GI Involvement in EDS
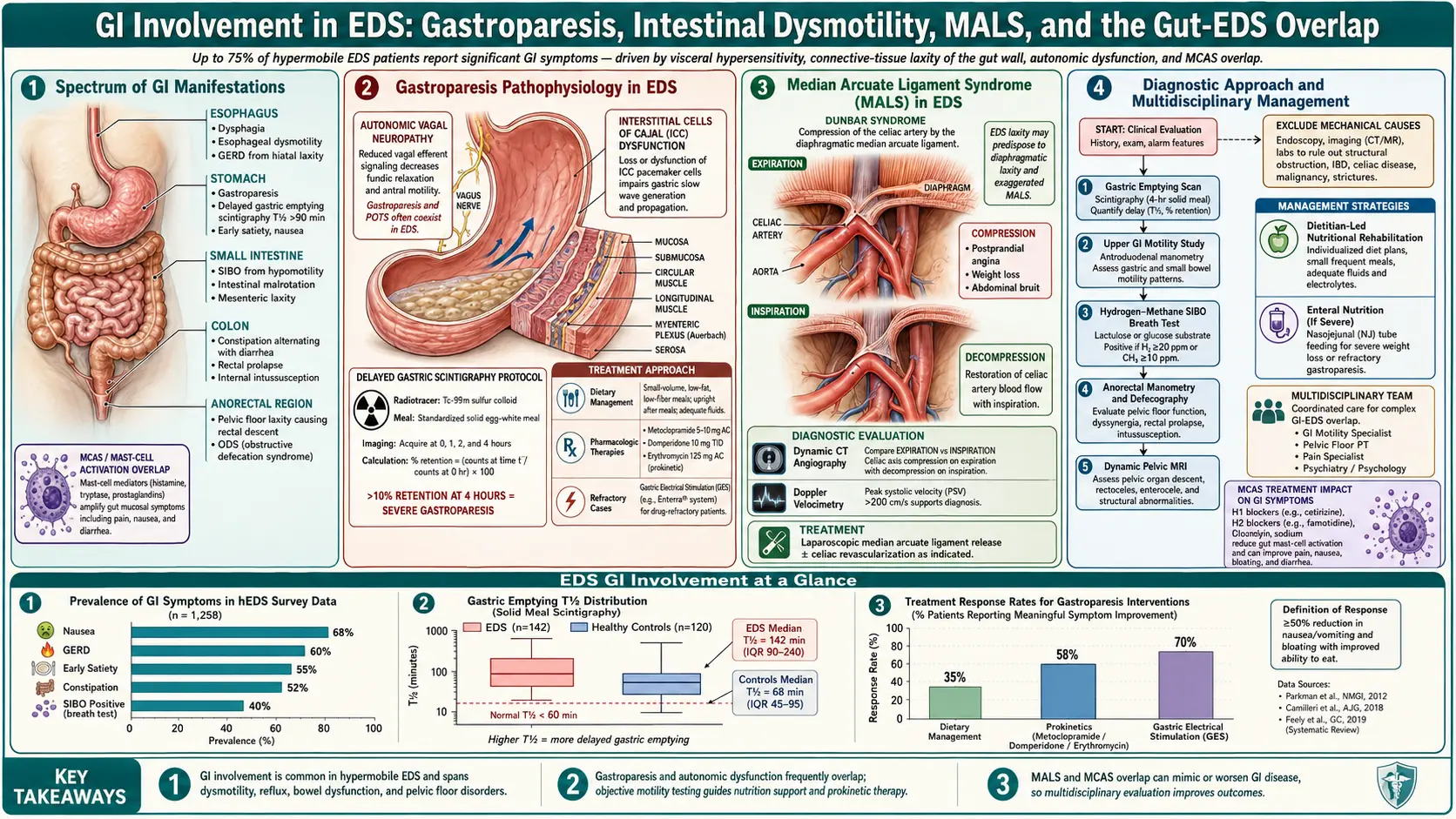
Table of Contents
- Why the EDS Gut Struggles
- Gastroparesis — The Stomach That Won’t Empty
- SIBO Overlap
- IBS-Type Symptoms & Functional Dyspepsia
- GERD, Hiatal Hernia, and PPI Cautions
- Organ Prolapse and Hernias
- Early Diverticular Disease
- The Dysautonomic Gut — POTS Effects
- Mast Cell GI Symptoms
- Eosinophilic Esophagitis and Gastritis
- Median Arcuate Ligament Syndrome (MALS)
- Superior Mesenteric Artery Syndrome
- Dysmotility Workup
- Nutrition Strategies
- vEDS and Spontaneous Bowel Rupture
- Surgery & Anesthesia Considerations
- Building Your Care Team
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
Why the EDS Gut Struggles
If you have Ehlers-Danlos syndrome and your gut is a daily problem — you are not imagining it, and you are not alone. Studies of hypermobile EDS (hEDS) cohorts consistently find gastrointestinal symptoms in 56–75% of patients. Fikree and colleagues at King’s College London documented this in their 2015 cohort study, and every major EDS clinic since has confirmed it. GI complaints are the second most common reason, after joint pain, that hEDS patients seek medical care.
