Biologics for Lupus: Belimumab, Anifrolumab, and Rituximab
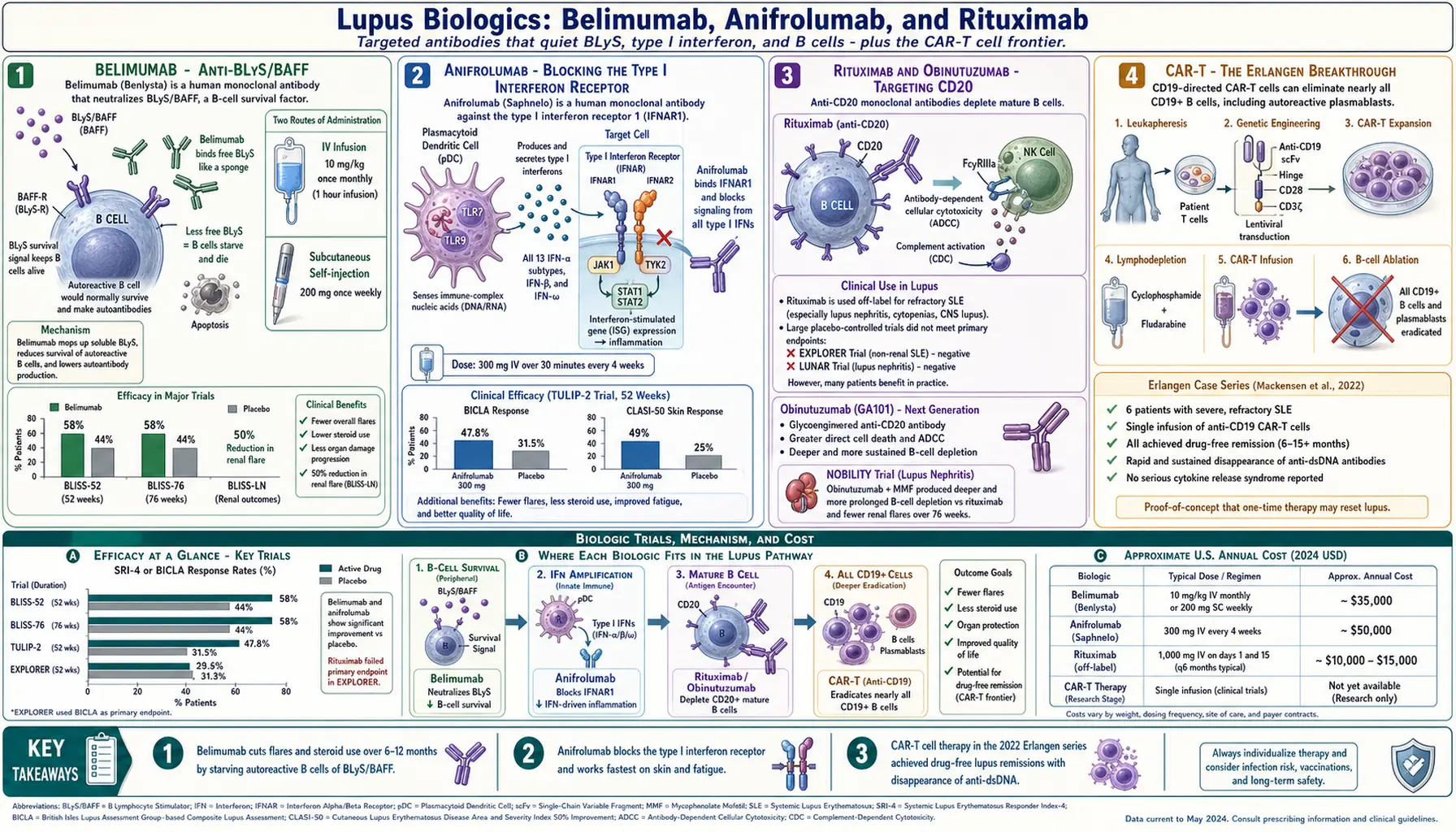
Table of Contents
- Why Biologics for Lupus at All
- Belimumab — Anti-BLyS/BAFF
- BLISS-52, BLISS-76, and BLISS-LN
- IV vs Subcutaneous Belimumab
- Anifrolumab — Blocking the Type I Interferon Receptor
- TULIP-1 and TULIP-2
- Rituximab — The Off-Label Workhorse
- Obinutuzumab and the NOBILITY Trial
- CAR-T Cell Therapy — The Erlangen Breakthrough
- Sequencing and Combinations
- Infection Risk and Vaccination Timing
- Pregnancy Considerations
- Cost, Access, and Insurance Reality
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
Why Biologics for Lupus at All
Lupus has always been the stubborn autoimmune disease. Rheumatoid arthritis has had targeted biologics since the late 1990s; psoriasis patients can choose from a dozen. Lupus, for decades, had only one treatment recipe: steroids plus an antimalarial plus an immunosuppressant, adjusted up and down in response to flares. Steroids worked but scarred every organ they touched. Mycophenolate and cyclophosphamide worked but hit bone marrow, ovaries, and the liver.
