ANA, dsDNA, and Lupus Autoantibodies
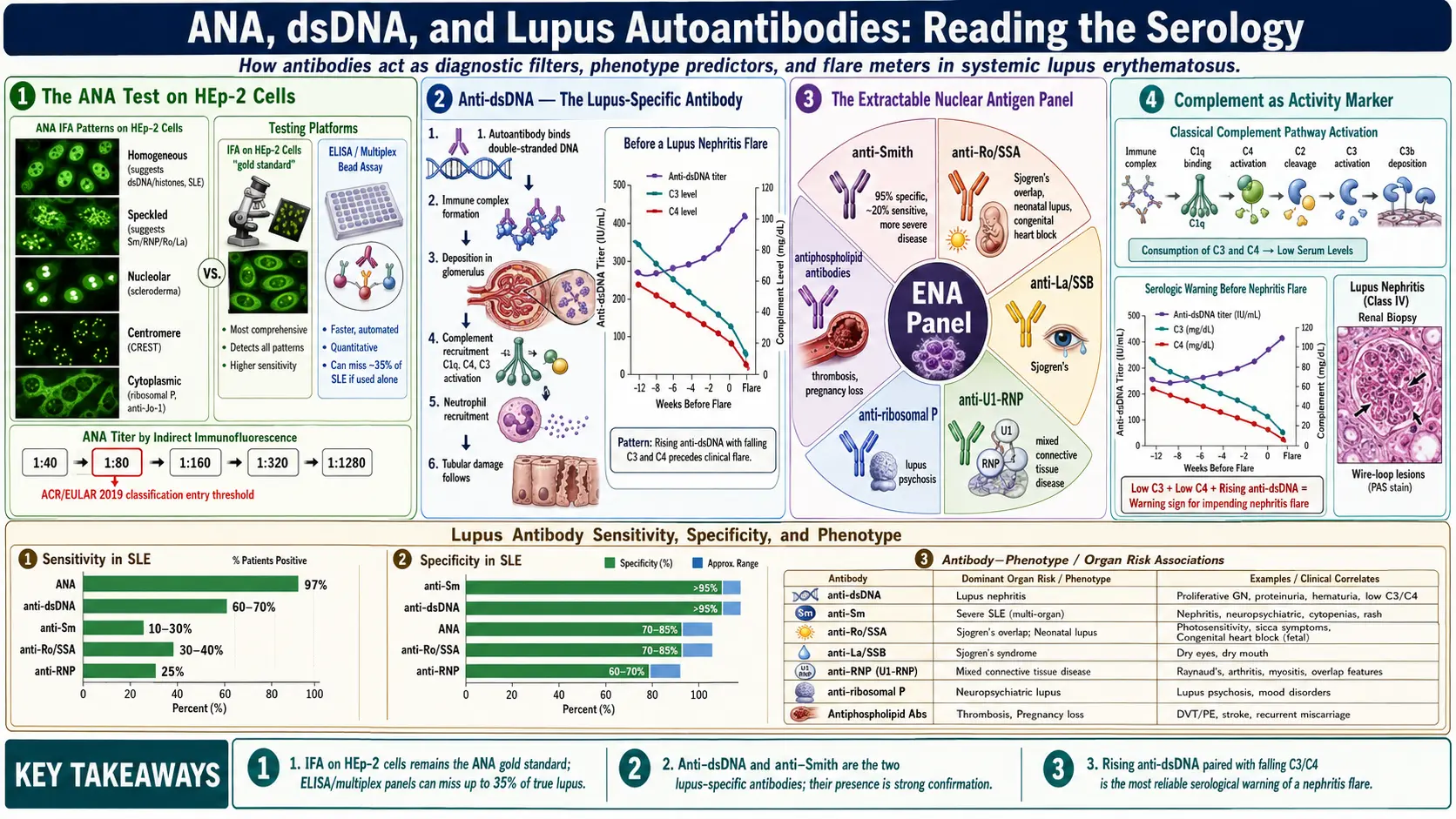
Table of Contents
- Why Antibodies Matter in Lupus
- The ANA Test — IFA vs. ELISA/Multiplex
- Reading Your ANA: Titer and Pattern
- Anti-dsDNA — The Lupus-Specific Antibody
- Anti-Smith (Sm) — Highly Specific, Often Missed
- Anti-Ro/SSA and Anti-La/SSB
- Ribosomal P and Neuropsychiatric Lupus
- U1-RNP and Mixed Connective Tissue Disease
- The Antiphospholipid Panel
- Complement: C3, C4, and CH50 as Activity Markers
- How Antibodies Predict Your Lupus Phenotype
- False Positives and Borderline Results
- A Practical Ordering Strategy
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
Why Antibodies Matter in Lupus
Systemic lupus erythematosus (SLE) is, at heart, a disease of broken self-tolerance. The immune system begins producing autoantibodies — antibodies directed at pieces of your own cells, most of them sitting inside the nucleus. These antibodies bind their targets, form immune complexes, deposit in tissues, and trigger the inflammation you feel as joint pain, rashes, kidney damage, fatigue, and brain fog.
