Pregnancy and EDS
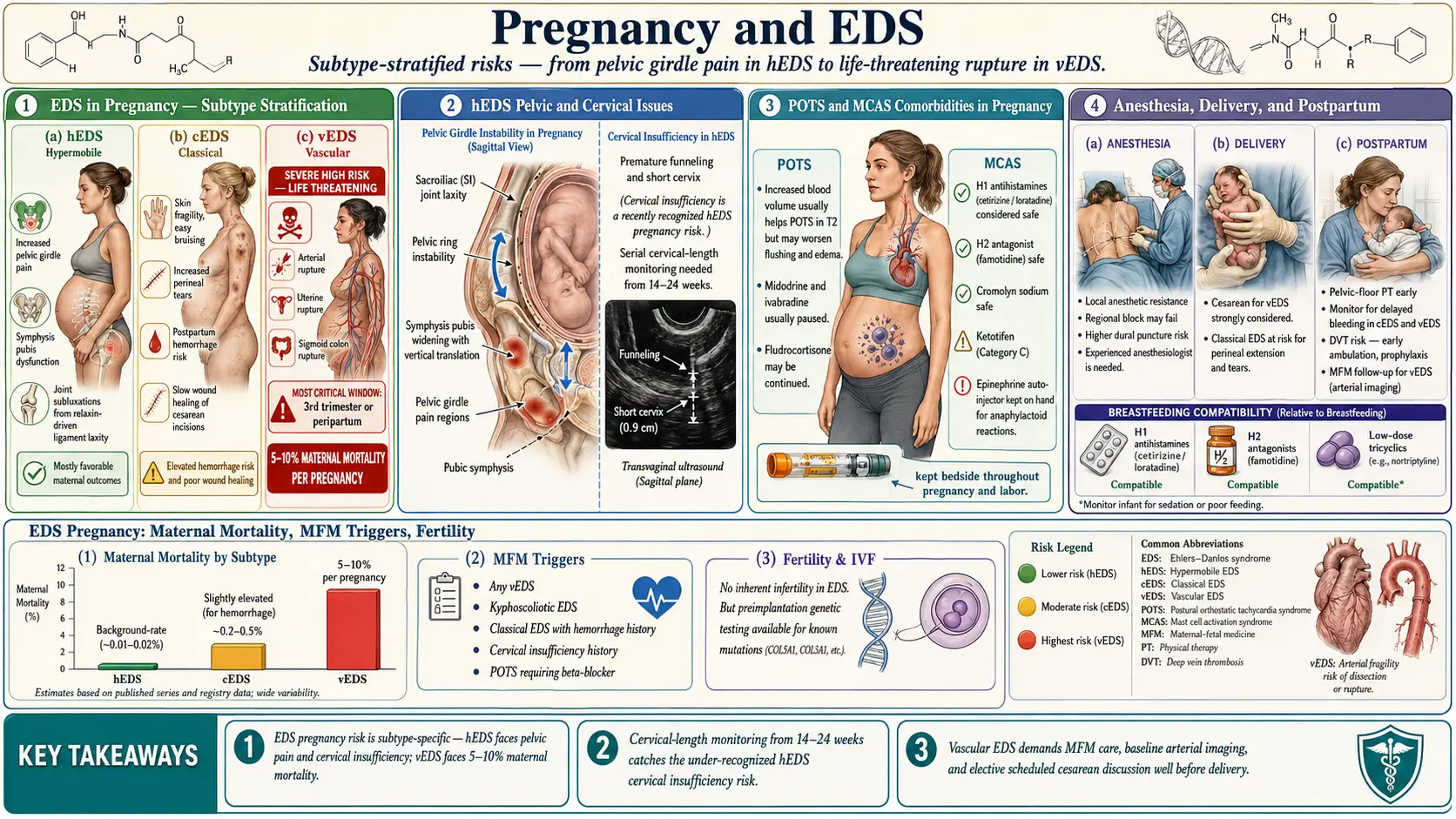
Table of Contents
- Overview — What Pregnancy Looks Like with EDS
- Hypermobile EDS (hEDS) in Pregnancy
- Symphysis Pubis Dysfunction and Pelvic Girdle Pain
- Cervical Insufficiency — The Underappreciated Risk
- Classical EDS (cEDS) in Pregnancy
- Vascular EDS (vEDS) — The High-Stakes Subtype
- POTS in Pregnancy
- MCAS Medications in Pregnancy
- Anesthesia Considerations
- Labor, Delivery & Cesarean Risks
- Postpartum Recovery — Pelvic Floor, Mood, Flares
- Breastfeeding with EDS
- Fertility, IVF & Family Planning
- Who Needs a High-Risk (MFM) Obstetrician
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
Overview — What Pregnancy Looks Like with EDS
Pregnancy with Ehlers-Danlos Syndrome is usually possible, frequently uncomfortable, and — in one specific subtype — genuinely dangerous. Most women with hypermobile EDS (hEDS) carry successful pregnancies, deliver healthy babies, and recover, though the process typically amplifies every connective-tissue complaint they had before conceiving. Women with classical EDS (cEDS) face similar issues plus a meaningful risk of premature rupture of membranes. Women with vascular EDS (vEDS) face a historically reported 12–15% maternal mortality per pregnancy, and every pregnancy decision must be individualized with a specialized team.
The dominant theme across all subtypes is the effect of relaxin — the hormone that softens connective tissue to allow the pelvis to open during delivery. In someone whose collagen is already loose, relaxin is a double dose of instability. Joints that were already subluxating pre-pregnancy now subluxate more often. Ligaments that were already stretchy now stretch to the point of injury. Skin that was already fragile now splits into striae at a rate that shocks first-time mothers. None of this is in your head, and none of it means you did something wrong.
The good news: knowledge matters. Women who enter pregnancy with a diagnosis, a specialist team, and a written plan do measurably better than those who are figuring it out as they go. This article walks through what to expect, what to screen for, and what to ask for.
Hypermobile EDS (hEDS) in Pregnancy
The majority of EDS pregnancies are hEDS pregnancies, because hEDS is by far the most common subtype. The overall obstetric outcome is generally good, but specific complications are common enough that they should be anticipated, not improvised around.
