Lupus and Pregnancy
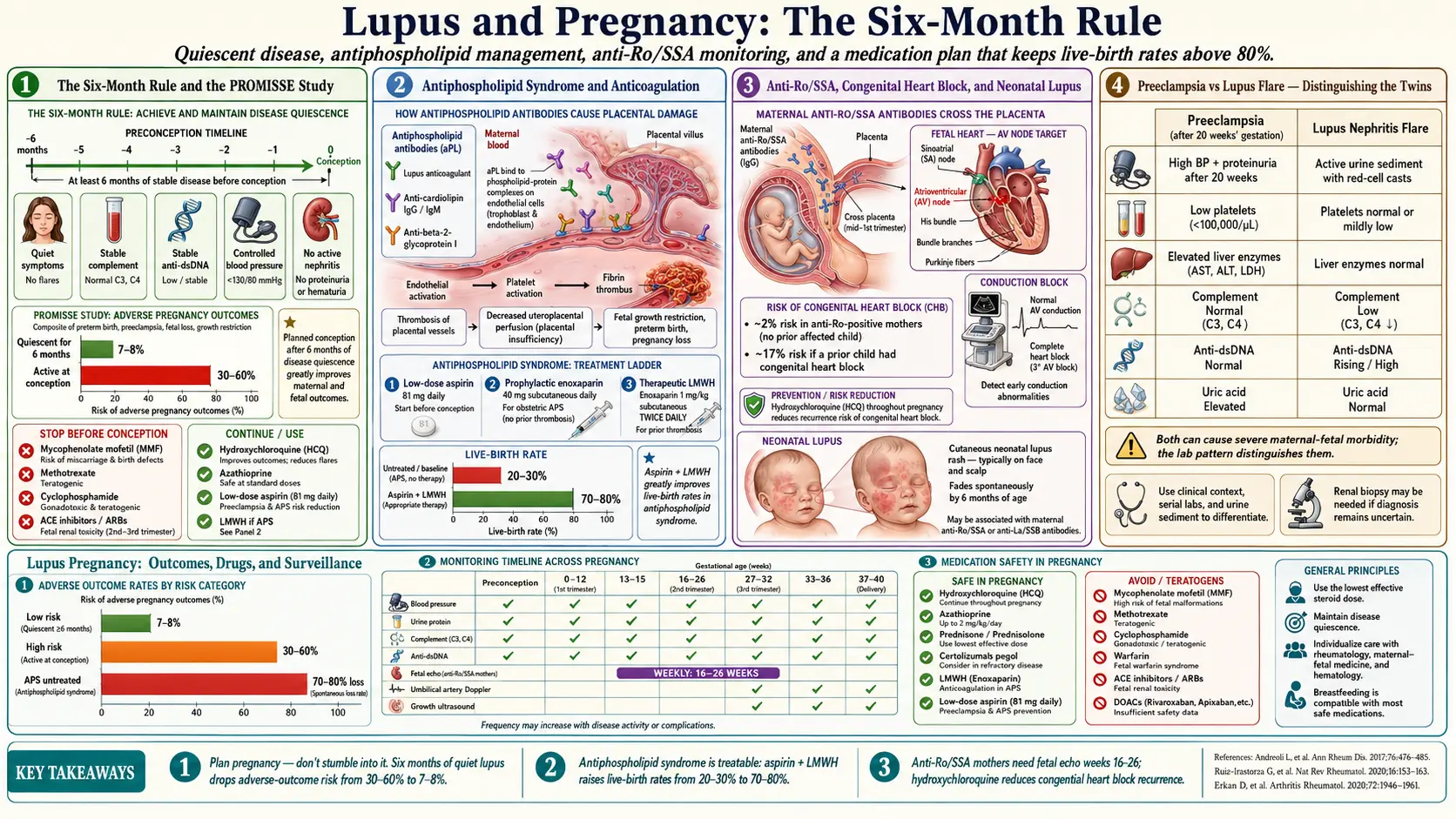
Table of Contents
- Why This Matters
- Preconception — The Six-Month Rule
- Risk Stratification — Who Is High-Risk?