The reason lupus resisted targeted therapy so long is that it is not one disease. It is dozens of overlapping immune failures: B cells that make pathogenic autoantibodies, T cells that help them, plasmacytoid dendritic cells that flood the blood with type I interferon, complement that gets consumed by immune complexes, and tissue that gets caught in the crossfire. Every patient's mix is different. A drug that shuts down one pathway helps the patients whose disease runs through that pathway, and does little for everyone else.
The modern biologic era started in 2011 with belimumab, the first FDA-approved lupus drug in 56 years. Anifrolumab followed in 2021. Rituximab never got an approved indication but is used constantly off-label. And in 2022, a German team published six cases of CAR-T cell therapy that achieved something no other treatment ever had: drug-free remission with disappearance of anti-dsDNA. The biologic landscape is changing faster now than in the previous fifty years combined.
Belimumab — Anti-BLyS/BAFF
Belimumab (brand name Benlysta) is a monoclonal antibody that binds and neutralizes BLyS, also called BAFF (B-cell activating factor). BLyS is a survival signal that B cells need in order to mature and stay alive. In lupus, BLyS is elevated in the blood, and the excess keeps autoreactive B cells — ones that would normally die off — alive and making autoantibodies.
By sponging up BLyS with belimumab, you slowly starve the autoreactive B-cell population. The effect is gentle and cumulative, not dramatic. Patients rarely feel a sudden difference. What they get, over six to twelve months, is fewer flares, lower steroid doses, and in many cases a noticeable drop in anti-dsDNA titers and a rise in complement C3 and C4.
Belimumab is FDA-approved for active SLE in adults and children age 5 and older, and since 2020 for active lupus nephritis. It is given either as a monthly IV infusion (10 mg/kg) or as a weekly subcutaneous injection (200 mg). Both work. The choice comes down to access, insurance, and whether you want to sit in an infusion chair once a month or learn to self-inject weekly.
BLISS-52, BLISS-76, and BLISS-LN
Belimumab's approval rests on three large randomized trials.
BLISS-52 (published in The Lancet, 2011) enrolled 865 patients, mostly from Asia, Latin America, and Eastern Europe. At 52 weeks, the SLE Responder Index (SRI-4) response rate was 58% on belimumab 10 mg/kg vs 44% on placebo — a 14-point absolute difference, statistically significant.
BLISS-76 (published in Arthritis & Rheumatism, 2011) enrolled 819 patients, mostly from North America and Europe. At 52 weeks, 43% on belimumab vs 34% on placebo responded — a smaller effect, but still significant. The difference between the two trials largely reflected background steroid doses and ethnic composition.
BLISS-LN (published in The New England Journal of Medicine, 2020) was the kidney trial. 448 patients with biopsy-proven class III, IV, or V lupus nephritis were randomized to belimumab plus standard therapy (mycophenolate or cyclophosphamide-azathioprine) or standard therapy alone. At two years, the primary efficacy renal response was achieved in 43% on belimumab vs 32% on placebo. More importantly, belimumab patients had half the risk of a renal flare or worsening over follow-up.