Joint instability. Relaxin levels rise early in the first trimester, peak around week 14, and remain elevated until delivery. On top of already lax ligaments, the result is predictable: more frequent subluxations of the hips, knees, shoulders, and SI joints; increased clumsiness and falls; and new instability in joints that had previously been stable. Bracing, careful transfer mechanics, and pregnancy-specific physical therapy help. Avoid deep squats, asymmetric weight-bearing, and carrying a toddler on one hip.
Striae and skin stretching. The combination of naturally elastic skin and rapid abdominal expansion produces striae gravidarum earlier, more extensively, and in more unusual distributions (flanks, breasts, thighs) than in the general obstetric population. Topicals do not prevent them. This is cosmetic, not dangerous.
Carpal tunnel and other compression neuropathies. Pregnancy-associated fluid shifts cause carpal tunnel in roughly a quarter of all pregnancies; the rate in hEDS is higher because the flexor retinaculum is already lax and peripheral nerves are more prone to mechanical injury. Night splints help. Most cases resolve by six to twelve weeks postpartum.
Rapid or precipitous labor. hEDS labor tends to be faster than average. Some women deliver within two to three hours of the first contraction, sometimes before reaching the hospital. A plan for early arrival and local paramedic awareness of the possibility is reasonable for anyone with a prior precipitous labor or a long commute to the hospital.
Postpartum hemorrhage. Connective-tissue fragility of the uterus and vaginal tissues increases the risk of heavier-than-average postpartum bleeding. Active management of the third stage (oxytocin, controlled cord traction) is standard and should not be skipped.
Symphysis Pubis Dysfunction and Pelvic Girdle Pain
Symphysis pubis dysfunction (SPD) — sometimes called pregnancy-related pelvic girdle pain — occurs in perhaps one in five unselected pregnancies. In hEDS it is more common, more severe, and lasts longer postpartum. The symphysis pubis, the SI joints, and the surrounding ligaments soften under relaxin; in EDS they soften further, and the resulting separation can be painful enough to make walking, stair-climbing, rolling over in bed, and putting on pants genuinely difficult.