- Antiphospholipid Syndrome and Anticoagulation
- Anti-Ro/SSA, Congenital Heart Block, and Neonatal Lupus
- Medications to Continue
- Medications to Stop Before Conception
- Biologics — Belimumab, Rituximab, and Anifrolumab
- Preeclampsia vs. Lupus Flare
- Prenatal Monitoring Schedule
- Delivery and Postpartum
- Breastfeeding
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
Why This Matters
If you have lupus and you want children, the good news up front: most pregnancies in women with lupus end with a healthy baby and a healthy mom. Twenty years ago rheumatologists routinely told lupus patients not to get pregnant. That advice is outdated. With modern risk-stratification and the right medication plan, live-birth rates exceed 80% — and in low-risk patients they approach the general-population rate.
But lupus pregnancy is not ordinary pregnancy. It sits at a complicated intersection: an autoimmune disease that can flare, a placenta that can fail, a panel of autoantibodies that can cross into fetal circulation, a list of medications that must be reshuffled, and a pregnancy complication (preeclampsia) that looks almost identical to one of the things lupus does on its own. The risks are real and include preeclampsia, preterm birth, intrauterine growth restriction, pregnancy loss, congenital heart block, and neonatal lupus. Nearly every one of those risks can be dramatically reduced by planning pregnancy carefully, staying on the right drugs, and getting watched closely by a team that knows what they are doing.
This article is the patient-facing version of what a maternal-fetal medicine specialist and rheumatologist would sit down and explain to you over an hour in a co-management clinic. Nothing here replaces that conversation — but it will help you show up prepared, ask the right questions, and catch errors when they happen.
Preconception — The Six-Month Rule
The single most important predictor of how your pregnancy will go is how stable your lupus was in the six months before conception. This is not a soft guideline. It is the finding that has been confirmed again and again, most rigorously in the PROMISSE study (Predictors of Pregnancy Outcome: Biomarkers in Antiphospholipid Antibody Syndrome and Systemic Lupus Erythematosus), a prospective multicenter cohort of several hundred lupus pregnancies.
What PROMISSE showed:
- Women whose lupus had been quiescent for at least six months before conception had adverse-pregnancy-outcome rates of roughly 7–8%.
- Women with active disease at conception had adverse-outcome rates of 30–60%, depending on what was active.
- The strongest predictors of a bad outcome were lupus anticoagulant positivity, active disease at baseline, low platelets, low complement, and non-white race/ethnicity (a signal for access-to-care and social-determinant issues as much as biology).
So the practical rule: plan the pregnancy, don't stumble into it. Six months of quiet lupus — no flare, stable labs, controlled blood pressure, safe medications — is the door you want to walk through before you start trying. If you are newly diagnosed, just off a flare, or still titrating medications, the right answer is usually contraception for now, pregnancy later.
"Quiescent" does not mean "feels fine." It means objective stability: no new clinical activity, stable or rising C3/C4 complement, stable or falling anti-dsDNA, normal blood pressure, no active nephritis on urine studies, and a SLEDAI or physician global assessment that your rheumatologist calls low.
Risk Stratification — Who Is High-Risk?
Before you conceive, you and your rheumatologist should run through a checklist. Any one of the following shifts you from "standard" to "high-risk" pregnancy and changes how closely you'll be monitored:
- Active lupus within the last 6 months — especially active nephritis.
- History of lupus nephritis, even if currently in remission. Pregnancy can unmask residual inflammation, and prior nephritis raises the risk of preeclampsia 2–3 fold. See Lupus Nephritis.
- Antiphospholipid antibody positivity — lupus anticoagulant, anticardiolipin IgG/IgM, or anti-beta-2-glycoprotein-I IgG/IgM. Triple-positive patients are the highest-risk subgroup.
- Anti-Ro/SSA or anti-La/SSB positivity — raises the risk of neonatal lupus and congenital heart block.
- Hypertension at baseline, or proteinuria, or elevated creatinine.
- Prior poor obstetric history — previous late miscarriage, stillbirth, severe preeclampsia, or preterm delivery under 34 weeks.
- Pulmonary hypertension, severe restrictive lung disease, heart failure, or advanced chronic kidney disease (creatinine > 2.8 mg/dL). These are the rare scenarios where pregnancy is considered medically contraindicated because maternal mortality is unacceptably high.
Most patients fall into the "standard-risk lupus pregnancy" category: well-controlled disease, no antiphospholipid antibodies, normal kidneys, normal blood pressure. These pregnancies do extremely well. High-risk pregnancies still usually do well — but they need a maternal-fetal medicine specialist co-managing with your rheumatologist from week one.