The take-home for patients: belimumab is a flare-reducer and a steroid-sparer. It does not send lupus into remission in most people. It tilts the odds over time, and for patients with residual activity despite hydroxychloroquine and a standard immunosuppressant, that tilt is usually worth having.
IV vs Subcutaneous Belimumab
The IV formulation (10 mg/kg monthly) was the original. Subcutaneous belimumab (200 mg weekly, self-injected) was approved in 2017 after the BLISS-SC trial showed non-inferiority.
- IV: monthly infusion, usually at an infusion center, about an hour in the chair. Dose is weight-based, so very large patients get proportionally more drug. Most insurance buckets it as a medical benefit (Part B for Medicare), which can mean lower out-of-pocket for some plans, higher for others.
- SC: weekly prefilled pen or syringe at home. Fixed 200 mg dose regardless of weight. Usually a pharmacy benefit (Part D for Medicare). More convenient for anyone with a job or kids; less convenient for anyone with needle phobia or dexterity issues.
Efficacy appears similar. Injection-site reactions are the most common SC-specific issue — redness, bruising, itch — usually mild and fading after the first few months. Most rheumatologists will let the patient choose, and many will switch formulations if the first one is not working logistically.
Anifrolumab — Blocking the Type I Interferon Receptor
Anifrolumab (brand name Saphnelo) is a monoclonal antibody against subunit 1 of the type I interferon receptor (IFNAR1). When it binds, it blocks signaling from all type I interferons — the 13 IFN-alphas, IFN-beta, IFN-omega, and others — at a single bottleneck.
Why does that matter? Because in lupus, a majority of patients have a high interferon signature: their blood cells show upregulation of hundreds of IFN-induced genes. Plasmacytoid dendritic cells sensing immune-complex debris pump out type I IFN, which in turn activates more B cells, drives more autoantibody production, and inflames skin and joints. Anifrolumab cuts the amplifier.
It is FDA-approved (since 2021) for moderate-to-severe SLE in adults on standard therapy. Dose is 300 mg IV every four weeks. There is no subcutaneous version yet approved in the U.S. (a SC formulation is in late-stage trials).
Patients often describe anifrolumab as working on skin and fatigue first — the two symptoms most tightly linked to IFN-alpha. Joint pain improvement follows. It is not a kidney drug; lupus nephritis trials are ongoing but not yet reported.
TULIP-1 and TULIP-2
TULIP-1 (published in The Lancet Rheumatology, 2019) enrolled 457 patients and initially failed its primary endpoint of SRI-4 response at 52 weeks. That looked like the end of anifrolumab. But when the sponsor re-analyzed using a different, more lupus-sensitive endpoint (the BICLA — BILAG-based Composite Lupus Assessment), the drug came back to life.
TULIP-2 (published in The New England Journal of Medicine, 2020) used BICLA as the primary endpoint from the start. 362 patients. At 52 weeks, 47.8% on anifrolumab vs 31.5% on placebo achieved BICLA response — a clean 16-point difference, highly significant. Cutaneous disease (as measured by the CLASI-50 response) improved in 49% vs 25%. Steroid tapering to 7.5 mg prednisone or less was achieved in 52% vs 30%.
That split between the two trials is instructive. It is not that the drug didn't work in TULIP-1 — it is that the SRI-4 was a poorly calibrated yardstick for this class of therapy. BICLA captures improvement in specific organ systems that SRI-4 misses. Modern lupus trials have largely moved to BICLA for this reason.
Rituximab — The Off-Label Workhorse
Rituximab is a chimeric anti-CD20 monoclonal antibody that destroys circulating B cells. It is FDA-approved for rheumatoid arthritis, several lymphomas, and ANCA-associated vasculitis — but not for lupus. Two pivotal trials, EXPLORER (extrarenal SLE, published in Arthritis & Rheumatism, 2010) and LUNAR (lupus nephritis, published in Arthritis & Rheumatism, 2012), both failed their primary endpoints.
