ACR/EULAR 2019 Classification Criteria for SLE
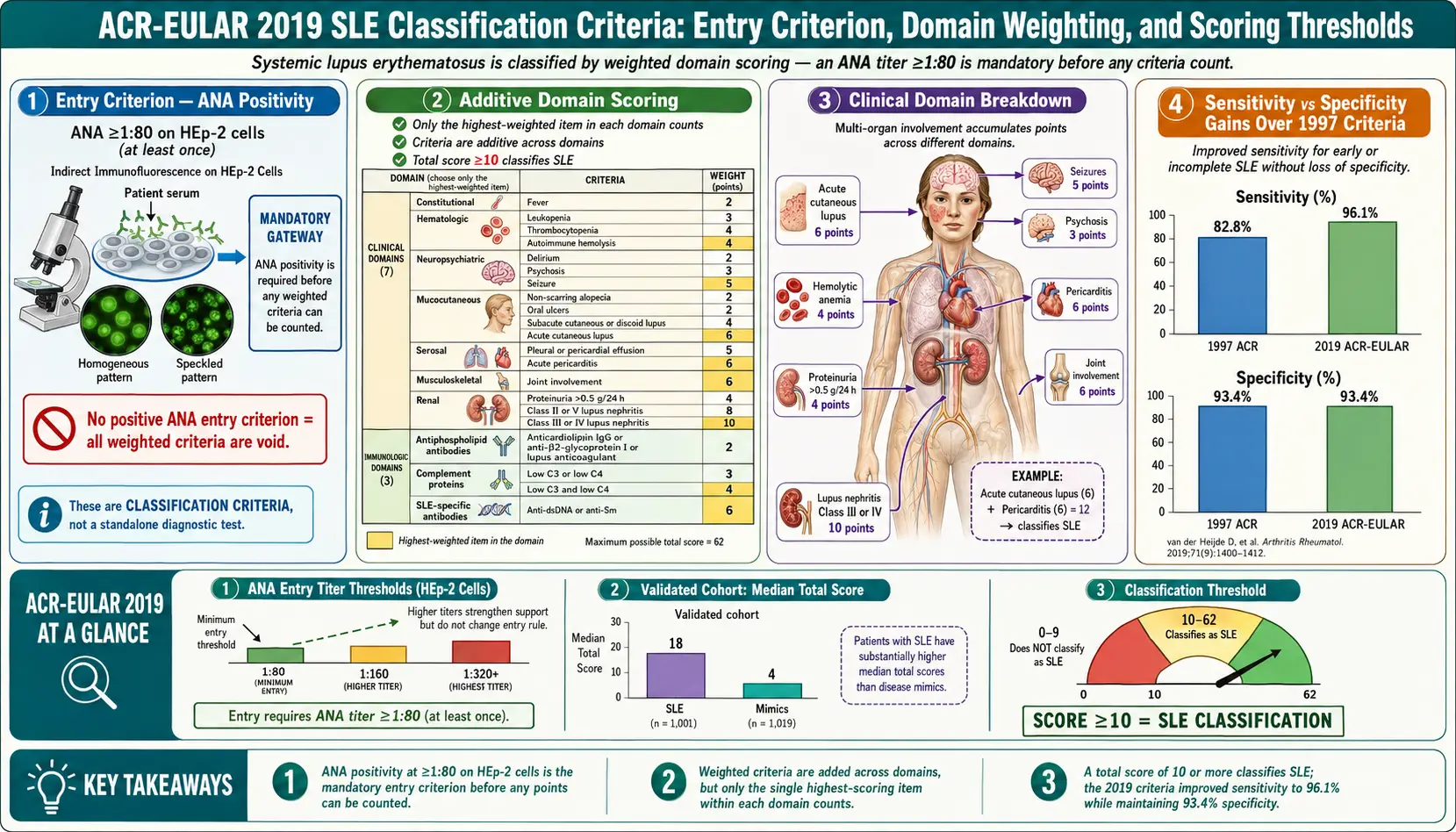
Table of Contents
- Why Classification Criteria Exist
- The Evolution: 1982 → SLICC 2012 → ACR/EULAR 2019
- The Entry Criterion: ANA ≥1:80
- The Ten Weighted Domains
- Scoring Rules — ≥10 Points and One Clinical Criterion
- How Well It Performs: 96% Sensitivity, 93% Specificity
- Classification Is Not Diagnosis
- Limitations: Young Patients, Early Disease, Rare Presentations
- Worked Examples
- What to Do With Your Score
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
Why Classification Criteria Exist
If you have been told you "meet lupus criteria" — or worse, that you "don't meet criteria" and therefore don't have lupus — it helps to understand what the criteria actually are, why they exist, and what they cannot do.