For a patient, the antibody panel is more than a diagnostic checkbox. It acts as:
- A diagnostic filter — a negative ANA in a person not on biologics makes SLE vanishingly unlikely.
- A phenotype predictor — which organs are most at risk (kidneys, skin, brain, blood vessels, the fetus).
- A disease-activity meter — anti-dsDNA and complement levels often rise and fall with flares.
- A pregnancy and family-planning tool — anti-Ro/SSA and antiphospholipid antibodies carry specific risks to a fetus.
This article walks through each antibody in plain language: what the test does, how to read the number, and what it means for you.
The ANA Test — IFA vs. ELISA/Multiplex
The antinuclear antibody (ANA) test is the front door. Over 97% of SLE patients are ANA-positive at some point in their disease course, which is why rheumatologists treat a persistently negative ANA as strong evidence against lupus. But not every ANA test is created equal.
Indirect Immunofluorescence (IFA) — the Gold Standard
A drop of your diluted serum is placed on a slide of HEp-2 cells (a human cell line). If ANAs are present, they latch onto nuclear targets. A fluorescent-tagged secondary antibody is added, and a technician reads the slide under a fluorescence microscope, recording two things: the titer (how many dilutions it takes before fluorescence disappears) and the pattern (homogeneous, speckled, nucleolar, centromere, and several others).
IFA is the method endorsed by the American College of Rheumatology (ACR) as the reference standard. It is labor-intensive and partly subjective — two technicians can disagree on a faint pattern — but it catches antibodies the cheaper methods miss.
ELISA and Multiplex Bead Assays — Fast but Fallible
To speed throughput, many commercial labs have switched to ELISA (enzyme-linked immunosorbent assay) or multiplex bead assays (Luminex, BioPlex). These use a fixed panel of purified nuclear antigens coated on plates or beads. The advantage is volume: hundreds of samples per run, automated, cheap. The disadvantage is that they only detect antibodies against the specific antigens on the panel. A real ANA directed at an antigen the panel does not include will be reported as negative, even when IFA would light up strongly.
This matters. Published series find that up to 35% of lupus patients test negative on solid-phase ANA assays while testing positive by IFA. If your clinical picture looks like lupus and your "ANA negative" came from a multiplex panel, insist on a reflex IFA before accepting the result.
Reading Your ANA: Titer and Pattern
An IFA result reports a ratio such as 1:80, 1:160, 1:320, 1:640, 1:1280. The denominator is the dilution at which fluorescence is still visible. Higher titers mean more antibody.
- 1:40 — present in up to 25-30% of healthy adults; usually not meaningful.
- 1:80 — the screening threshold used in ACR/EULAR 2019 classification; a single-entry requirement for SLE classification.
- 1:160 — clinically suspicious.
- 1:320 and above — strongly suggestive of a connective-tissue disease; time for a full autoantibody workup.
The pattern narrows the suspect list:
- Homogeneous (diffuse) — even staining of the whole nucleus. Suggests antibodies to dsDNA, histones, or chromatin. Common in SLE and drug-induced lupus.
- Speckled — fine or coarse dots throughout the nucleus. Suggests anti-Sm, anti-RNP, anti-Ro/SSA, anti-La/SSB. Seen in SLE, Sjögren's, and mixed connective tissue disease.
- Nucleolar — staining concentrated in the nucleoli. More classic for systemic sclerosis (scleroderma) than for lupus.
- Centromere — discrete dots corresponding to chromosome centromeres. Tightly linked to limited scleroderma (CREST).
- Cytoplasmic — staining outside the nucleus. Suggests antibodies to ribosomal P (lupus), Jo-1 (myositis), or mitochondria (primary biliary cholangitis).
The pattern is a routing signal, not a diagnosis. It tells the lab and the rheumatologist which follow-up antibodies to run.
Anti-dsDNA — The Lupus-Specific Antibody
Antibodies against double-stranded DNA are one of two antibodies (with anti-Sm) considered genuinely specific for SLE. Roughly 60-70% of lupus patients test positive at some point; if you have anti-dsDNA, the probability you have lupus is very high.