The reason is mechanical and neurological at once. Your gut is a long tube of smooth muscle wrapped in collagen-rich connective tissue, suspended by collagen-rich ligaments, and coordinated by an autonomic nervous system that also happens to be dysregulated in most EDS patients. When the collagen scaffold is faulty, the tube stretches, sags, and empties unevenly. When the autonomic wiring is off, the tube’s peristaltic rhythm breaks down. You end up with a gut that is simultaneously too floppy and too slow.
This article walks through every layer of that dysfunction — from the stomach that won’t empty to the pelvic floor that won’t hold — and what actually helps.
Gastroparesis — The Stomach That Won’t Empty
Gastroparesis is delayed gastric emptying in the absence of mechanical obstruction. In plain language: your stomach is a bag that has forgotten how to squeeze on schedule. Food sits. You feel full after three bites. An hour later you are nauseous. Two hours later you are still full. By dinner you feel bloated from breakfast.
The classic EDS gastroparesis picture:
- Early satiety — full after small amounts.
- Postprandial bloating and nausea — worst 30–90 minutes after eating.
- Upper abdominal fullness that does not respond to bowel movements.
- Unintentional weight loss in moderate-to-severe cases.
- Reflux symptoms as undigested food sits and pressurizes the lower esophageal sphincter.
The diagnostic gold standard is the 4-hour gastric emptying scintigraphy scan. You eat a standardized egg-white meal labeled with technetium-99m, and a camera tracks how much remains in your stomach at 1, 2, and 4 hours. More than 10% retention at 4 hours is abnormal; more than 35% is moderate; more than 90% is severe. The older 90-minute version of this test is unreliable — insist on the 4-hour protocol.
Treatment That Actually Helps
Meal structure is the first lever, and often the most effective one:
- Small frequent meals — five or six small plates a day beat three large ones. Aim for 300–400 calories per meal.
- Soft and liquid phase during flares — smoothies, soups, yogurt, mashed vegetables, protein shakes. Liquids empty faster than solids and faster than fat.
- Lower fat and lower fiber during bad weeks — both slow gastric emptying. This is the one situation where low fiber is therapeutic.
- Upright posture for 60–90 minutes after eating — gravity helps.
Prokinetics are drugs that nudge the stomach to squeeze. None are perfect, and each has trade-offs:
- Metoclopramide (Reglan) — the FDA-approved option. Effective, but carries a black-box warning for tardive dyskinesia (a sometimes-irreversible movement disorder) if used continuously beyond 12 weeks. Use short courses or intermittent pulses.
- Erythromycin — an antibiotic that, at low doses, acts as a motilin-receptor agonist and drives a strong stomach contraction. The catch: tachyphylaxis (loss of effect) within 4–6 weeks of daily use. Most motility specialists dose it in pulses — two weeks on, two weeks off — or only before the largest meal of the day.
- Prucalopride (Motegrity) — a 5-HT4 agonist originally approved for chronic constipation; off-label use for gastroparesis is growing, especially when constipation coexists.
- Domperidone — not FDA-approved in the U.S. but available through compounding pharmacies with an Investigational New Drug permit. Comparable efficacy to metoclopramide without the neurological risk. Requires baseline ECG because of QT-prolongation potential.