Classification criteria were invented so that researchers running clinical trials could be sure that everyone enrolled in a "lupus" study actually had the same disease. Without a standard, one hospital's trial of 100 "lupus patients" might overlap only loosely with another hospital's 100 "lupus patients," and the drug results would be impossible to compare. Classification criteria solve that problem — they create a shared rulebook for research.
That is all they were designed to do. They were never meant to be a diagnostic checklist a physician uses in clinic to decide whether to treat you. The person making your diagnosis is a rheumatologist applying clinical judgment to your full picture: symptoms, physical exam, labs, imaging, biopsies, family history, and response to therapy. Criteria are a reference point, not a verdict. This distinction matters enormously, and we come back to it in section 7.
The Evolution: 1982 → SLICC 2012 → ACR/EULAR 2019
Three generations of criteria have shaped how we classify systemic lupus erythematosus (SLE):
The 1982 ACR Criteria (revised 1997)
The original American College of Rheumatology (ACR) criteria listed eleven items — malar rash, discoid rash, photosensitivity, oral ulcers, arthritis, serositis, renal disorder, neurologic disorder, hematologic disorder, immunologic disorder, and a positive antinuclear antibody (ANA). A patient needed at least 4 of 11, either simultaneously or serially, to be classified as having SLE. The criteria were simple and durable, used in thousands of studies, but they had known gaps. Lupus nephritis confirmed on biopsy — a slam-dunk piece of evidence — only counted as one item. Patients with severe single-organ disease often failed to reach four and were excluded from trials.
The SLICC 2012 Criteria
The Systemic Lupus International Collaborating Clinics (SLICC) group expanded the list to 17 items split into clinical and immunologic categories. A patient met criteria with at least 4 items including at least one from each category, or with biopsy-proven lupus nephritis plus a positive ANA or anti-dsDNA. Sensitivity improved (about 97%) but specificity dropped (about 84%) — SLICC 2012 pulled in more true cases but also more false positives.
The ACR/EULAR 2019 Criteria
In 2019 the American College of Rheumatology and the European Alliance of Associations for Rheumatology (EULAR) jointly published a new system built from the ground up. Three innovations set it apart:
- A mandatory entry criterion — ANA positive at ≥1:80 — before any other item is counted.
- Weighted scoring. Items are not all worth one point. Lupus nephritis class III/IV on biopsy is worth 10. Leukopenia is worth 3. The weights reflect how strongly each finding points toward SLE.
- An “attribution rule.” An item only counts if there is no better explanation than lupus. Fever from a clear urinary tract infection does not count. Arthritis from a fresh ankle sprain does not count.
The result is a more flexible, more discriminating rulebook. A single heavily-weighted finding plus the right background can cross the threshold; a scatter of weak findings cannot. The full reference is Aringer et al., Arthritis & Rheumatology 2019 (DOI below).
The Entry Criterion: ANA ≥1:80
The 2019 system starts with a gatekeeper: you must have a positive antinuclear antibody (ANA) at a titer of at least 1:80, measured by the reference method — indirect immunofluorescence on HEp-2 cells — at some point in your history. If you have never had an ANA at 1:80 or higher, the criteria stop there. Nothing else you could score matters; you are not classified as SLE by this system.
The logic is straightforward. Roughly 97–98% of people with lupus have a positive ANA at 1:80 or higher at some point. Requiring it up front dramatically reduces false positives — patients with unrelated diseases whose rash or joint pain might otherwise accidentally score points.
