Hydroxychloroquine and Antimalarials for Lupus
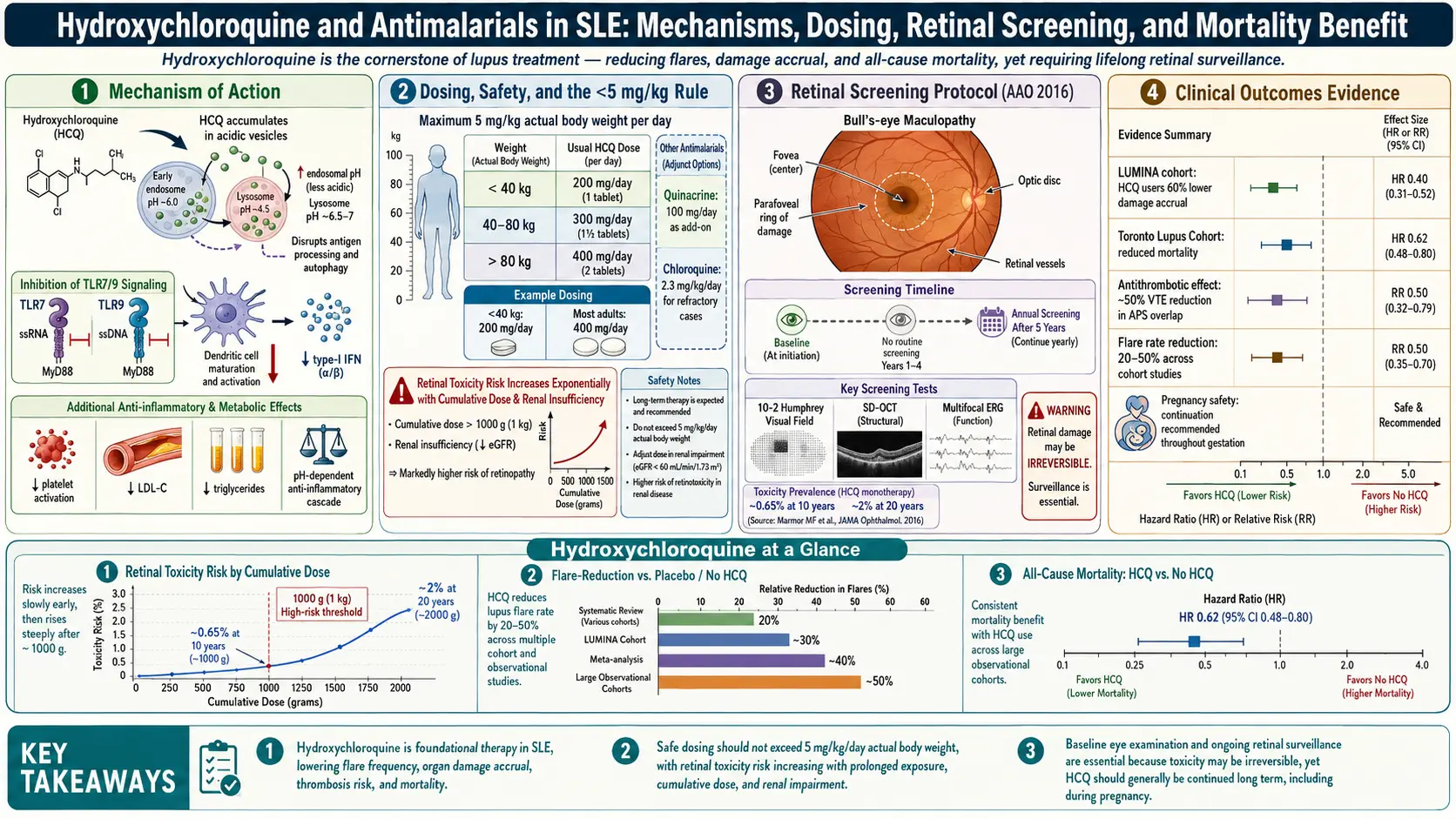
Table of Contents
- What Hydroxychloroquine Is and Why It Matters
- How It Actually Works: TLR7/9 and Lysosomal Alkalization
- The Survival Benefit: LUMINA and the Canadian Study
- Fewer Flares, Lower Damage Scores
- Blood Clot Reduction and Antiphospholipid Syndrome
- Lipid, Glucose, and Cardiovascular Perks
- The Pregnancy-Safe Anchor Drug
- Dosing: The 5 mg/kg Rule
- Blood Level Monitoring (400-500 ng/mL)
- Retinal Toxicity Screening
- Other Side Effects, Including Skin Hyperpigmentation
- Drug Interactions
- Quinacrine as an Add-On for Refractory Skin Disease
- Chloroquine: The Older Cousin
- Practical Tips for Taking It
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
What Hydroxychloroquine Is and Why It Matters
Hydroxychloroquine — brand name Plaquenil, usually abbreviated HCQ — is the single most important drug in lupus. Every major guideline (ACR, EULAR, the British Society for Rheumatology) says the same thing: every lupus patient who can tolerate it should be on it, for life. It is the "anchor drug," the one you keep taking through remission, through pregnancy, through every other medication change.