And yet rituximab is one of the most-used biologics in real-world lupus care. Why? Because the trials probably had the wrong design, not the wrong drug. EXPLORER and LUNAR allowed high background steroid doses that washed out any rituximab signal. Observational cohorts, registries like BILAG-BR and LRRPS, and single-center series have consistently shown that rituximab helps refractory patients — the ones who failed mycophenolate, cyclophosphamide, and often belimumab, and who are heading toward irreversible organ damage.
Rituximab has a particularly strong real-world track record in:
- Refractory lupus nephritis after mycophenolate failure.
- Severe autoimmune cytopenias in lupus — immune thrombocytopenia, autoimmune hemolytic anemia.
- Catastrophic antiphospholipid syndrome (CAPS) and some difficult secondary APS cases.
- Severe neuropsychiatric lupus unresponsive to cyclophosphamide — see the neuropsychiatric lupus article.
Typical dosing is 1000 mg IV on day 1 and day 15, repeated every 6 months or at B-cell repopulation. Insurance coverage off-label is possible but usually requires peer-to-peer advocacy by the rheumatologist citing published cohort data and prior treatment failures.
Obinutuzumab and the NOBILITY Trial
Obinutuzumab is a newer anti-CD20 antibody, glycoengineered for stronger B-cell depletion than rituximab. The NOBILITY phase 2 trial (published in Annals of the Rheumatic Diseases, 2022) tested it in 125 patients with active proliferative lupus nephritis on top of mycophenolate. At 76 weeks, complete renal response was achieved in 41% on obinutuzumab vs 23% on placebo.
A larger phase 3 trial (REGENCY) reported in 2024 confirmed the benefit in lupus nephritis, and obinutuzumab is expected to reach FDA review for this indication. For patients with refractory proliferative nephritis, this will likely become a major option within the next few years, possibly displacing rituximab for kidney disease specifically.
CAR-T Cell Therapy — The Erlangen Breakthrough
In 2022, a team at University Hospital Erlangen in Germany, led by Georg Schett, reported five patients with severe refractory lupus treated with CD19-directed chimeric antigen receptor (CAR) T cells — the same technology used in blood cancers. The concept: take the patient's own T cells, engineer them in a lab to recognize and kill any cell expressing CD19 (all B cells), infuse them back, and let them wipe B cells far more deeply than any antibody can.
The results, published in Nature Medicine (2022) and updated in The New England Journal of Medicine (2024), were extraordinary. All treated patients achieved drug-free remission. Anti-dsDNA became undetectable. Complement normalized. When B cells eventually repopulated from the bone marrow (typically around month 4), they came back as naive, non-autoreactive — the autoimmune "memory" had been erased. Follow-up is now past two years with continued drug-free remission in most patients.
This is the first therapy that has plausibly reset lupus rather than suppressed it. CAR-T for lupus is now in multi-center trials worldwide. Access in 2026 is still limited to a handful of academic centers and clinical trials, with cost estimates in the $400,000–$500,000 range per treatment — more than any biologic, but potentially a one-time price. Serious risks include cytokine release syndrome, infection during the B-cell aplasia window, and the long-term unknowns of engineered T cells. Candidates so far have been patients with severe disease unresponsive to every other option. Within five years, that bar is likely to come down.
Sequencing and Combinations
There is no universally agreed-upon order, but the practical rheumatology reality in 2026 looks roughly like this:
- Hydroxychloroquine for essentially everyone with lupus, at all severity levels. See the hydroxychloroquine article.
- Steroids for flares, tapered as fast as the disease allows.
- Mycophenolate, azathioprine, or methotrexate as a conventional immunosuppressant backbone.
- Belimumab added for persistent moderate activity or frequent flares — often alongside mycophenolate in nephritis.
- Anifrolumab for skin-dominant or fatigue-dominant disease, or as an alternative to belimumab if belimumab has failed. A handful of centers now use belimumab and anifrolumab together in severe cases, but this is not yet standard and not always covered.
- Rituximab (or increasingly obinutuzumab) for refractory nephritis, cytopenias, NPSLE, or APS complications.
