Acute Kidney Injury
Acute Kidney Injury (AKI) is a sudden episode of kidney failure or damage that occurs within hours to days, resulting in accumulation of waste products in the blood and disruption of fluid and electrolyte balance. It is a common and serious condition associated with high morbidity and mortality, particularly in hospitalized patients.
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research and Advances
- References
- Featured Videos
1. Overview
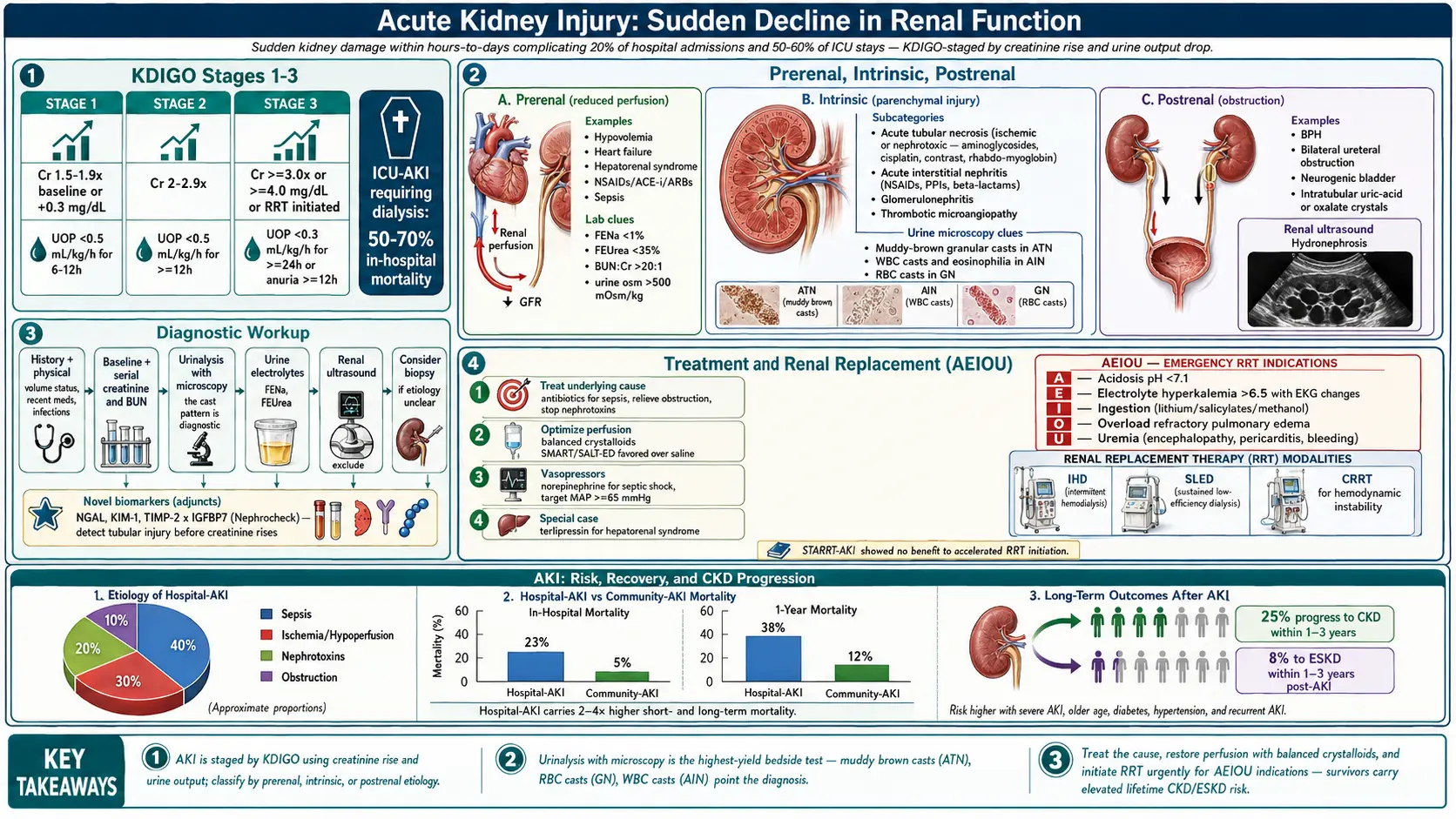
Acute Kidney Injury is defined by the Kidney Disease: Improving Global Outcomes (KDIGO) 2012 guidelines as any of the following: a rise in serum creatinine by 0.3 mg/dL or more within 48 hours; a rise in serum creatinine to 1.5 times or more the baseline within 7 days; or urine output less than 0.5 mL/kg/hour for 6 hours or more. AKI represents a spectrum of injury ranging from mild functional impairment to complete cessation of kidney function requiring renal replacement therapy (RRT).
AKI is classified into three stages based on KDIGO criteria, with Stage 1 representing the mildest and Stage 3 the most severe form. The condition may be prerenal (reduced perfusion), intrinsic renal (direct parenchymal injury), or postrenal (obstructive) in etiology. Early recognition and intervention are critical to improving outcomes.
2. Epidemiology
AKI affects approximately 13.3 million people per year worldwide, with an estimated 1.7 million deaths annually attributable to the condition. In high-income countries, AKI complicates approximately 20% of all hospital admissions and up to 50–60% of intensive care unit (ICU) admissions. The incidence in critically ill patients ranges from 30–67% depending on the definition used and population studied.