Three Testing Methods, Three Meanings
- Crithidia luciliae immunofluorescence (CLIFT) — uses a protozoan whose kinetoplast is essentially pure dsDNA. Considered the most specific but less sensitive. A positive CLIFT is meaningful.
- Farr radioimmunoassay — the classic quantitative method, correlates best with kidney disease. Largely phased out because of radioactivity.
- ELISA — the everyday clinical assay. Sensitive, quantitative, but less specific — can pick up low-affinity antibodies that are not clinically meaningful. Most labs now use this.
Anti-dsDNA as a Flare Meter
In a subset of patients — often called "serologically active" — anti-dsDNA titers rise weeks to months before a clinical flare, particularly lupus nephritis. A rising dsDNA paired with a falling C3 or C4 is one of the most reliable warning signs in rheumatology and often prompts pre-emptive treatment intensification. See the lupus nephritis article for how this pairing guides biopsy and induction therapy.
Not every lupus patient tracks this way. Some have persistently high dsDNA and stable disease; others flare with normal dsDNA. Your own pattern, once your rheumatologist has watched it for a year or two, is more informative than the textbook pattern.
Anti-Smith (Sm) — Highly Specific, Often Missed
Anti-Sm targets a set of small nuclear ribonucleoproteins involved in splicing pre-mRNA. It is the second disease-specific antibody for SLE: specificity exceeds 95%, meaning almost no one without lupus has it. The catch is sensitivity — only 10-30% of lupus patients are Sm-positive, with higher prevalence in patients of African and Asian ancestry.
Because Sm rarely turns positive after initial diagnosis, it is usually checked once as part of the extractable nuclear antigen (ENA) panel at diagnosis. Its presence is strong confirmation of lupus and has been associated with more severe disease, including nephritis and central nervous system involvement, though the association is not strong enough to drive individual treatment decisions.
Anti-Ro/SSA and Anti-La/SSB
These two antibodies target small ribonucleoprotein particles (Ro60, Ro52, and La). They are not lupus-specific — in fact, they are the hallmark antibodies of Sjögren's syndrome, where 60-80% of patients are positive. But they appear in roughly 30-40% of SLE patients and carry three very practical implications.
1. Subacute Cutaneous Lupus and Photosensitivity
Anti-Ro is tightly linked to the photosensitive annular or psoriasiform rash of subacute cutaneous lupus erythematosus (SCLE). If you have anti-Ro and your rash flares with sun exposure, rigorous photoprotection is non-negotiable. See the cutaneous lupus article for specific sunscreen and clothing strategies.
2. Sjögren's Overlap
Up to 20% of lupus patients develop secondary Sjögren's — dry eyes, dry mouth, swollen parotid glands — and anti-Ro/La predict this overlap. The Lupus & Sjögren's overlap article covers the full workup.
3. Neonatal Lupus and Congenital Heart Block
This is the single most important piece of family-planning information for any woman with anti-Ro or anti-La. These antibodies cross the placenta, and in roughly 1-2% of first pregnancies they cause congenital heart block in the fetus — an irreversible conduction defect that can require a lifelong pacemaker or, rarely, prove fatal in utero. Prior affected pregnancies raise the recurrence risk to 15-20%. The risk window is 16-26 weeks, during which serial fetal echocardiograms and prophylactic hydroxychloroquine are used. The lupus and pregnancy article lays out the full monitoring protocol.
Ribosomal P and Neuropsychiatric Lupus
Anti-ribosomal P antibodies target three phosphoproteins on the large ribosomal subunit. They are present in roughly 10-20% of lupus patients, are highly specific (>95%), and show a cytoplasmic ANA pattern.
Their clinical signature is neuropsychiatric lupus (NPSLE) — psychosis, severe depression, and lupus cerebritis. The association is imperfect: some patients with ribosomal P never develop psychiatric disease, and many NPSLE patients never have the antibody. But in a lupus patient with new-onset psychosis or depression, a positive ribosomal P helps argue that the symptoms are lupus-driven rather than, say, steroid-induced or situational. See neuropsychiatric lupus for the full differential.
U1-RNP and Mixed Connective Tissue Disease
Anti-U1-RNP targets small nuclear ribonucleoproteins that partner with Sm but are a separate antibody. It appears in roughly 30-40% of lupus patients at low-to-moderate titers. At very high titers, in the absence of anti-dsDNA or anti-Sm, the diagnosis shifts from SLE to mixed connective tissue disease (MCTD) — a syndrome blending features of lupus, scleroderma, and polymyositis, often with Raynaud's, puffy hands, and pulmonary hypertension risk.