It started its career in the 1940s as an antimalarial for American soldiers in the Pacific. Dermatologists noticed that troops taking it had unexpectedly clear skin — their rashes improved. By the 1950s it was being tested in lupus and rheumatoid arthritis, and by the 1990s a landmark trial had proven that stopping it triggers flares. If you have lupus, HCQ is not optional background noise. It is the drug keeping your disease quiet.
And yet patients constantly ask: "If I feel fine, can I stop it?" The answer is almost always no — and this article explains why, in detail, with the actual studies that settled the question.
How It Actually Works: TLR7/9 and Lysosomal Alkalization
Hydroxychloroquine is a weak base. When you swallow a tablet, the molecule drifts through your bloodstream and gets concentrated inside a tiny compartment in every cell called the lysosome — the cell's acidic recycling bin. Normally the lysosome sits at pH 4.5. HCQ soaks up protons and nudges that pH up toward 6. That sounds trivial. It isn't.
Two immune sensors live inside those acidified compartments: Toll-like receptor 7 (TLR7) and Toll-like receptor 9 (TLR9). Their job is to detect viral RNA and DNA. In lupus, they misfire — they bind the patient's own nucleic acids (leaking out of dying cells), mistake them for a virus, and trigger the type I interferon cascade that drives most of the disease. HCQ raises lysosomal pH just enough that TLR7 and TLR9 can no longer engage their ligands properly. The false alarm quiets down. Interferon production drops. B cells stop producing as many autoantibodies.
HCQ also interferes with antigen processing (lysosomes are where your cells chop up proteins for MHC II presentation), dampens cytokine production by macrophages, and stabilizes lysosomal membranes so the enzymes that cause tissue damage stay contained. The net effect is a gentle, broad immune recalibration — not immunosuppression in the classic sense. You do not get more infections on HCQ. In fact, patients on it appear to do better against many viral illnesses.
The Survival Benefit: LUMINA and the Canadian Study
The single most important fact about HCQ is this: it keeps lupus patients alive longer. Two studies nailed this down.
The Canadian Hydroxychloroquine Study Group trial (1991, published in NEJM) was the first randomized, placebo-controlled withdrawal trial. Forty-seven lupus patients in remission on HCQ were randomized either to continue the drug or to switch to placebo. Within six months, patients on placebo flared at 2.5 times the rate of those who stayed on HCQ. The trial was stopped early. Long-term follow-up showed the placebo group had higher damage accrual years later.
The LUMINA cohort (LUpus in MInorities: NAture vs. nurture) — a multi-ethnic U.S. study led by Graciela Alarcón — went further. Following hundreds of Hispanic, African-American, and Caucasian lupus patients, LUMINA found that HCQ use was independently associated with reduced mortality, with hazard ratios around 0.12–0.38 depending on the analysis. Translated: lupus patients on HCQ died at roughly one-fifth to one-third the rate of those not on it, after adjusting for disease severity, ethnicity, and other medications. No other lupus drug has shown a comparable survival signal in observational data.
This is the evidence base behind "never stop Plaquenil." Flare risk, organ damage, cardiovascular events, thrombosis, and all-cause mortality all move in the right direction when HCQ is on board.
Fewer Flares, Lower Damage Scores
Beyond survival, HCQ measurably reduces lupus flare frequency. Pooled data suggest flare rates drop by roughly 50% on continuous HCQ versus off it. Organ damage — measured by the SLICC/ACR Damage Index, which tracks irreversible changes like kidney scarring, avascular necrosis, cataracts, and stroke — accumulates more slowly on HCQ. Patients started on it early in their disease course have better long-term outcomes than those started years in.
This is why rheumatologists frequently prescribe HCQ the same day a lupus diagnosis is made, even before a full workup is complete, and even when symptoms are mild. Starting early pays dividends for decades.