Practical management:
- Pelvic support belt worn daily from the second trimester onward. Multiple brands exist; fit matters more than brand.
- Keep knees together for all transitional movements — getting in and out of the car, rolling over, getting up from a chair. Asymmetric leg movement widens the symphysis painfully.
- Crutches or a walker are not overkill in severe cases. They preserve mobility and reduce SI strain.
- Pelvic-floor-informed physical therapy with a therapist experienced in hypermobility. Generic "prenatal yoga" can worsen things by stretching already lax structures.
- Side-lying with a pillow between the knees is the least painful sleep position.
SPD often lingers six to twelve months postpartum in hEDS, and occasionally longer. It should not be dismissed as "just pregnancy pain."
Cervical Insufficiency — The Underappreciated Risk
Cervical insufficiency — the cervix opening silently and prematurely — is under-recognized in EDS. The cervix is a connective-tissue structure. In women with collagen disorders, it is mechanically weaker than in the general population, and preterm dilation without contractions is a documented risk.
A reasonable screening protocol for any EDS pregnancy:
- Transvaginal cervical length measurement every two to four weeks from 16 to 24 weeks. This is the window in which cervical shortening typically appears.
- Cerclage (a surgical stitch to reinforce the cervix) if the cervix shortens below 25 mm or if there is a history of preterm delivery or prior second-trimester loss. Cerclage in EDS is technically more challenging because the tissue holds sutures less reliably; some surgeons place reinforcing sutures.
- Vaginal progesterone may be added for short-cervix findings.
Ask for this monitoring explicitly. Standard-risk pregnancies do not get routine cervical length surveillance, and many obstetricians unfamiliar with EDS will not add it unless asked.
Classical EDS (cEDS) in Pregnancy
cEDS — caused by mutations in COL5A1 or COL5A2 — shares the hEDS concerns listed above and adds its own. The central issue is amniotic sac fragility. Some case series report premature rupture of membranes (PROM) in up to 50% of cEDS pregnancies, sometimes weeks before the due date. This can precipitate preterm delivery.
Additional cEDS concerns:
- Skin wound healing is slower. If a cesarean is required, sutures are typically left in longer (often double the standard duration), and some surgeons use retention sutures or a layered closure to compensate for tissue that holds stitches poorly.
- Vaginal tears and episiotomy wounds heal slowly and dehisce more often. Some obstetricians favor elective episiotomy in cEDS to make a clean controllable incision rather than risk an irregular tear; others argue the opposite. Discuss ahead of time.
- Uterine prolapse risk after parity is higher than in the general population, particularly after multiple deliveries.
- Neonatal screening — the baby may have inherited cEDS. Careful handling at delivery (to avoid clavicle fracture, cephalohematoma) is reasonable, and pediatric follow-up for hypermobility features should be arranged.
Vascular EDS (vEDS) — The High-Stakes Subtype
vEDS is different. It is caused by mutations in COL3A1, the gene for type III collagen, which is the scaffold of arteries, the uterus, and hollow organs. Pregnancy in vEDS is a genuine, life-threatening undertaking, and the literature has historically reported a maternal mortality of 12–15% per pregnancy, driven by uterine rupture, arterial dissection, and aortic rupture during labor and the early postpartum period.
No part of this article is a reason to panic if you already have vEDS and are pregnant. Modern specialized care — early diagnosis, preconception vascular imaging, celiprolol, planned delivery at a tertiary center with cardiothoracic surgery on site, and elective cesarean timing — has improved outcomes significantly in reported cohorts. But the decision to pursue pregnancy in vEDS is a decision that deserves a detailed individualized counseling session, ideally with a geneticist, a maternal-fetal medicine specialist, and a vascular surgeon at a center that sees vEDS regularly.
Key points that come up repeatedly in vEDS pregnancy counseling:
- Preconception whole-body MRA (magnetic resonance angiography) to map aneurysms, dissections, and baseline arterial anatomy.