For treatment purposes the MCTD/SLE distinction matters: MCTD has a higher baseline rate of pulmonary arterial hypertension, which warrants periodic echocardiogram screening regardless of symptoms.
The Antiphospholipid Panel
Up to 30-40% of lupus patients carry antiphospholipid antibodies (aPL), and roughly a third of those develop clinical antiphospholipid syndrome (APS) — arterial or venous thrombosis, recurrent miscarriage, or both. Every lupus patient should have this panel checked at diagnosis and repeated if new thrombotic events occur.
Three tests make up the panel, and you need all three because they detect overlapping but non-identical antibody populations:
- Lupus anticoagulant (LA) — a functional clotting-time assay (despite the misleading name, LA prolongs test-tube clotting but promotes clotting in living blood). Requires screening plus confirmatory and mixing studies, and cannot be reliably measured while a patient is on warfarin or direct oral anticoagulants.
- Anti-cardiolipin (aCL) — IgG and IgM against cardiolipin, reported in GPL and MPL units. Moderate-to-high positive (>40 units) on two occasions 12 weeks apart is the diagnostic threshold.
- Anti-β2-glycoprotein I (anti-β2-GPI) — the most specific of the three. High-titer IgG positivity is the strongest risk marker for thrombosis.
The most dangerous phenotype is "triple positive" — lupus anticoagulant, anti-cardiolipin, and anti-β2-GPI all positive. Triple-positive patients carry the highest recurrent-thrombosis risk, typically require lifelong warfarin (direct oral anticoagulants are inferior in this setting), and face specific pregnancy-management protocols. Any positive aPL antibody must be confirmed on a second sample at least 12 weeks apart because transient positivity happens with infections, including the Epstein-Barr virus.
Complement: C3, C4, and CH50 as Activity Markers
The complement system is a cascade of blood proteins that immune complexes consume. In active lupus, immune complexes form faster than the liver can replace the proteins, so complement levels fall. Tracking them is one of the simplest and cheapest ways to monitor disease activity.
- C3 — the most commonly followed. Normal range roughly 90-180 mg/dL. Falls with active lupus, especially nephritis.
- C4 — normal range roughly 10-40 mg/dL. Tends to fall earlier than C3 in a flare. Complication: partial C4 null alleles are present in up to 30% of the population, producing chronically low C4 at baseline regardless of disease activity. Your rheumatologist should establish your personal baseline.
- CH50 — a functional assay of the whole classical pathway. Useful when you need to check for inherited complement deficiency, which is itself a strong risk factor for lupus.
The combination that predicts trouble is rising anti-dsDNA + falling C3/C4, particularly with new proteinuria on urinalysis. That triad should trigger a same-month call to your rheumatologist, not a wait for the next scheduled visit.
How Antibodies Predict Your Lupus Phenotype
Lupus is not one disease. It is a family of overlapping clinical patterns, and your antibody profile tells you which one you most resemble. Rough associations:
- Anti-dsDNA + low C3/C4 — kidney-predominant lupus; higher nephritis risk; flare-driven course.
- Anti-Sm — more severe disease overall; higher nephritis and CNS risk, especially in patients of African or Asian ancestry.
- Anti-Ro/SSA — photosensitive skin disease, secondary Sjögren's, neonatal lupus risk in pregnancy.
- Anti-ribosomal P — psychosis, depression, lupus cerebritis.
- High-titer anti-U1-RNP (isolated) — mixed connective tissue disease; pulmonary hypertension risk; Raynaud's.
- Antiphospholipid antibodies — thrombosis, stroke, miscarriage, livedo, thrombocytopenia.
- Anti-histone (predominant) — classic for drug-induced lupus (hydralazine, procainamide, minocycline, TNF inhibitors).
None of these profiles is deterministic. They shift probabilities, not certainties. But they shape what your rheumatologist screens for and how often — urinalysis every visit for dsDNA-positive patients, echocardiogram every 1-2 years for high-titer RNP, fetal echo during pregnancy for Ro-positive women.
False Positives and Borderline Results
A low-titer ANA by itself is not a diagnosis. Reasons a perfectly healthy person might have a positive ANA:
- Age. ANA prevalence rises with age; roughly 20-30% of adults over 65 are ANA-positive at 1:40, with no illness.
- Family history. Healthy first-degree relatives of lupus patients are more often ANA-positive than the general population.