Antiphospholipid Syndrome and Anticoagulation
Antiphospholipid antibodies (aPL) — lupus anticoagulant, anticardiolipin, and anti-beta-2-glycoprotein-I — are present in roughly 30–40% of lupus patients. A smaller subset (maybe a third of those) meet clinical criteria for antiphospholipid syndrome (APS), which requires both persistent antibody positivity and a history of thrombosis or pregnancy morbidity (recurrent early loss, fetal loss after 10 weeks, or severe preeclampsia / placental insufficiency requiring preterm delivery).
APS in pregnancy is treatable, and the treatment works. The standard regimen:
- Low-dose aspirin 81 mg daily, ideally started before conception and continued throughout pregnancy and into the early postpartum period.
- Prophylactic low-molecular-weight heparin (LMWH) — typically enoxaparin 40 mg subcutaneously once daily — for women with obstetric APS (recurrent loss or prior late fetal loss) but no prior thrombosis.
- Therapeutic-dose LMWH — enoxaparin 1 mg/kg twice daily — for women with a prior thrombotic event.
Aspirin plus LMWH takes live-birth rates in APS pregnancy from roughly 20–30% with no treatment to 70–80% with treatment. That is one of the most powerful obstetric interventions in modern medicine. Warfarin is teratogenic and is not used during pregnancy; direct oral anticoagulants (DOACs like apixaban, rivaroxaban) are not recommended because safety data are inadequate. LMWH, by contrast, does not cross the placenta and has decades of pregnancy safety data.
Aspirin 81 mg is also now recommended by ACOG and the USPSTF for all lupus patients starting in the first trimester (ideally by 12–16 weeks), regardless of antiphospholipid status, because lupus itself is an independent risk factor for preeclampsia. Many rheumatologists start it at the positive pregnancy test.
Anti-Ro/SSA, Congenital Heart Block, and Neonatal Lupus
About 30–40% of lupus patients are positive for anti-Ro/SSA antibodies, and a subset also have anti-La/SSB. These antibodies cross the placenta freely starting around week 12. In the fetus, they can bind cardiac tissue and trigger two distinct problems:
Neonatal cutaneous lupus. A transient rash — often on the face and scalp, sometimes triggered by light exposure in the newborn nursery — that appears in the first few weeks of life and resolves spontaneously by 6–8 months as maternal antibodies clear the baby's circulation. It looks alarming but leaves no lasting damage.
Congenital heart block (CHB). The serious one. Maternal anti-Ro antibodies damage the fetal atrioventricular (AV) node, producing complete heart block that is usually permanent. It affects roughly 2% of first pregnancies in anti-Ro-positive women, and the recurrence rate in a subsequent pregnancy after an affected child is 15–20%. Most affected babies need a pacemaker, often within the first year of life. The mortality rate is around 15–20%.
The critical window is weeks 16 to 26 — that's when the fetal conduction system develops and when antibody-mediated damage occurs. Standard practice in anti-Ro-positive pregnancies:
- Weekly or every-two-week fetal echocardiograms from 16 to 26 weeks, then every 2–4 weeks through 34 weeks.
- Monitor the fetal PR interval (a measure of AV conduction time). A prolonging PR interval may represent first-degree block and a chance to intervene before complete block sets in.
- If first-degree or second-degree block is caught early, treatment with dexamethasone or IVIG plus hydroxychloroquine may halt progression in some cases, though evidence is mixed and dexamethasone has its own fetal-growth costs.
- Hydroxychloroquine (HCQ) taken by the mother throughout pregnancy has been shown in the PATCH trial and other studies to cut the recurrence rate of CHB roughly in half in anti-Ro-positive women who had a prior affected child. This is one of the strongest reasons to stay on HCQ.
Medications to Continue
The most dangerous thing you can do before or during a lupus pregnancy is stop your medications out of a vague fear that "drugs are bad for the baby." Untreated lupus is far more dangerous to the baby than the drugs below. The current EULAR and ACR guidelines are clear:
Hydroxychloroquine (HCQ) — continue, and this is not optional. HCQ crosses the placenta but does not harm the fetus. It reduces lupus flares, reduces preeclampsia, reduces preterm birth, reduces the risk of congenital heart block in anti-Ro-positive women, and improves live-birth rates. Stopping HCQ in pregnancy is associated with a measurable increase in flares and worse outcomes. Dose is usually 200–400 mg daily. See Hydroxychloroquine and Antimalarials.