Blood Clot Reduction and Antiphospholipid Syndrome
Lupus patients have a higher baseline risk of blood clots — deep vein thrombosis, pulmonary embolism, stroke, and pregnancy loss — especially those with antiphospholipid antibodies (lupus anticoagulant, anticardiolipin, anti-beta-2-glycoprotein I). HCQ cuts this risk. Multiple cohort studies have shown lower thrombotic event rates on HCQ, with effect sizes in the range of a 30–70% reduction.
The mechanism is several-fold: HCQ inhibits platelet aggregation directly, reduces antiphospholipid antibody binding to cell membranes, and reverses some of the pro-coagulant surface changes that antiphospholipid antibodies cause. For a patient with lupus plus antiphospholipid antibodies, HCQ is a non-negotiable part of the regimen — it is doing work anticoagulants cannot do.
Lipid, Glucose, and Cardiovascular Perks
HCQ has a cluster of metabolic side benefits that surprise most patients:
- Lowers LDL and total cholesterol by roughly 5–15% in most cohorts. The effect is small but real and additive to statins.
- Improves insulin sensitivity. HCQ has been repurposed in small trials as a treatment for type 2 diabetes, where it lowers HbA1c by roughly 0.5 percentage points. In lupus, this matters because chronic steroid use drives insulin resistance — HCQ partly counteracts that.
- Reduces cardiovascular event rates. Lupus patients are at roughly 2–4× general-population risk of heart attack and stroke; HCQ appears to blunt this excess risk.
None of these are reasons to start HCQ on their own — but they are reasons not to stop it because your lupus "feels fine."
The Pregnancy-Safe Anchor Drug
HCQ is one of the very few lupus medications that is explicitly recommended to continue during pregnancy. Stopping it in pregnancy increases flare risk, which is bad for both mother and baby. Continuing it:
- Reduces lupus flares during pregnancy.
- Lowers the risk of neonatal lupus and congenital heart block in babies of mothers with anti-Ro/SSA antibodies (see the Lupus and Pregnancy article).
- Reduces preeclampsia risk.
- Reduces pregnancy loss in antiphospholipid-positive women.
HCQ crosses the placenta and is also secreted in breast milk at low levels, but decades of data show no birth defects, no growth restriction, and no developmental harm. Both the American College of Rheumatology and EULAR strongly recommend continuation throughout pregnancy and breastfeeding. If you are planning pregnancy on HCQ, do not stop it — in fact, if you are not on HCQ and you have lupus, your rheumatologist will very likely start it before conception.
Dosing: The 5 mg/kg Rule
The old dosing instruction was 6.5 mg/kg of ideal body weight, up to 400 mg daily. That dose is now considered too high. In 2016, the American Academy of Ophthalmology (AAO) revised its guidelines in light of data showing that retinal toxicity risk rises sharply above 5 mg/kg of real body weight per day.
Current consensus dosing:
- Maximum: 5 mg/kg/day of real (actual) body weight.
- Most adults: 200–400 mg daily, taken as a single dose or split (200 mg twice daily).
- Tablets: 200 mg scored tablets are standard; 100 mg tablets exist but are less common.
- A 60 kg (132 lb) adult: maximum 300 mg/day — often dosed as 200 mg and 400 mg on alternating days, averaging 300 mg.
- A 50 kg (110 lb) adult: maximum 250 mg/day — typically 200 mg daily, or 200/300 alternating.
If your prescription says 400 mg/day and you weigh less than 80 kg (176 lb), ask your rheumatologist whether the dose can be lowered. Older prescriptions sometimes lag the updated guidelines.
Take HCQ with food — it reduces nausea, which is the main reason patients abandon the drug in the first month. A single evening dose with dinner works well for most people.
Blood Level Monitoring (400-500 ng/mL)
HCQ has a long half-life (about 40 days), so steady-state blood levels take three to six months to reach. A growing number of rheumatologists now order HCQ whole-blood levels to do two things:
- Confirm adherence. A level under about 200 ng/mL suggests the patient is missing doses. Nonadherence is the most common reason for "HCQ failure."
- Target a therapeutic window. Most evidence suggests optimal efficacy at 400–500 ng/mL of blood HCQ. Higher than roughly 1,200 ng/mL may increase retinal toxicity risk; much lower than 500 ng/mL predicts flares.
LabCorp and Quest offer HCQ blood level testing; it is a simple blood draw, usually covered by insurance when ordered by a rheumatologist. If your levels are low despite you taking the drug faithfully, it may be an absorption issue (antacids, sucralfate, and kaolin-pectin reduce HCQ absorption) rather than a dosing failure. If levels are high, a small dose reduction can lower eye toxicity risk without losing efficacy.
