams and nerve conduction studies are warranted.
- Sedation, constipation, thrombosis risk.
Because of the neuropathy and teratogenicity, many dermatologists now use lenalidomide (a thalidomide analog originally developed for multiple myeloma) at 5–10 mg daily instead. Lenalidomide has a much lower neuropathy risk and similar efficacy in refractory DLE, though it remains teratogenic and expensive.
These drugs are reserved for patients with relentless scarring disease — not for mild cosmetic concerns — and are always prescribed by a dermatologist or rheumatologist experienced with them.
Anifrolumab and the Interferon Pathway
Anifrolumab (Saphnelo) is a monoclonal antibody that blocks the type I interferon receptor. It was approved by the FDA in 2021 for moderate-to-severe SLE, and the pivotal TULIP-1 and TULIP-2 trials showed that skin scores (CLASI) improved dramatically in patients with active cutaneous disease — more than 50% of anifrolumab patients achieved a 50% reduction in CLASI activity score, compared to about 25% on placebo. For SLE patients whose dominant active feature is skin, anifrolumab is often the biologic of choice.
Dosing is a monthly intravenous infusion (300 mg). The main risks are infections (particularly viral upper respiratory) and herpes zoster reactivation; shingles vaccination before starting is strongly recommended. Anifrolumab has not been studied enough in pregnancy to recommend during conception. See the lupus biologics page for full comparisons with belimumab and rituximab.
Belimumab (Benlysta), a BLyS inhibitor, has less dramatic skin-specific data but also helps cutaneous disease as part of overall SLE control. For purely skin-limited CLE without systemic activity, neither biologic is formally approved, but off-label use is reasonable in severe refractory cases.
Managing Scars, Hair Loss, and Hyperpigmentation
Once DLE scars or SCLE pigment changes are set, no drug reverses them quickly — but several interventions help.
- Hyperpigmentation (dark spots) — topical azelaic acid 15–20%, tranexamic acid (topical or oral), niacinamide, and hydroquinone 4% (short courses, under dermatologist supervision) fade dark macules over 3–6 months. Daily SPF 50+ tinted sunscreen is non-negotiable — without it, nothing works.
- Hypopigmentation (pale spots) — harder to treat. Pigment sometimes returns on its own over a year or more. Excimer laser and narrow-band UVB (carefully targeted, under dermatologist supervision only — never do home phototherapy for lupus skin) can sometimes restore melanocyte function.
- Scarring alopecia — if follicles are destroyed, no drug regrows hair. Options are hair systems (wigs and toppers), scalp micropigmentation, and — once disease is quiescent for at least two years — hair transplantation into scarred areas, though grafts can be poorly vascularized. Minoxidil doesn't help scarred follicles.
- Panniculitis scars (depressions in the face or arm) — hyaluronic acid fillers can cosmetically raise sunken areas. Autologous fat transfer is another option. Only attempt once disease has been stable for at least 6–12 months, because active panniculitis can destroy injected material.
- Makeup camouflage — waterproof high-coverage brands (Dermablend, Kevyn Aucoin, Estée Lauder Double Wear) cover both red active lesions and pigment changes. Color-correcting green or peach primers neutralize red and brown tones before foundation. This is not vanity; it is daily functioning for many patients.
A Realistic Daily Routine
What does photoprotection actually look like on a Tuesday? Something like this:
- Morning: gentle cleanser; bland moisturizer; tinted broad-spectrum mineral sunscreen SPF 50+ with iron oxides (full teaspoon for face, neck, ears, V of chest, and tops of hands).
- Before leaving the house: UPF 50 long-sleeve layer, wide-brim hat, wraparound sunglasses, UV driving gloves if going to the car.
- Every two hours outside: reapply sunscreen — a mineral powder or stick is practical over makeup.
- At work: sit more than an arm's length from sunlit windows; use tinted window film or blinds on the worst offenders.
- Evening: gentle cleanser to remove sunscreen (double-cleanse if wearing a heavy mineral product); topical medication (tacrolimus to active patches, clobetasol to thick scalp plaques per dermatologist); bland moisturizer; HCQ with dinner to reduce nausea.
- Monthly: check sunscreen and medication supplies; update the hat or UPF shirt if the elastic is dying; re-schedule dermatology follow-up every 3–6 months during active disease.
- Annually: eye exam (uveitis screen, HCQ retinal screening by year five), vitamin D level (aggressive sun avoidance causes deficiency — most lupus patients need 1000–2000 IU daily), full skin check, and lupus bloodwork per rheumatology.
This is a lot. It gets easier once it becomes habit — the sunscreen and hat are just what you do before leaving the house, the same way brushing your teeth is. Patients who push through the first three months of new routines almost always report fewer flares within a year.
Key Research Papers
- Okon LG, Werth VP. Cutaneous lupus erythematosus: diagnosis and treatment. Best Pract Res Clin Rheumatol. 2013. (Foundational review of CLE classification and management.)
- Morand EF, et al. Trial of anifrolumab in active systemic lupus erythematosus (TULIP-2). N Engl J Med. 2020.
- Furie R, et al. Type I interferon inhibitor anifrolumab in active systemic lupus erythematosus (TULIP-1). Lancet Rheumatol. 2019.
- Kuhn A, et al. Photoprotection in patients with cutaneous lupus erythematosus. Br J Dermatol. 2015.
- Kohli I, et al. Effect of visible light on the skin and the role of iron oxides in sunscreen protection. J Invest Dermatol. 2020.
- Cortés-Hernández J, et al. Thalidomide in the treatment of refractory cutaneous lupus erythematosus. Br J Dermatol. 2012.
- Fennira F, et al. Lenalidomide for refractory chronic and subacute cutaneous lupus erythematosus. J Am Acad Dermatol. 2016.
- Fanouriakis A, et al. 2019 update of the EULAR recommendations for the management of systemic lupus erythematosus. Ann Rheum Dis. 2019.
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on cutaneous lupus subtypes, photoprotection, and treatment:
- Cutaneous lupus erythematosus classification
- Discoid lupus and scarring alopecia
- Drug-induced subacute cutaneous lupus
- Cutaneous lupus photoprotection and iron oxide sunscreen
- Visible light and cutaneous lupus
- Hydroxychloroquine plus quinacrine in cutaneous lupus
- Thalidomide and lenalidomide for refractory discoid lupus
- Anifrolumab and cutaneous lupus CLASI response
Connections
- Lupus Overview
- Lupus and Pregnancy
- Biologics: Belimumab, Anifrolumab and Rituximab
- ANA, dsDNA and Lupus Autoantibodies
- Hydroxychloroquine and Antimalarials
- Neuropsychiatric Lupus
- ACR-EULAR 2019 Classification Criteria
- Lupus-Sjogren's Overlap
- Lupus Nephritis and Kidney Involvement
- Raynaud's Disease
- Sjogren's Syndrome
- Vitamin D3
- Alopecia
- Full Body MRI
- Psoriasis