Azathioprine — compatible. The fetal liver lacks the enzyme (inosinate pyrophosphorylase) needed to convert azathioprine to its active metabolite, so fetal drug exposure is limited. Azathioprine up to 2 mg/kg/day is standard for lupus nephritis maintenance in pregnancy and is compatible with breastfeeding.
Tacrolimus — compatible. Often used in active or prior lupus nephritis during pregnancy. Monitor trough levels monthly because pregnancy changes drug clearance.
Prednisone — use the lowest effective dose. Prednisone is extensively metabolized by placental 11-beta-hydroxysteroid dehydrogenase, so only about 10% reaches the fetus. Low doses (under 10–15 mg/day) are generally considered acceptable. Higher doses carry real risks: maternal hypertension, gestational diabetes, preeclampsia, preterm rupture of membranes, and cleft palate if used in high doses in the first trimester. Use prednisone for flares, not as a ceiling. Dexamethasone and betamethasone do cross the placenta and are reserved for fetal indications (lung maturity before preterm delivery, or congenital heart block treatment).
Cyclosporine — compatible. Less commonly used than tacrolimus but acceptable.
Sulfasalazine — compatible. Rarely used in lupus but worth knowing.
Low-dose aspirin 81 mg — start it. See the APS and preeclampsia-prevention discussion above.
LMWH — use as indicated. Does not cross the placenta.
Medications to Stop Before Conception
Several lupus drugs are clearly teratogenic or associated with serious fetal harm. These need to be stopped before you start trying, with washout periods built into the timing.
Methotrexate — STOP. Causes miscarriage and a specific pattern of birth defects (craniofacial, limb, CNS). Washout: 3 months before conception (the drug sits in tissues well beyond its plasma half-life). If you are on methotrexate and planning pregnancy, switch to azathioprine or another compatible agent three months before you start trying, and supplement with folic acid throughout the transition.
Mycophenolate mofetil (MMF, CellCept) — STOP. A potent teratogen. Causes a characteristic pattern of congenital malformations (cleft lip/palate, ear malformations, cardiac defects, microtia) and early miscarriage. This is one of the most important drug-switch conversations a lupus nephritis patient will have. Washout: 6 weeks minimum, but most specialists want the switch done and the patient stable on azathioprine or tacrolimus for 3–6 months before conception. Both partners should use contraception while either is on MMF.
Cyclophosphamide — STOP. Teratogenic, causes infertility, and is reserved for severe disease. Not used in pregnancy except in true life-threatening maternal indications after the first trimester.
Leflunomide — STOP with an active washout. Leflunomide's active metabolite has a half-life of weeks and can persist for up to two years. The washout protocol uses cholestyramine 8 g three times daily for 11 days, followed by two separate blood levels (at least 14 days apart) showing serum concentrations below 0.02 mg/L, before conception is safe.
Warfarin — switch to LMWH. Teratogenic in the first trimester (warfarin embryopathy) and raises fetal bleeding risk later. Transition to therapeutic-dose LMWH before conception or at the positive pregnancy test.
ACE inhibitors and ARBs — STOP. Cause fetal renal dysgenesis, oligohydramnios, and lung hypoplasia, particularly in the second and third trimesters. Substitute labetalol, nifedipine, or methyldopa for blood pressure control.
NSAIDs — limited use. Avoid after 20 weeks (FDA warning: oligohydramnios and renal dysfunction) and definitely after 30 weeks (premature closure of the ductus arteriosus). Short-term use in the first and early second trimester is generally acceptable if clearly needed.
Biologics — Belimumab, Rituximab, and Anifrolumab
The biologic lupus drugs are newer and pregnancy data are thinner. The general framework: IgG monoclonal antibodies cross the placenta actively via the neonatal Fc receptor starting in the second trimester and rising steeply through the third. That means late-pregnancy dosing delivers the highest fetal exposure — which matters for vaccination decisions in the baby's first six months (live vaccines should be delayed in infants exposed to biologics late in pregnancy).
Belimumab (Benlysta). An anti-BLyS monoclonal antibody. Pregnancy safety data are limited but expanding. The manufacturer's pregnancy registry and recent cohort analyses do not show a clear teratogenic signal. Current practice: avoid starting belimumab in a woman trying to conceive, but if a patient is stable and well-controlled on it and pregnancy occurs, shared decision-making often continues it, sometimes dropping the last dose in the third trimester. See Biologics in Lupus.