A few nuances patients ask about:
- Once positive, always counts. If your ANA was 1:160 five years ago and is 1:40 today, the entry criterion is still met. ANA titers fluctuate; the system treats any historical positive at 1:80+ as qualifying.
- Method matters. Many commercial labs use ELISA or multiplex bead assays rather than HEp-2 immunofluorescence. Results do not always match. If an ELISA-based ANA is negative but clinical suspicion is strong, ask for a HEp-2 immunofluorescence ANA specifically. This is the method the criteria reference.
- A low-titer ANA is not lupus. About 15–20% of healthy adults have a positive ANA at 1:40, and the number climbs with age. An ANA alone — without symptoms, without other labs — is not a diagnosis of anything. See the ANA and autoantibody article for the full interpretation guide.
The Ten Weighted Domains
Past the entry criterion, findings are grouped into ten domains — seven clinical and three immunologic. Within each domain, only the single highest-scoring item counts. You do not add up multiple items from the same domain. This prevents, say, three different skin findings from quadruple-counting.
Clinical Domains
- Constitutional — unexplained fever over 38.3°C (2 points).
- Hematologic — leukopenia, white count below 4,000 (3 points); thrombocytopenia, platelet count below 100,000 (4 points); autoimmune hemolysis (4 points).
- Neuropsychiatric — delirium (2), psychosis (3), seizure (5). Each demands careful exclusion of other causes. See the neuropsychiatric lupus article.
- Mucocutaneous — non-scarring alopecia (2), oral ulcers (2), subacute cutaneous or discoid lupus (4), acute cutaneous lupus (6, the classic malar "butterfly" rash). See the cutaneous lupus page.
- Serosal — pleural or pericardial effusion (5), acute pericarditis (6).
- Musculoskeletal — joint involvement (6): at least two joints with synovitis or tender joints with more than 30 minutes of morning stiffness.
- Renal — proteinuria over 0.5 g / 24 hours (4), kidney biopsy showing class II or V lupus nephritis (8), kidney biopsy showing class III or IV lupus nephritis (10). Class III/IV is a one-shot ticket across the threshold. See the lupus nephritis article.
Immunologic Domains
- Antiphospholipid antibodies — anti-cardiolipin, anti-beta-2-glycoprotein I, or lupus anticoagulant positive at medium-high titer (2 points).
- Complement proteins — low C3 or low C4 (3 points); low C3 and low C4 (4 points).
- SLE-specific antibodies — anti-dsDNA or anti-Smith, either one present (6 points).
A subtle point about the "only the highest counts within a domain" rule: a patient with both thrombocytopenia and autoimmune hemolytic anemia scores 4 (not 4+4=8) in the hematologic domain. A patient with both low C3 and low C4 scores 4 in the complement domain. Across different domains the scores add freely.
Scoring Rules — ≥10 Points and One Clinical Criterion
Three rules must all be met:
- Entry criterion — ANA ≥1:80 at least once.
- Attribution — each item is only counted if lupus is the most likely explanation. The attending rheumatologist makes the call.
- Total ≥10 points with at least one clinical criterion. You cannot meet the threshold on immunologic findings alone, no matter how striking the labs look.
The "at least one clinical" rule matters. A hypothetical patient with a positive ANA, high-titer anti-dsDNA (6), low C3 and C4 (4), and antiphospholipid antibodies (2) scores 12 on labs alone — but without a single clinical feature they do not classify as SLE. In real practice this almost never happens; labs this striking usually come with at least some clinical finding. The rule is a backstop against pure-lab classifications.
Time window: items need not appear simultaneously. Anything documented at any point in the patient's history counts, as long as the attribution rule still holds.
How Well It Performs: 96% Sensitivity, 93% Specificity
In the derivation and validation cohorts published with the 2019 criteria, performance measured:
- Sensitivity: 96.1% — of patients with a rheumatologist's diagnosis of SLE, 96% also met the 2019 criteria.