Non-drug adjuncts that some patients find useful: ginger (250–1000 mg before meals), peppermint oil capsules, acupressure at the P6 (Neiguan) point on the inner wrist, and gastric-emptying-friendly supplements like Iberogast (STW 5).
SIBO Overlap
Small intestinal bacterial overgrowth is so common in hEDS that some motility clinics now screen for it routinely. Studies place SIBO in more than 50% of hEDS patients — several times the general-population rate. The mechanism is mechanical: slow small-bowel transit gives bacteria time to settle and multiply where they don’t belong.
The overlap with gastroparesis is direct — the stomach empties slowly, the small bowel receives intermittent boluses, and the migrating motor complex (the housekeeping wave that sweeps the gut clean between meals) runs weak or absent. Bacteria ferment carbohydrates, produce hydrogen and methane, and you bloat within 30 minutes of eating anything starchy or sweet.
Diagnosis is a 3-hour lactulose or glucose breath test. Treatment begins with rifaximin (550 mg three times daily for 14 days), often combined with neomycin or metronidazole if methane predominates. Relapse is common — which is why EDS patients often cycle through multiple protocols and benefit from prokinetics and elemental diets between rounds.
For a full SIBO workup, see the SIBO overview, the Rifaximin Protocols deep dive, and the Elemental Diet deep dive.
IBS-Type Symptoms & Functional Dyspepsia
Many hEDS patients carry an IBS label from earlier in their medical journey — alternating constipation and diarrhea, bloating that waxes with stress, cramping relieved by a bowel movement. That IBS label is often correct, but it is incomplete: the underlying driver is frequently a combination of dysmotility, SIBO, autonomic dysfunction, and mast-cell reactivity, any of which is individually treatable.
Zeitoun and colleagues (2013) reported that hEDS patients with IBS-type symptoms had measurably abnormal colonic transit and rectal sensation compared to IBS patients without EDS — meaning the gut is not behaving normally and “functional” does not mean “in your head.”
Functional dyspepsia — upper abdominal discomfort, early satiety, postprandial distress — overlaps heavily with mild gastroparesis and is often treated as the same problem. If the 4-hour scintigraphy is normal but symptoms are classic, trial a prokinetic anyway; many patients improve.
Full workup and subtype-directed therapy live on the IBS hub, including the Rome IV subtype breakdown and the IBS Medications Guide.
GERD, Hiatal Hernia, and PPI Cautions
Reflux is nearly universal in hEDS. The lower esophageal sphincter (LES) is a collagen-reinforced ring; when the collagen is faulty, the ring is lax. Stomach contents reflux easily, especially if gastroparesis is also pressurizing the stomach from below. Hiatal hernia — where the upper stomach slides through the diaphragm into the chest — is more common in EDS than in the general population, for the same connective-tissue reason.
Symptoms include heartburn, regurgitation, chronic cough, hoarseness, chest pain that mimics cardiac pain, and dental erosion. Long-standing untreated reflux raises the risk of Barrett’s esophagus — a precancerous change in the esophageal lining. Endoscopic screening every 3–5 years is reasonable if reflux has been ongoing for more than a decade.
PPI Trade-Offs
Proton-pump inhibitors (omeprazole, pantoprazole, esomeprazole) work well for reflux but are not benign long-term. In EDS patients specifically, two concerns loom larger than in the general population:
- Calcium absorption. PPIs reduce gastric acid, which reduces ionization of dietary calcium. EDS patients already trend toward lower bone density; adding a PPI compounds the risk. If long-term PPI use is necessary, supplement with calcium citrate (which absorbs acid-independently) rather than calcium carbonate.
- SIBO risk. Gastric acid is the first line of defense against bacterial overgrowth. Suppress it for years and you invite SIBO into a gut that is already mechanically vulnerable.
The pragmatic approach: use a PPI at the lowest effective dose, consider H2 blockers (famotidine) for milder reflux, elevate the head of the bed 6–8 inches, stop eating three hours before lying down, and revisit whether the PPI is still needed every 6–12 months.
Organ Prolapse and Hernias
Collagen holds organs where they belong. When the collagen is faulty, gravity and pressure eventually win. Nelson and colleagues (2015) documented elevated rates of pelvic-organ prolapse, rectal prolapse, and hernia across EDS subtypes.