Rituximab. An anti-CD20 monoclonal. First-trimester exposure is generally considered low-risk (placental transfer is minimal before week 16). Avoid in the second and third trimesters: rituximab crosses the placenta heavily in later pregnancy and causes transient B-cell depletion in the newborn, which can last several months and interferes with vaccine responses and infection defense. If rituximab is medically necessary, time infusions so that the last dose is at least several months before conception when possible.
Anifrolumab. A type-I interferon receptor antibody approved for moderate-to-severe SLE in 2021. Pregnancy data are essentially absent. Current guidance: avoid during pregnancy. Women of reproductive age on anifrolumab should use effective contraception, and the drug should be stopped before conception with a reasonable washout.
Preeclampsia vs. Lupus Flare
This is the single hardest clinical problem in lupus pregnancy, and understanding it will help you advocate for yourself when you're in triage at 3 a.m. in the third trimester. Both preeclampsia and a lupus flare can present with:
- Rising blood pressure
- Proteinuria
- Edema
- Thrombocytopenia
- Headache
- A feeling of "something is wrong"
They require opposite treatments. Preeclampsia is treated by delivering the baby — the placenta is the problem. A lupus flare is treated by intensifying immunosuppression, and delivering the baby won't fix it. Getting this wrong in either direction is harmful.
Features that suggest preeclampsia over lupus flare:
- Onset after 20 weeks (preeclampsia is very rare before 20 weeks except in the setting of molar pregnancy or severe APS)
- New or rising uric acid
- Rising liver transaminases (AST/ALT) — the "H" and "EL" of HELLP syndrome
- Normal or rising complement (C3, C4)
- Normal or stable anti-dsDNA titers
- No active findings elsewhere (no new rash, no synovitis, no serositis, no oral ulcers)
- Rapid resolution within 24–48 hours of delivery
Features that suggest a lupus flare:
- Falling complement (C3 and C4 drop)
- Rising anti-dsDNA
- Active urinary sediment (red-cell casts, dysmorphic RBCs)
- Low C1q level or positive anti-C1q antibodies
- Extra-renal lupus activity: new rash, arthritis, oral ulcers, pleuritis, pericarditis, hair loss
- Onset before 20 weeks
- No improvement after delivery
In practice, patients often have both — preeclampsia and a lupus flare can and do coexist, especially in women with prior lupus nephritis. That's one reason the co-management model (rheumatologist plus maternal-fetal medicine plus nephrologist if needed) is standard of care.
Prenatal Monitoring Schedule
A typical lupus pregnancy monitoring plan, modified for risk level:
- Preconception: baseline CBC, creatinine, urinalysis, spot urine protein/creatinine ratio, C3, C4, anti-dsDNA, anti-Ro/SSA, anti-La/SSB, full antiphospholipid panel (lupus anticoagulant, anticardiolipin IgG/IgM, anti-beta-2-GPI IgG/IgM), TSH, vitamin D. Control blood pressure. Optimize HCQ. Stop teratogens with washout.
- First trimester: early dating ultrasound; confirm medication list; start aspirin 81 mg by 12 weeks; repeat labs above at 8–12 weeks.
- Second trimester: detailed anatomy scan at 18–20 weeks; lupus labs every 4–6 weeks (or monthly if high-risk); uterine artery Dopplers around 20–24 weeks in high-risk patients; fetal echocardiography weekly or biweekly from week 16 to week 26 in anti-Ro-positive women, then every 2–4 weeks until 34 weeks.
- Third trimester: growth ultrasounds every 3–4 weeks (lupus raises the risk of intrauterine growth restriction); antenatal fetal testing (non-stress tests, biophysical profiles) starting around 32 weeks in high-risk patients; continue lupus labs monthly.
- Delivery planning: most lupus patients can aim for vaginal delivery at term. Cesarean for obstetric indications. Stress-dose steroids are given during labor and delivery if the mother has been on prednisone greater than 5 mg/day for more than 3 weeks.
Delivery and Postpartum
The postpartum period is a high-risk window for lupus flare. Rates of flare in the first 3–6 months after delivery are higher than the baseline flare rate, driven partly by immune-system rebound and partly by sleep deprivation, stress, and breastfeeding hormones.
Expect:
- A rheumatology visit within 4–6 weeks after delivery (sooner if symptoms).