- Continue celiprolol if tolerated and if your prescriber judges the benefit outweighs the fetal risk. Celiprolol is the one medication with randomized evidence (BBEST trial) for reducing arterial events in vEDS.
- Deliver at a center with 24/7 cardiothoracic surgery capable of emergency aortic repair. This is not negotiable.
- Some experts recommend elective cesarean at 34–36 weeks under controlled conditions (Pepin 2014, Murray 2014 approaches) to avoid the hemodynamic stress and uterine contractility of full-term labor. Others individualize based on imaging and prior history. There is no single accepted protocol — the point is that the decision is made deliberately, not in an emergency.
- The early postpartum period (the first two weeks) carries ongoing risk of vascular events and requires close monitoring.
- Family planning options for women with known COL3A1 mutations who want biological children but want to avoid or reduce pregnancy risk: preimplantation genetic diagnosis (PGD) with IVF to select unaffected embryos, gestational surrogacy, donor egg with a non-vEDS surrogate, or adoption. All four are medically reasonable. This is a values discussion, not just a medical one.
POTS in Pregnancy
Postural orthostatic tachycardia syndrome is common in hEDS (see POTS), and its behavior in pregnancy is counterintuitive.
- First trimester: usually worse. Blood volume has not yet expanded but vasodilation has, so standing tachycardia and presyncope intensify. Morning sickness and reduced fluid/salt intake make it worse.
- Second trimester: often better. Plasma volume has expanded by 30–50%, which happens to be the exact intervention POTS patients chase with IV fluids. Many women feel the best they have felt in years during the second trimester.
- Third trimester: mixed. Volume is still high, but mechanical compression of the inferior vena cava by the uterus can provoke supine symptoms and syncope.
- Postpartum: often rebounds harder than pre-pregnancy. Volume drops back over weeks, diuresis is rapid, and sleep deprivation is severe. Plan for a rough 6–12 weeks postpartum and consider resuming salt, fluids, and compression immediately after delivery.
Medication during pregnancy is individualized. Beta-blockers (metoprolol, propranolol) are widely used in pregnancy with a long safety record, though formally category C. Midodrine is category C with less data. Fludrocortisone is used in some pregnancies when volume expansion is inadequate. Ivabradine data in pregnancy are too limited to recommend routinely. Non-pharmacologic measures — compression stockings (preferably abdominal compression too), 3–5 grams/day of sodium, 2.5–3 liters of fluid, and left-lateral sleeping — should be maximized first.
MCAS Medications in Pregnancy
Mast cell activation syndrome (see MCAS) commonly travels with EDS. Pregnancy itself can either quiet or inflame mast cells, and it is not predictable which way a given woman will go. Whatever baseline regimen works should generally be continued, with a few medication-specific notes:
- H1 antihistamines, second generation: cetirizine, loratadine, and levocetirizine have reassuring pregnancy safety data and are the usual workhorses.
- H1 first generation: diphenhydramine and hydroxyzine are used in pregnancy but more sedating; hydroxyzine is typically avoided in the first trimester.
- H2 antihistamines: famotidine is the default; ranitidine was withdrawn in 2020 for NDMA contamination and is no longer available.
- Cromolyn sodium (oral): poorly absorbed, broadly considered safe in pregnancy, useful for GI mast cell symptoms.
- Ketotifen: not available in the United States, though used abroad.
- Montelukast: used when needed; data are reassuring overall.
- Aspirin, low-dose: used selectively for prostaglandin-mediated symptoms and also, independently, for preeclampsia prevention in high-risk patients.
General principle: do not introduce new mast-cell triggers in pregnancy. This is not the time to try a novel food, supplement, or medication unless clearly necessary. And do not abruptly stop a working regimen — a severe mast-cell reaction mid-pregnancy is worse for the fetus than continuing well-tolerated antihistamines.
Anesthesia Considerations
Anesthesia in EDS deserves its own conversation before labor begins. The issues are real, well-documented, and routinely under-anticipated by general obstetric anesthesia teams.