- Rectal prolapse — the rectum slides through the anus, especially with straining. Can occur decades earlier than in non-EDS patients.
- Uterine prolapse and cystocele — common postpartum and post-menopause, but also seen in nulliparous young women with hEDS.
- Internal intussusception — the rectum telescopes into itself, causing incomplete evacuation, pelvic pressure, and the sensation of a blockage that won’t pass. Diagnosed by defecography (MRI or fluoroscopic).
- Hernias — umbilical, inguinal, incisional. All heal poorly because the surrounding tissue is friable. Surgical repair in EDS patients has a higher recurrence rate, and mesh is often preferred over suture repair alone.
Pelvic-floor physical therapy is the first-line conservative treatment for prolapse. Surgical repair is reserved for symptomatic cases and should be performed by a surgeon who understands EDS tissue fragility.
Early Diverticular Disease
Diverticula are small out-pouchings of the colon wall where the muscle layer is thin. In the general population, they are rare before age 40. In EDS, earlier-onset diverticular disease is well-documented — sometimes appearing in patients in their 20s and 30s. The mechanism is the same connective-tissue weakness that produces hernias and prolapse, now applied to the colonic wall itself.
Symptoms range from intermittent left-lower-quadrant pain to frank diverticulitis (fever, tenderness, elevated white count). Imaging workup (CT) is identical to non-EDS diverticulitis, but the threshold for considering the diagnosis in a young patient should be lower when EDS is known.
The Dysautonomic Gut — POTS Effects
The gut is an autonomic organ. When the autonomic nervous system misbehaves — as it does in the majority of hEDS patients with POTS — gut motility suffers directly. Chelimsky and colleagues (2014) documented the gut-dysautonomia link in detail.
The POTS-driven gut picture includes:
- Gastric stasis worsened by upright posture — blood pools in the lower body, splanchnic perfusion drops, and gastric squeeze weakens.
- Slow colonic transit and constipation — the migrating motor complex is sympathetically driven; sympathetic dysregulation slows the housekeeping wave.
- Postprandial hypotension — eating shunts blood to the gut, and an EDS-POTS patient can feel faint, foggy, or profoundly fatigued for an hour after meals.
- Paradoxical diarrhea when sympathetic surges trigger urgent evacuation.
Volume loading (salt, fluids, compression garments) and POTS-directed therapy often improve gut symptoms as a side effect — which is why the EDS triad needs to be managed as a single system, not three separate problems.
Mast Cell GI Symptoms
Mast cells line the gut from the mouth to the anus. When they degranulate inappropriately — the hallmark of mast cell activation syndrome — the GI tract is often the first place symptoms appear.
Typical MCAS GI picture:
- Abdominal cramping within 15–60 minutes of eating certain foods (high-histamine foods are common triggers).
- Sudden-onset diarrhea, sometimes with flushing and heart racing.
- Bloating, nausea, and early satiety that wax and wane without obvious pattern.
- Food sensitivities that shift over time — a food tolerated yesterday triggers a reaction today.
Treatment layers H1 blockers (cetirizine, loratadine, fexofenadine) with H2 blockers (famotidine) and adds oral cromolyn sodium 100–200 mg 15–30 minutes before meals for direct stabilization of gut mast cells. A low-histamine diet and a low-FODMAP diet often overlap and are worth a structured trial under a dietitian’s guidance.
Eosinophilic Esophagitis and Gastritis
Eosinophilic esophagitis (EoE) and eosinophilic gastritis are increasingly recognized in EDS cohorts, particularly in patients who also have atopic disease and MCAS features. Symptoms include food impaction, difficulty swallowing solids, chest pain with eating, and persistent reflux unresponsive to standard PPI therapy. Diagnosis requires endoscopy with multiple biopsies; treatment is usually topical swallowed steroids (budesonide or fluticasone), elimination diets, or biologics (dupilumab) in refractory cases.
Median Arcuate Ligament Syndrome (MALS)
MALS is compression of the celiac artery by the median arcuate ligament of the diaphragm. In people with normal connective tissue, the ligament sits clear of the artery. In EDS, the shifted tissue geometry — combined with weight loss from gastroparesis — allows the ligament to pinch the vessel during expiration.