- Continued aspirin 81 mg for 1–6 weeks postpartum.
- Continued LMWH for 6 weeks postpartum in APS patients (the highest thrombosis risk of the whole pregnancy cycle is the first six postpartum weeks).
- Contraception counseling. Estrogen-containing contraceptives (combined pills, patch, ring) are contraindicated in APS-positive women and relatively contraindicated in active lupus because of thrombosis risk. Progestin-only pills, the levonorgestrel IUD, the copper IUD, and the subdermal implant are all safe. Discuss this before discharge.
Breastfeeding
Most lupus medications are compatible with breastfeeding at the doses used in clinical practice. Specifically:
- Hydroxychloroquine — compatible. Minimal infant exposure via breast milk.
- Azathioprine — compatible. Active metabolite levels in breast milk are negligible.
- Tacrolimus, cyclosporine — compatible.
- Prednisone — compatible. At doses over 20 mg/day, some clinicians recommend waiting 4 hours between a dose and a feed, though this is more tradition than strong evidence.
- NSAIDs (ibuprofen, etc.) — compatible short-term; prefer ibuprofen over others.
- Warfarin — compatible (does not enter breast milk in clinically significant amounts).
- LMWH — compatible (too large to pass into milk, and not orally absorbed by the infant anyway).
- Belimumab, rituximab, anifrolumab — limited data; IgG antibodies in breast milk are mostly digested in the infant gut, so transfer is thought to be low, but shared decision-making is required.
- Methotrexate, mycophenolate, cyclophosphamide, leflunomide — contraindicated during breastfeeding, the same way they are in pregnancy.
For infants whose mothers received biologics late in pregnancy, live vaccines (rotavirus, later MMR) should be deferred until the baby's drug level has cleared — typically at least 6 months of age, sometimes longer depending on the drug. Inactivated vaccines (DTaP, Hib, pneumococcal, hepatitis B, inactivated polio) are given on the normal schedule.
Key Research Papers
- Buyon JP, Kim MY, Guerra MM, et al. Predictors of pregnancy outcomes in patients with lupus: a cohort study. Ann Intern Med. 2015 (PROMISSE).
- Andreoli L, Bertsias GK, Agmon-Levin N, et al. EULAR recommendations for women's health and the management of family planning, assisted reproduction, pregnancy and menopause in patients with SLE and/or antiphospholipid syndrome. Ann Rheum Dis. 2017.
- Sammaritano LR, Bermas BL, Chakravarty EE, et al. 2020 American College of Rheumatology Guideline for the Management of Reproductive Health in Rheumatic and Musculoskeletal Diseases. Arthritis Rheumatol. 2020.
- Izmirly P, Kim M, Friedman DM, et al. Hydroxychloroquine to prevent recurrent congenital heart block in fetuses of anti-SSA/Ro-positive mothers. J Am Coll Cardiol. 2020 (PATCH).
- Empson M, Lassere M, Craig J, Scott J. Prevention of recurrent miscarriage for women with antiphospholipid antibody or lupus anticoagulant. N Engl J Med. 2003;349(12):1133–1138.
- Fanouriakis A, Kostopoulou M, Andersen J, et al. EULAR recommendations for the management of systemic lupus erythematosus: 2023 update. Ann Rheum Dis. 2024.
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on lupus pregnancy:
- PROMISSE and lupus pregnancy outcomes
- Antiphospholipid syndrome, aspirin, and LMWH in pregnancy
- Anti-Ro/SSA and congenital heart block
- Hydroxychloroquine in lupus pregnancy
- Mycophenolate teratogenicity
- Preeclampsia vs. lupus flare differentiation
- Belimumab pregnancy registry data
- Lupus nephritis and pregnancy outcomes
- Neonatal cutaneous lupus
- Lupus medications and breastfeeding
Connections
- Lupus Overview
- Hydroxychloroquine and Antimalarials
- Biologics: Belimumab, Anifrolumab and Rituximab
- Lupus Nephritis and Kidney Involvement
- Preeclampsia
- Neuropsychiatric Lupus
- ACR-EULAR 2019 Classification Criteria
- Cutaneous Lupus and Photoprotection
- Lupus-Sjogren's Overlap
- ANA, dsDNA and Lupus Autoantibodies
- Sjogren's Syndrome
- Pregnancy and Hashimoto's
- Pregnancy and IBD
- Gestational Diabetes
- Kidney Disease