Retinal Toxicity Screening
This is the side effect patients fear most, and fair enough — long-term HCQ can, in a minority, damage the retina in a pattern called bull's-eye maculopathy. Once established, the damage does not reverse. But modern screening catches it so early that visible vision loss has become rare.
The AAO 2016 screening schedule:
- Baseline exam within the first year of starting HCQ. This gives your ophthalmologist a reference point and also screens for pre-existing retinal disease that would change the risk calculation.
- No annual screening needed during the first five years on HCQ, unless you have risk factors (see below).
- Annual exams starting at year 5 for everyone on the drug.
Risk factors that warrant earlier annual screening: dose above 5 mg/kg/day, kidney disease (HCQ is renally cleared), concurrent tamoxifen use, or pre-existing macular disease.
The screening test is not a standard eye exam. It must include:
- Spectral-domain optical coherence tomography (SD-OCT) — a high-resolution cross-section of the retina, looking for thinning of the outer nuclear layer (the earliest sign).
- Automated visual field testing — in most patients a 10-2 pattern (testing the central 10 degrees of vision). In Asian patients, a wider 24-2 or 30-2 may be needed because toxicity can start outside the central macula.
- Fundus autofluorescence (FAF) — imaging that picks up early retinal pigment epithelium damage.
When you schedule the appointment, tell the office: "I need Plaquenil screening per the 2016 AAO guidelines — OCT, 10-2 visual field, and fundus autofluorescence." If a clinic says, "We don't do those, but we'll check your vision," that is not a valid screen. Go elsewhere. Most academic and large private ophthalmology practices have the equipment.
At the currently recommended dose (≤5 mg/kg/day), retinal toxicity risk is under 1% at five years, about 2% at ten years, and rises to 20% only after 20+ years of use. If screening catches it early and HCQ is stopped, progression after stopping is minimal.
Other Side Effects, Including Skin Hyperpigmentation
Beyond the retina, HCQ is remarkably well tolerated. The most common issues:
- Nausea and stomach upset. Usually resolves by week 4–6; taking with food handles most cases.
- Skin hyperpigmentation. Roughly 5–25% of long-term users develop slate-gray, blue-black, or brown patches, most often on the shins, forearms, face, or hard palate. Mechanism is thought to be iron and melanin deposition in the skin. It is cosmetic, not dangerous, and often partially fades after stopping HCQ — but the fading is slow and incomplete. Patients should be warned about this before starting.
- Hair changes. Mild hair lightening or thinning in a minority.
- Myopathy and cardiomyopathy. Rare but recognized with very long-term use; muscle weakness should prompt evaluation.
- Hypoglycemia. HCQ can lower blood sugar, occasionally significantly in patients also on insulin or sulfonylureas.
- QT prolongation. At normal lupus doses this is clinically trivial for healthy patients, but becomes relevant when combined with other QT-prolonging drugs (see interactions below).
- Rash. Uncommon; a true HCQ allergy sometimes forces switching to quinacrine.
Drug Interactions
HCQ has relatively few clinically important interactions, but the ones that exist matter:
- Antacids and sucralfate — reduce absorption. Separate by at least four hours.
- Digoxin — HCQ increases digoxin levels; monitor closely.
- Cyclosporine — levels rise on HCQ.
- Metoprolol and other beta-blockers metabolized by CYP2D6 — modest level increases.
- Insulin and sulfonylureas — additive hypoglycemia risk.
- Other QT-prolonging drugs — azithromycin, fluoroquinolones, ondansetron, methadone, certain antipsychotics. At HCQ lupus doses the risk is small but worth mentioning to any prescribing physician.
- Tamoxifen — increases retinal toxicity risk; if both are needed, screen more frequently.
Quinacrine as an Add-On for Refractory Skin Disease
Quinacrine (mepacrine, brand name Atabrine) is the forgotten third antimalarial. It was used extensively during WWII as a malaria prophylactic. It is not manufactured commercially in the United States any more but is readily available from compounding pharmacies for around $50–150/month out of pocket.
Quinacrine is particularly useful for cutaneous lupus that has not responded to HCQ alone. Adding quinacrine 100 mg/day to standard HCQ can clear persistent skin lesions in a substantial fraction of patients. Crucially, quinacrine does not cause retinal toxicity — it does not bind to the retinal pigment epithelium the way HCQ and chloroquine do. So it can be layered on without increasing eye screening burden.