- Recent infection. Viral infections (Epstein-Barr, parvovirus B19, hepatitis) can produce transient ANA positivity and even transient antiphospholipid antibodies.
- Other autoimmune disease. Autoimmune thyroid disease, primary biliary cholangitis, and autoimmune hepatitis commonly produce a positive ANA.
- Medications. Hydralazine, procainamide, isoniazid, minocycline, certain anticonvulsants, and TNF inhibitors can induce ANA and drug-induced lupus. The predominant antibody in drug-induced lupus is anti-histone, and the syndrome usually resolves when the drug is stopped.
- Pregnancy. A mild rise in ANA titer can occur even in healthy pregnancies.
The corollary is also true: a positive ANA in someone with no symptoms, normal labs, and no family history should be followed, not treated. Referral to rheumatology for a one-time assessment is reasonable; lifelong surveillance is not. Many people with an incidentally found ANA live normal lives and never develop autoimmune disease.
A Practical Ordering Strategy
If you are a patient advocating for a full workup, or a clinician tuning your lupus panel, this is a sensible sequence:
- ANA by IFA on HEp-2 cells with titer and pattern. Insist on IFA, not multiplex-only.
- If ANA ≥ 1:80 and clinical suspicion is present, add an ENA panel (Sm, RNP, Ro/SSA, La/SSB, Scl-70, Jo-1, centromere).
- Anti-dsDNA by ELISA plus CLIFT if available. Recheck quantitatively every 3-6 months once baseline is established.
- Complement C3, C4, and CH50 if you suspect inherited deficiency. Recheck C3/C4 with each rheumatology visit.
- Antiphospholipid panel: lupus anticoagulant, anti-cardiolipin IgG/IgM, anti-β2-GPI IgG/IgM. Repeat any positive result at 12 weeks.
- Ribosomal P if neuropsychiatric symptoms appear.
- CBC, urinalysis with microscopy, creatinine, and spot urine protein/creatinine ratio — not antibodies, but essential companions that catch organ involvement the antibodies only hint at.
Cost at major U.S. commercial labs: the full panel typically runs $300-700 cash-pay, and is covered by insurance when ordered with a relevant ICD-10 code (M32.9 for SLE unspecified, R76.0 for abnormal ANA). Ask the lab for itemized CPT codes if you need to negotiate.
Key Research Papers
- Aringer M, et al. 2019 EULAR/ACR classification criteria for systemic lupus erythematosus. Ann Rheum Dis. 2019.
- Pisetsky DS, et al. ANA as an entry criterion for the classification of SLE. Ann Rheum Dis. 2019.
- Kaul A, et al. Systemic lupus erythematosus. Nat Rev Dis Primers. 2016.
- Miyakis S, et al. International consensus statement on an update of the classification criteria for definite antiphospholipid syndrome (APS). J Thromb Haemost. 2006.
- Pengo V, et al. Rivaroxaban vs warfarin in high-risk patients with antiphospholipid syndrome. Blood. 2018.
- Izmirly PM, et al. Maternal use of hydroxychloroquine and prevention of recurrent congenital heart block. J Am Coll Cardiol. 2020.
- Agmon-Levin N, et al. International recommendations for the assessment of autoantibodies to cellular antigens referred to as anti-nuclear antibodies. Ann Rheum Dis. 2014.
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on lupus autoantibody testing, interpretation, and clinical correlation:
- ANA testing by HEp-2 immunofluorescence in lupus
- Anti-dsDNA and lupus nephritis flare prediction
- Anti-Smith antibody in systemic lupus
- Anti-Ro/SSA, neonatal lupus, and congenital heart block
- Ribosomal P antibody and neuropsychiatric lupus
- U1-RNP and mixed connective tissue disease
- Triple-positive antiphospholipid syndrome and thrombosis
- Complement C3/C4 and lupus disease activity
- Drug-induced lupus and anti-histone antibodies
- ANA-negative lupus and solid-phase assay limitations
Connections
- Lupus Overview
- Neuropsychiatric Lupus
- Lupus and Pregnancy
- Lupus-Sjogren's Overlap
- Cutaneous Lupus and Photoprotection
- Lupus Nephritis and Kidney Involvement
- ACR-EULAR 2019 Classification Criteria
- Biologics: Belimumab, Anifrolumab and Rituximab
- Hydroxychloroquine and Antimalarials
- Sjogren's Syndrome
- Raynaud's Disease
- Kidney Disease
- Arthritis
- Full Body MRI
- Urinalysis
- Thrombocytopenia