The classic MALS picture:
- Epigastric pain triggered by eating, often starting 15–30 minutes into a meal.
- Post-prandial nausea, weight loss, fear of eating.
- An epigastric bruit heard on exam, louder during expiration.
Screening is by mesenteric duplex ultrasound with inspiratory/expiratory maneuvers — a noninvasive test that measures celiac-artery flow velocity. Confirmation uses CT or MR angiography. Treatment of symptomatic, imaging-confirmed MALS is laparoscopic release of the median arcuate ligament, sometimes combined with celiac ganglion neurolysis. Outcomes in EDS cohorts are more variable than in the general population, and patient selection matters — MALS anatomy is common, but symptomatic MALS is rarer.
Superior Mesenteric Artery Syndrome
SMA syndrome is compression of the third part of the duodenum between the aorta behind it and the superior mesenteric artery in front of it. Normally, a fat pad keeps the two vessels apart. Weight loss, scoliosis, or loss of abdominal muscle tone (all common in EDS) narrows the angle and pinches the duodenum.
Symptoms: post-prandial epigastric pain, vomiting of partially digested food, bloating that improves lying on the left side or in a knee-chest position, and further weight loss that worsens the anatomy in a vicious cycle. Diagnosis is by CT with oral contrast showing the narrowed aortomesenteric angle. Treatment begins with nutritional rehabilitation (sometimes via NJ tube to restore the fat pad); surgical bypass (duodenojejunostomy) is reserved for refractory cases.
Dysmotility Workup
When symptoms don’t fit neatly into one category, a full motility panel sorts out what is happening where:
- Gastric emptying scintigraphy — 4-hour protocol, as described above.
- Whole-gut transit scintigraphy — measures transit through stomach, small bowel, and colon in a single study.
- Wireless motility capsule (SmartPill) — a swallowable sensor that records pH, pressure, and temperature as it traverses the GI tract, giving segmental transit times without radiation.
- Anorectal manometry and balloon expulsion test — for patients with constipation or incomplete evacuation to distinguish slow transit from pelvic floor dysfunction.
- Defecography — MRI or fluoroscopic imaging of defecation to identify rectocele, intussusception, or pelvic descent.
- Breath testing — lactulose or glucose for SIBO; lactose, fructose for sugar malabsorption.
Insurance often balks at these studies. Document each symptom and failed empirical therapy in your chart before requesting them — a motility specialist’s referral helps considerably.
Nutrition Strategies
Eating is a project in moderate-to-severe EDS GI disease. The hierarchy of intervention:
- Small frequent meals — five or six small plates, never large ones.
- Liquid nutrition as needed — commercial formulas (Boost, Ensure, Orgain, Kate Farms) for calorie insurance when solid food is failing. Kate Farms is often better tolerated because it is plant-based and free of common allergens.
- Elemental diet during flares — pre-digested amino-acid formulas (Vivonex, Physicians’ Elemental Diet, homemade versions) that require no digestion and starve SIBO bacteria. Typically run 2–3 weeks. See the Elemental Diet deep dive for protocols and cost strategies.
- NG or NJ tube feeding — for short- to medium-term failure-to-thrive situations.
- PEG (gastric) or PEG-J (jejunal) tubes — reserved for severe chronic cases where oral nutrition cannot maintain weight and hydration. Used by a minority of patients with severe gastroparesis or chronic intestinal pseudo-obstruction.
Overlap diets matter. A patient with hEDS + MCAS + SIBO may benefit from a combined low-histamine + low-FODMAP approach — but only for limited periods, and with a dietitian’s help, because combined restriction diets are nutritionally risky.
vEDS and Spontaneous Bowel Rupture
Vascular EDS (vEDS) is a separate and more dangerous subtype. The hallmark is fragility of arteries and hollow organs. Spontaneous sigmoid colon rupture is a recognized catastrophic event in vEDS, sometimes with little or no warning beyond abdominal pain.
Any vEDS patient with sudden severe abdominal pain needs urgent imaging (CT) and surgical evaluation. Elective colonoscopy in vEDS is approached with great caution; many vEDS clinicians avoid it unless absolutely necessary because of perforation risk.