- Specificity: 93.4% — of patients with a non-SLE connective tissue disease (Sjögren's, rheumatoid arthritis, scleroderma, and so on), 93% did not meet criteria.
Compared to the earlier systems, the 2019 criteria improved specificity substantially over SLICC 2012 (84%) while slightly improving sensitivity over the 1997 ACR criteria (82%). It is the most accurate classification tool SLE has ever had.
Sensitivity varies by disease duration. In patients with more than one year of disease, sensitivity approaches 98%. In patients in the first six months of symptoms — when organ damage may not yet have accumulated and antibody profiles may still be evolving — sensitivity drops. This is an important limitation for young adults in the early window, discussed further below.
Classification Is Not Diagnosis
This is the single most common source of confusion, and it is worth being blunt about it.
Classification criteria are for research cohorts. Diagnosis is what a rheumatologist does in clinic. A physician can, and routinely does, diagnose lupus in patients who do not meet classification criteria — because the clinical picture is obvious, because a biopsy shows classic pathology, because the patient responds to hydroxychloroquine the way lupus patients respond, because other diseases have been ruled out. Physicians can also withhold a lupus diagnosis from a patient who technically meets criteria if the individual items are better explained by something else, even after attribution review.
If a clinician tells you "you don't meet criteria, so you don't have lupus," that is a misuse of the tool. The correct question is "what do you think this is, and how are we going to work it up?" — not "did you score ten points on a rubric?"
The inverse is also true. "Meeting criteria" is not the same as "definitely has lupus." It means "looks enough like lupus that a research study would enroll this person." Most people who meet criteria do indeed have SLE, but about 7% of people who meet the 2019 criteria turn out, on longer follow-up or autopsy, to have had a different connective tissue disease. Classification is an approximation, not a pronouncement.
Limitations: Young Patients, Early Disease, Rare Presentations
Several real-world situations expose the limits of any classification tool:
- Early disease. In the first six to twelve months, many patients have only a handful of features — photosensitive rash plus arthralgia plus a positive ANA, for example — that do not yet total ten points. They may still have lupus. Early-SLE cohorts consistently show that criteria accumulate over time; the clock does not start at ten and count down.
- Pediatric and adolescent lupus. Children with SLE often present with kidney or hematologic disease that scores heavily and tends to meet criteria earlier. But rare juvenile variants (for example, monogenic interferonopathies that mimic lupus) may score high without being classical SLE.
- Drug-induced lupus. Hydralazine, procainamide, minocycline, TNF inhibitors, and a few others cause a lupus-like syndrome that can score points. The attribution rule is supposed to screen these out, but the clinical call can be difficult when a patient has been on the drug for years.
- Isolated cutaneous lupus. Discoid lupus confined to the skin, without systemic involvement, often does not cross the threshold. That is appropriate — skin-limited disease is biologically distinct and treated differently. See the cutaneous lupus article.
- Overlap syndromes. Patients with mixed connective tissue disease, undifferentiated connective tissue disease, or lupus-Sjögren's overlap may score points from features belonging to either condition. The attribution rule helps, but the lines are fuzzy. See the lupus-Sjögren's overlap article.
- ANA-negative lupus. Roughly 2–3% of SLE patients have a persistently negative ANA by immunofluorescence, often with anti-Ro/SSA antibodies instead. These patients cannot meet the 2019 criteria by definition. They still have lupus. A rheumatologist can diagnose and treat them; they simply would not be enrolled in a strictly 2019-criteria trial.
Worked Examples
Three composite cases illustrate how the arithmetic plays out.
Example 1: Meets Criteria Easily
A 28-year-old woman with:
- ANA 1:640 (entry met)
- Malar rash — acute cutaneous lupus (6)
- Arthritis of wrists and MCP joints (6)
- Anti-dsDNA positive (6)
- Low C3 and C4 (4)
Total: 22. Meets criteria comfortably, with multiple clinical items.
Example 2: Borderline, Pending Biopsy
A 34-year-old man with:
- ANA 1:320 (entry met)
- Oral ulcers (2)
- Non-scarring alopecia (2)
- Proteinuria 1.2 g / 24 hours (4)
- Low C3 only (3)
Total: 11, with clinical criteria present. Meets 2019 criteria. A kidney biopsy would add weight (class III nephritis would push the total to 17 and dictate treatment).