- Resistance to local anesthetics. A well-described phenomenon in EDS — injected lidocaine, bupivacaine, and similar agents wear off faster than expected or fail to fully anesthetize. Bring the EDS Society anesthesia card to your delivery hospital and ask the anesthesiologist to review it in advance. Plan for double-checking block effectiveness before the procedure starts.
- Difficult epidural placement. Atypical spinal anatomy, ligamentum flavum laxity, and the loss of the classic "loss of resistance" endpoint make epidural placement technically harder. A senior anesthesiologist, not a trainee, is a reasonable request.
- Extended duration of block required. Plan catheter placement early if labor is progressing slowly; do not wait until transition.
- Dural puncture and post-dural-puncture headache. Connective-tissue fragility increases the chance of inadvertent dural puncture and may prolong the resulting CSF leak headache. Epidural blood patches work but may need to be repeated.
- Cervical spine caution. If general anesthesia is required, cervical instability may complicate intubation. Flag this to the anesthesia team.
Write the anesthesia plan into your birth plan, share it with your obstetrician in advance, and request an antepartum anesthesia consultation — typically at 32–36 weeks — so the team meets you before you are in active labor.
Labor, Delivery & Cesarean Risks
Vaginal delivery is possible and often preferred in hEDS. Cesarean is sometimes chosen electively (maternal request, prior severe SPD, fetal or positional indications, vEDS) and sometimes required for obstetric reasons. Both routes carry EDS-specific risks.
Vaginal delivery concerns: precipitous labor, perineal tears (possibly extending further than expected through fragile tissue), episiotomy that heals slowly, and postpartum hemorrhage from uterine or vaginal tissue fragility.
Cesarean concerns: intraoperative tissue friability (fascia, uterus, and skin all hold sutures less reliably), slow wound healing, higher rates of wound dehiscence, higher rates of incisional hernia, and in vEDS the risk of arterial injury during surgery. Cesarean in EDS typically involves longer suture duration, slower activity progression postpartum, and a longer hospital stay for monitoring.
Either way, expect a longer recovery than an average-risk mother. Three to six weeks of explicit lifting restrictions is reasonable, and "getting back to normal" at six weeks is an unrealistic expectation that many women blame themselves for. Block out three months.
Postpartum Recovery — Pelvic Floor, Mood, Flares
Postpartum is frequently the hardest phase of an EDS pregnancy. Three problems cluster here:
Joint flare. Relaxin lingers for several months, particularly while breastfeeding. Joints that were hanging on during pregnancy often destabilize in the first six to twelve weeks postpartum, with a wave of subluxations, pain, and functional loss. Plan for it. Have braces, PT appointments, and help with the baby lined up before delivery.
Pelvic floor dysfunction. Urinary incontinence, fecal incontinence, and early pelvic organ prolapse are all more common after delivery in EDS. An early (6–8 week) pelvic-floor PT evaluation — not a generic one, but with someone who works with hypermobility — is one of the highest-yield interventions for long-term quality of life. Long-term pelvic organ prolapse risk after parity is elevated in EDS, with symptoms sometimes appearing years after delivery.
Postpartum mood. Postpartum depression and anxiety rates are higher in EDS. The drivers are obvious in retrospect: severe sleep deprivation in someone who already slept poorly, hormonal collapse, joint flare, pain, and a body that does not feel like it is recovering. This is a medical problem, not a character flaw. SSRIs, therapy, sleep support, and a low threshold for formal postpartum mental health care should all be on the table.
Breastfeeding with EDS
Breastfeeding is usually possible in EDS, but positioning is the deal-breaker. Holding a newborn in a cradle position for 30–45 minutes, eight to twelve times a day, routinely subluxates shoulders, strains wrists, and aggravates neck pain in hEDS. Solutions:
- Side-lying nursing for the first weeks — far less load on shoulders and wrists.
- Nursing pillows (Boppy, My Brest Friend, or similar) to take the weight off the arms.
- Football hold after cesarean to keep pressure off the incision.
- Pumping as a full or partial substitute if latch-related strain becomes unsustainable.
- Medication compatibility: use the NIH LactMed database to look up any EDS-related medication (beta-blockers, antihistamines, NSAIDs, SSRIs). Most commonly used agents have reassuring data.