Its main side effect is a yellow skin discoloration (from the drug itself deposited in skin) that can be dramatic and is why some patients decline it. The yellow fades over weeks to months after stopping. Less commonly, quinacrine can cause hematologic side effects (aplastic anemia is rare but reported), so CBC monitoring is standard for the first few months. It is not used in pregnancy.
If your cutaneous lupus is not adequately controlled on HCQ and photoprotection, ask your rheumatologist about adding compounded quinacrine. See also the cutaneous lupus article.
Chloroquine: The Older Cousin
Chloroquine was the original antimalarial used in lupus, before HCQ (a hydroxylated derivative) was shown to have a better safety profile. Chloroquine is more potent mg-for-mg but carries a substantially higher retinal toxicity risk and more GI side effects. It is rarely prescribed for lupus in the United States today — HCQ has essentially replaced it — but it remains common in parts of Latin America, Asia, and Europe. If you travel and are offered chloroquine for malaria prophylaxis while already on HCQ for lupus, do not double up; speak with your rheumatologist about alternatives.
Practical Tips for Taking It
- Take it every single day. Missing doses is the main reason people decide HCQ "isn't working." The drug only works at steady-state levels that take months to build.
- Pair it with a meal. Dinner is the easiest anchor for most people.
- Do not panic if you start it and feel worse for a few days. Some patients have transient nausea or headaches for 1–2 weeks that self-resolve.
- Do not expect rapid improvement. HCQ takes 2–6 months to show its full effect. Joint pain and fatigue are often the first symptoms to budge; skin can take longer.
- Get your baseline eye exam. Schedule it in the first year. Put the year-5 exam on your calendar today.
- Keep your weight current with your prescriber. Dose is weight-based; if you lose 15 pounds the right dose may have changed.
- Ask for a blood level after 6 months. If your level is low despite full adherence, something is blocking absorption — often an antacid or PPI taken too close to the dose.
- Do not stop without your rheumatologist. Even "I feel fine and I want to simplify my pill box" is a conversation to have first, not after.
Key Research Papers
- The Canadian Hydroxychloroquine Study Group. A randomized study of the effect of withdrawing hydroxychloroquine sulfate in systemic lupus erythematosus. N Engl J Med. 1991.
- Alarcón GS, et al. Effect of hydroxychloroquine on the survival of patients with systemic lupus erythematosus: data from LUMINA, a multiethnic US cohort. Ann Rheum Dis. 2007.
- Marmor MF, et al. Recommendations on screening for chloroquine and hydroxychloroquine retinopathy (2016 revision). Ophthalmology. 2016.
- Melles RB, Marmor MF. The risk of toxic retinopathy in patients on long-term hydroxychloroquine therapy. JAMA Ophthalmol. 2014.
- Fanouriakis A, et al. 2019 update of the EULAR recommendations for the management of systemic lupus erythematosus. Ann Rheum Dis. 2019.
- Ruiz-Irastorza G, et al. Effect of antimalarials on thrombosis and survival in patients with systemic lupus erythematosus. Lupus. 2006.
- Costedoat-Chalumeau N, et al. Hydroxychloroquine in systemic lupus erythematosus: results of a French multicentre controlled trial (PLUS Study). Ann Rheum Dis. 2013.
- Petri M. Use of hydroxychloroquine to prevent thrombosis in systemic lupus erythematosus and in antiphospholipid antibody-positive patients. Curr Rheumatol Rep. 2011.
Research Papers
For further reading, these PubMed topic searches surface current peer-reviewed work on hydroxychloroquine pharmacology, monitoring, and use in lupus:
- Hydroxychloroquine and lupus survival
- Hydroxychloroquine mechanism: TLR7 and TLR9 inhibition
- Hydroxychloroquine retinal toxicity and screening
- Hydroxychloroquine blood level monitoring
- Hydroxychloroquine in lupus pregnancy
- Hydroxychloroquine and antiphospholipid-related thrombosis
- Quinacrine for cutaneous lupus
- Hydroxychloroquine effects on lipids and glucose
Connections
- Lupus Overview
- Lupus and Pregnancy
- Cutaneous Lupus and Photoprotection
- Neuropsychiatric Lupus
- ACR-EULAR 2019 Classification Criteria
- Biologics: Belimumab, Anifrolumab and Rituximab
- Lupus-Sjogren's Overlap
- ANA, dsDNA and Lupus Autoantibodies
- Lupus Nephritis and Kidney Involvement
- Arthritis
- Sjogren's Syndrome
- Kidney Disease
- Stroke
- Cataracts
- Malaria