- CAR-T trial referral for patients who have exhausted the above and have life-threatening or rapidly damaging disease.
An important 2023 French cohort (the BEAT-LUPUS approach, published in The Lancet Rheumatology) showed that rituximab followed by belimumab produced better long-term disease control than rituximab alone — the idea being that rituximab wipes the B cells, belimumab then prevents the newly emerging B cells from finding the BLyS signals they need to re-establish autoimmunity. This sequence is now a reasonable option for refractory patients, at least at specialist centers.
Infection Risk and Vaccination Timing
All B-cell-targeting and interferon-blocking biologics raise infection risk. The pattern differs by mechanism.
- Belimumab: modest increase in serious infections vs placebo in trials. Sinopulmonary infections, urinary tract infections, and herpes zoster top the list.
- Anifrolumab: meaningfully higher rates of herpes zoster (shingles) — about three times background. Upper respiratory infections also more common. All patients starting anifrolumab should receive the recombinant zoster vaccine (Shingrix) beforehand if possible.
- Rituximab and obinutuzumab: deep B-cell depletion means impaired vaccine responses for 6–12 months post-infusion, persistent hypogammaglobulinemia in a minority, and an increased risk of hepatitis B reactivation (screen with HBsAg and anti-HBc before starting). Rare but catastrophic: progressive multifocal leukoencephalopathy (PML) in longstanding lupus patients on long-term B-cell depletion.
Vaccination timing rules of thumb:
- Give all inactivated vaccines (influenza, pneumococcal PCV20, Tdap, HPV, Shingrix, hepatitis B) before starting a biologic when possible, or at least 2 weeks before the next dose once on therapy.
- Live vaccines (MMR, varicella, yellow fever, live zoster, nasal flu) are contraindicated on B-cell-depleting therapy and generally avoided on belimumab and anifrolumab as well. Shingrix is a recombinant (non-live) vaccine and is safe on all these biologics.
- For rituximab, ideally vaccinate at least 4 weeks before infusion. Vaccine response after rituximab is blunted for 6–12 months.
Pregnancy Considerations
Lupus hits women of childbearing age hardest, so pregnancy planning is central to biologic choice. See the lupus and pregnancy article for the full discussion; the biologic-specific summary:
- Hydroxychloroquine: continue throughout pregnancy. Strongly recommended — reduces flares and neonatal lupus risk.
- Belimumab: there is a pregnancy registry (Belimumab Pregnancy Registry) with several hundred reported outcomes showing no clear teratogenic signal, but data are still limited. Guidelines currently suggest stopping before conception if disease allows, continuing if disease control requires it and the patient is informed. Crosses the placenta in the third trimester.
- Anifrolumab: minimal human pregnancy data. Generally avoided. Wash out before conception when possible.
- Rituximab: causes transient B-cell depletion in the infant if given in the second or third trimester. Live vaccines must be held for the baby until B cells recover. First-trimester exposure appears low risk.
Breastfeeding: IgG biologics cross into breast milk in tiny amounts and are generally not absorbed intact from the infant gut. Belimumab, anifrolumab, and rituximab are all considered compatible with breastfeeding in current rheumatology guidelines, though decisions are individualized.
Cost, Access, and Insurance Reality
Biologics are expensive. Ballpark U.S. list prices in 2026:
- Belimumab IV: roughly $3,500–$4,500 per monthly infusion (weight-dependent), about $45,000–$55,000/year.
- Belimumab SC: roughly $1,000 per weekly 200 mg pen, about $52,000/year.
- Anifrolumab: roughly $5,000 per monthly infusion, about $60,000/year.
- Rituximab (off-label): roughly $8,000–$10,000 per 1000 mg dose; a standard induction (day 1 + day 15) runs $16,000–$20,000. Biosimilars (rituximab-pvvr, rituximab-abbs, rituximab-arrx) can cut this by 15–40%.
- Obinutuzumab: when used off-label for lupus, similar to rituximab pricing.
- CAR-T (investigational): $400,000–$500,000 one-time, generally only accessible through trial enrollment.