Example 3: Does Not Meet — But May Still Have Lupus
A 19-year-old woman, three months into symptoms:
- ANA 1:160 (entry met)
- Photosensitive rash that is not clearly acute cutaneous lupus (0, unless dermatology attributes it)
- Arthralgia without objective synovitis (0 — tenderness alone does not count without morning stiffness ≥30 min)
- Mild leukopenia at 3,800 (3)
Total: 3. Does not meet criteria. She does not "not have lupus." She has early, evolving disease, and the rheumatologist will follow her closely, check dsDNA and complement over time, and reassess. Half of patients in this pattern go on to meet criteria within two years.
What to Do With Your Score
If a clinician has quoted your ACR/EULAR 2019 score, here is how to use that information:
- Ask for the breakdown. Which domains contributed which points. You are entitled to see the arithmetic.
- Ask about attribution. Any item the rheumatologist attributed away — and why.
- Ask about missing information. Has anti-dsDNA been checked in the last year? Has complement been checked? A kidney biopsy if there is any proteinuria? These change scores and treatment plans.
- Do not treat the score as your diagnosis. Ask: "What is your clinical diagnosis, independent of the score? What is your confidence? What is the differential?"
- If you don't meet criteria but have symptoms, ask about undifferentiated connective tissue disease, follow-up intervals, and whether hydroxychloroquine is reasonable to start now — in some early-SLE cohorts, early hydroxychloroquine slows progression to full SLE. See the hydroxychloroquine article.
For the full picture of how classification interacts with treatment decisions, medication selection, and long-term monitoring, start at the lupus overview. If kidneys are involved, the lupus nephritis article covers biopsy classes and induction therapy in depth; see also kidney disease for broader context. Pregnancy changes monitoring considerably — see lupus and pregnancy.
Key Research Papers
- Aringer M, Costenbader K, Daikh D, et al. 2019 European League Against Rheumatism/American College of Rheumatology classification criteria for systemic lupus erythematosus. Arthritis Rheumatol. 2019;71(9):1400–1412.
- Aringer M, Costenbader K, Daikh D, et al. 2019 EULAR/ACR classification criteria for systemic lupus erythematosus. Ann Rheum Dis. 2019;78(9):1151–1159.
- Petri M, Orbai AM, Alarcón GS, et al. Derivation and validation of the Systemic Lupus International Collaborating Clinics classification criteria for systemic lupus erythematosus. Arthritis Rheum. 2012;64(8):2677–2686.
- Hochberg MC. Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum. 1997;40(9):1725.
- Tan EM, Cohen AS, Fries JF, et al. The 1982 revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum. 1982;25(11):1271–1277.
- Fanouriakis A, Kostopoulou M, Andersen J, et al. EULAR recommendations for the management of systemic lupus erythematosus: 2023 update. Ann Rheum Dis. 2024.
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on SLE classification, validation cohorts, and real-world application of the 2019 criteria:
- ACR/EULAR 2019 classification criteria for SLE
- SLICC 2012 classification criteria validation
- ANA as entry criterion in lupus classification
- Early undifferentiated connective tissue disease progressing to SLE
- ANA-negative lupus and anti-Ro antibodies
- Pediatric SLE and classification criteria performance
- Drug-induced lupus and classification criteria
- Lupus nephritis biopsy classes (ISN/RPS)
Connections
- Lupus Overview
- ANA, dsDNA and Lupus Autoantibodies
- Neuropsychiatric Lupus
- Lupus and Pregnancy
- Cutaneous Lupus and Photoprotection
- Lupus-Sjogren's Overlap
- Hydroxychloroquine and Antimalarials
- Lupus Nephritis and Kidney Involvement
- Biologics: Belimumab, Anifrolumab and Rituximab
- Kidney Disease
- Arthritis
- Sjogren's Syndrome
- Thrombocytopenia
- Alopecia
- Full Body MRI