Do not let an aggressive lactation consultant push you into positions that are injuring you. Fed is not just a slogan — for a recovering EDS mother, sustainable is the whole game.
Fertility, IVF & Family Planning
Baseline fertility in hEDS is generally normal. cEDS and vEDS fertility rates do not appear substantially reduced either, though the decision to attempt pregnancy in vEDS is dominated by risk, not fertility. EDS overlaps heavily with endometriosis, which can reduce fertility and cause significant cyclical pain; women in this overlap population sometimes need assisted reproduction (see Infertility).
If IVF is pursued:
- Oocyte retrieval requires extra care — the vaginal wall and ovarian tissue are more fragile, and bleeding from retrieval puncture sites is more common. Communicate the diagnosis clearly to the reproductive endocrinologist.
- Ovarian hyperstimulation syndrome (OHSS) is treated the same way as in non-EDS patients, but fluid shifts can destabilize coexisting POTS.
- Preimplantation genetic diagnosis (PGD) is available for families with a known COL3A1 (vEDS) mutation, and increasingly for families with known COL5A1/COL5A2 (cEDS) mutations. hEDS does not yet have a reliable genetic marker, so PGD is not available for hEDS.
Who Needs a High-Risk (MFM) Obstetrician
Not every EDS pregnancy needs maternal-fetal medicine, but many should have at least a consultation. A practical cutoff:
- Vascular EDS — always. Full MFM co-management, delivery at a tertiary center with cardiothoracic surgery, individualized delivery plan.
- Classical EDS — usually. PROM risk, cervical-length surveillance, and delivery planning justify MFM involvement.
- Hypermobile EDS — selectively. Refer to MFM if any of the following apply: prior preterm delivery, prior second-trimester loss, severe POTS or MCAS, cervical shortening on surveillance, prior precipitous labor with complications, prior postpartum hemorrhage, or severe joint instability requiring specific delivery planning.
- Any subtype + significant comorbidity — thyroid disease (see Pregnancy and Hashimoto's), cardiac disease, prior preeclampsia (see Preeclampsia) — is worth at least one MFM visit.
The goal is not to medicalize a pregnancy that does not need it. The goal is to make sure the team knows what EDS is before a complication arrives, not after.
Key Research Papers
- Pepin MG, Murray ML, Byers PH. Survival is affected by mutation type and molecular mechanism in vascular Ehlers-Danlos syndrome (EDS type IV). Genet Med. 2014.
- Murray ML, Pepin M, Peterson S, Byers PH. Pregnancy-related deaths and complications in women with vascular Ehlers-Danlos syndrome. Obstet Gynecol. 2014.
- Castori M, et al. Pregnancy and delivery in Ehlers-Danlos syndrome hypermobility type. Ultrasound Obstet Gynecol. 2012.
- Ehlers-Danlos pregnancy outcomes — PubMed current literature.
Research Papers
The following PubMed topic searches return current peer-reviewed work on EDS in pregnancy:
- Hypermobile EDS and pregnancy outcomes
- Vascular EDS pregnancy and maternal mortality
- Classical EDS and premature rupture of membranes
- Cervical insufficiency in Ehlers-Danlos syndrome
- Symphysis pubis dysfunction and joint hypermobility
- POTS in pregnancy
- Mast cell activation and antihistamines in pregnancy
- Anesthesia and local anesthetic resistance in EDS
- Celiprolol and the BBEST trial in vascular EDS
- Preimplantation genetic diagnosis for COL3A1
Connections
- Ehlers-Danlos Syndrome
- Hypermobile EDS and 2017 Diagnostic Criteria
- Classical and Vascular EDS
- Craniocervical Instability and AAI
- GI Involvement in EDS
- POTS, MCAS and the EDS Triad
- Pain Management in hEDS
- Pediatric EDS and Transition of Care
- Physical Therapy and Joint Protection
- POTS
- MCAS
- Infertility
- Preeclampsia
- Endometriosis
- Hashimoto's Thyroiditis
- Pregnancy and Hashimoto's
- Chronic Fatigue Syndrome
- Migraine