Access tactics that actually work:
- Manufacturer patient-assistance programs. GSK (belimumab) and AstraZeneca (anifrolumab) both run copay-assistance and free-drug programs for uninsured or underinsured patients. These are under-used; ask your rheumatologist's office or search "[drug name] patient access."
- Peer-to-peer review for denials. When an insurer denies belimumab or anifrolumab citing "not enough prior therapy tried," a 15-minute phone call between the rheumatologist and the insurance medical director reverses most denials.
- Specialty pharmacy steering. For SC belimumab, the cheaper path is usually the insurer's preferred specialty pharmacy, not the practice's in-house one. Ask.
- Rituximab biosimilars. If your insurer denies branded rituximab, pivot to a biosimilar — the off-label lupus coverage argument is identical, and the lower price often unsticks approval.
- Medicare Part B vs Part D. IV biologics given in a clinic are billed under Part B and have different cost-sharing than SC biologics filled at a pharmacy under Part D. For Medicare patients, the choice of formulation can change the annual out-of-pocket by thousands of dollars. Run the numbers both ways before committing.
Insurance will rarely pay for two biologics at once. Combinations like belimumab + anifrolumab or the rituximab-then-belimumab sequence are typically only approved at academic centers willing to fight sustained appeals — or in clinical trial contexts where the drug is free.
Key Research Papers
- Navarra SV, et al. Efficacy and safety of belimumab in patients with active systemic lupus erythematosus: a randomised, placebo-controlled, phase 3 trial (BLISS-52). Lancet. 2011.
- Furie R, et al. A phase III, randomized, placebo-controlled study of belimumab, a monoclonal antibody that inhibits B lymphocyte stimulator, in patients with systemic lupus erythematosus (BLISS-76). Arthritis Rheum. 2011.
- Furie R, et al. Two-year, randomized, controlled trial of belimumab in lupus nephritis (BLISS-LN). N Engl J Med. 2020.
- Morand EF, et al. Trial of anifrolumab in active systemic lupus erythematosus (TULIP-2). N Engl J Med. 2020.
- Furie RA, et al. Type I interferon inhibitor anifrolumab in active systemic lupus erythematosus (TULIP-1). Lancet Rheumatol. 2019.
- Merrill JT, et al. Efficacy and safety of rituximab in moderately-to-severely active systemic lupus erythematosus: the EXPLORER trial. Arthritis Rheum. 2010.
- Rovin BH, et al. Efficacy and safety of rituximab in patients with active proliferative lupus nephritis: the LUNAR study. Arthritis Rheum. 2012.
- Furie RA, et al. B-cell depletion with obinutuzumab for the treatment of proliferative lupus nephritis: NOBILITY phase 2 trial. Ann Rheum Dis. 2022.
- Mackensen A, et al. Anti-CD19 CAR T cell therapy for refractory systemic lupus erythematosus. Nat Med. 2022.
- Müller F, et al. CD19 CAR T-cell therapy in autoimmune disease — a case series with follow-up. N Engl J Med. 2024.
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on lupus biologics and emerging therapies:
- Belimumab and the BLISS trial program
- Belimumab in lupus nephritis
- Anifrolumab and the TULIP trials
- Type I interferon signature in SLE
- Rituximab in refractory lupus
- Rituximab in antiphospholipid syndrome
- Obinutuzumab and the NOBILITY trial
- CAR-T cell therapy in lupus and autoimmunity
- Infection risk with biologics in SLE
- Belimumab pregnancy registry outcomes
Connections
- Lupus Overview
- Neuropsychiatric Lupus
- Lupus and Pregnancy
- Hydroxychloroquine and Antimalarials
- ACR-EULAR 2019 Classification Criteria
- Cutaneous Lupus and Photoprotection
- Lupus-Sjogren's Overlap
- ANA, dsDNA and Lupus Autoantibodies
- Lupus Nephritis and Kidney Involvement
- Arthritis
- Sjogren's Syndrome
- Kidney Disease
- Myasthenia Gravis
- Thrombocytopenia