Details of vEDS management live on the Classical and Vascular EDS page.
Surgery & Anesthesia Considerations
Any surgery in an EDS patient is higher-stakes than in a non-EDS patient. GI surgery is no exception.
- Tissue friability. Sutures tear, tissue pulls apart, and anastomoses leak more often than in the general population. Surgeons familiar with EDS use smaller, more frequent sutures, longer healing times, and mesh where appropriate.
- Longer hospital stays. Recovery is slower; plan for it.
- Elective-surgery thresholds should be higher. Don’t operate for marginal indications. Cosmetic or convenience procedures should be reconsidered carefully.
- Bariatric surgery in particular deserves extreme caution — anastomotic leaks, staple-line dehiscence, and internal hernias are all more common.
Anesthesia considerations specific to EDS:
- Local anesthetic (lidocaine) is often reported to be less effective or shorter-acting in EDS patients. This is patient-reported and partly supported by case literature. Dentists and minor-procedure providers should be warned in advance.
- Epidural and spinal anesthesia may require higher volumes or longer durations to achieve adequate block.
- Cervical spine instability in some hEDS patients makes intubation riskier; warn the anesthesia team in advance.
- Autonomic lability (POTS-type) responds poorly to anesthetic-induced vasodilation; expect larger blood-pressure swings.
Building Your Care Team
No single clinician manages all of this. The realistic roster:
- GI motility specialist — a gastroenterologist with specific training in dysmotility, SIBO, and functional gut disorders. Motility-focused clinics exist at most academic medical centers.
- Registered dietitian familiar with low-FODMAP, low-histamine, gastroparesis diets, and elemental feeding. Essential for navigating restrictive overlap diets without nutritional deficiency.
- Primary care physician willing to coordinate — the one person who holds the full picture and makes sure nothing falls through the cracks.
- Pain psychology or GI-focused behavioral health — not because the pain is psychological, but because chronic GI disease is exhausting and evidence-based cognitive strategies reduce symptom severity.
- Pelvic-floor physical therapist for prolapse, incomplete evacuation, or pelvic descent.
- EDS-aware rheumatologist or geneticist to anchor the systemic picture.
You are the general contractor on this project. Keep a single document with every test result, every medication trial, every flare, and every referral — and bring it to every appointment. The gastroenterologist who sees you for 20 minutes cannot reconstruct a decade of dysfunction; your notes can.
Key Research Papers
- Fikree A, Chelimsky G, Collins H, Kovacic K, Aziz Q. Gastrointestinal involvement in the Ehlers-Danlos syndromes. Neurogastroenterol Motil. 2015.
- Nelson AD, et al. Ehlers-Danlos syndrome and gastrointestinal manifestations: a 20-year experience. J Pediatr Gastroenterol Nutr. 2015.
- Zeitoun JD, et al. Functional digestive symptoms and quality of life in patients with Ehlers-Danlos syndromes: results of a national cohort study on 134 patients. PLoS One. 2013.
- Chelimsky G, et al. Gastrointestinal dysfunction in autonomic disorders. Neurogastroenterol Motil. 2014.
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on EDS-related gastrointestinal disease:
- Ehlers-Danlos and gastrointestinal involvement
- Hypermobile EDS and gastroparesis
- Ehlers-Danlos and small intestinal bacterial overgrowth
- Ehlers-Danlos and irritable bowel syndrome
- Ehlers-Danlos, hiatal hernia, and GERD
- Median arcuate ligament syndrome and EDS
- Superior mesenteric artery syndrome
- Vascular EDS and bowel rupture
- Mast cell activation and gastrointestinal disease
- POTS and gastrointestinal motility
Connections
- Ehlers-Danlos Syndrome
- Hypermobile EDS and 2017 Diagnostic Criteria
- Classical and Vascular EDS
- Craniocervical Instability and AAI
- POTS, MCAS and the EDS Triad
- Pain Management in hEDS
- Pediatric EDS and Transition of Care
- Physical Therapy and Joint Protection
- Pregnancy and EDS
- SIBO
- Rifaximin Protocols
- Elemental Diet for SIBO
- Irritable Bowel Syndrome
- IBS Rome IV Subtypes
- Gastroesophageal Reflux Disease
- POTS