Community-acquired AKI accounts for approximately 40% of cases, while hospital-acquired AKI comprises the remaining 60%. In low- and middle-income countries, AKI is predominantly caused by infections (sepsis, malaria, diarrheal illness) and obstetric complications. The condition disproportionately affects the elderly, with incidence rising sharply after age 65 due to reduced nephron reserve and comorbidity burden.
AKI requiring dialysis carries an in-hospital mortality of 50–70% in ICU settings. Survivors face an increased risk of developing chronic kidney disease (CKD), with approximately 25% progressing to CKD and 8% to end-stage kidney disease (ESKD) within 1–3 years.
3. Pathophysiology
The pathophysiology of AKI is multifactorial and varies by etiology. The KDIGO staging system provides a framework for severity classification:
- Stage 1: Serum creatinine 1.5–1.9 times baseline OR increase of ≥0.3 mg/dL; urine output <0.5 mL/kg/h for 6–12 hours
- Stage 2: Serum creatinine 2.0–2.9 times baseline; urine output <0.5 mL/kg/h for ≥12 hours
- Stage 3: Serum creatinine ≥3.0 times baseline, or ≥4.0 mg/dL, or initiation of RRT, or GFR <35 mL/min/1.73 m² in patients <18 years; urine output <0.3 mL/kg/h for ≥24 hours or anuria for ≥12 hours
Prerenal AKI
Reduced effective arterial blood volume leads to decreased renal perfusion pressure. The kidney compensates by activating the renin-angiotensin-aldosterone system (RAAS) and increasing antidiuretic hormone (ADH) secretion, resulting in sodium and water retention. Prolonged prerenal states lead to ischemic tubular injury and intrinsic AKI.
Intrinsic Renal AKI
Acute tubular necrosis (ATN) is the most common form of intrinsic AKI, caused by ischemia or nephrotoxic injury. Ischemic ATN results from sustained hypoperfusion leading to tubular epithelial cell death, loss of brush border integrity, and intraluminal cast formation. Inflammatory mediators, including reactive oxygen species, cytokines (TNF-α, IL-1β, IL-6), and complement activation, amplify tubular injury. Tubular cell death occurs via apoptosis and necrosis, with subsequent desquamation causing tubular obstruction and back-leak of glomerular filtrate.
Glomerulonephritis, vasculitis, and acute interstitial nephritis represent other forms of intrinsic AKI with distinct immunopathological mechanisms.
Postrenal AKI
Bilateral ureteral obstruction or obstruction at the bladder or urethra increases intratubular pressure, reducing glomerular filtration rate (GFR). Prolonged obstruction leads to irreversible tubular atrophy and interstitial fibrosis.
4. Etiology and Risk Factors
Prerenal Causes
- Hypovolemia: hemorrhage, burns, vomiting, diarrhea, excessive diuresis
- Reduced cardiac output: heart failure, cardiogenic shock, pericardial tamponade
- Hepatorenal syndrome in cirrhosis
- Medications: NSAIDs, ACE inhibitors/ARBs, calcineurin inhibitors
- Sepsis-associated hemodynamic compromise
Intrinsic Renal Causes
- Acute tubular necrosis (ischemic or nephrotoxic)
- Nephrotoxins: aminoglycosides, cisplatin, contrast media, myoglobin (rhabdomyolysis), hemoglobin
- Glomerulonephritis: rapidly progressive GN, anti-GBM disease, ANCA-associated vasculitis
- Acute interstitial nephritis: drug-induced (NSAIDs, beta-lactams, PPIs), infectious, autoimmune
- Thrombotic microangiopathy: HUS, TTP, HELLP syndrome
- Renal vascular occlusion: renal artery thrombosis, cholesterol emboli
Postrenal Causes
- Benign prostatic hyperplasia, prostate or cervical carcinoma
- Bilateral ureteral obstruction (stones, retroperitoneal fibrosis, malignancy)
- Neurogenic bladder, urethral stricture
- Intratubular obstruction: uric acid crystals (tumor lysis), oxalate, acyclovir, methotrexate
Risk Factors
- Advanced age (>65 years), pre-existing CKD, diabetes mellitus, hypertension
- Heart failure, cirrhosis, sepsis, major surgery (cardiac, vascular, orthopedic)
- Nephrotoxic medication exposure, radiocontrast use, volume depletion
- Male sex, Black ethnicity, obesity
5. Clinical Presentation
The clinical presentation of AKI is highly variable and depends on the underlying etiology, severity, and rate of progression. Many patients with mild AKI are asymptomatic, with the condition detected only through laboratory abnormalities.
Symptoms
- Oliguria or anuria: Urine output <400 mL/day (oliguria) or <100 mL/day (anuria) in severe cases; non-oliguric AKI occurs in 30–50% of patients
- Fluid overload: peripheral edema, pulmonary edema, hypertension, weight gain
- Uremic symptoms: nausea, vomiting, anorexia, lethargy, confusion, asterixis, pericarditis (uremic frost in late stages)
- Electrolyte disturbances: muscle weakness or cramps (hyponatremia, hyperkalemia), tetany (hypocalcemia)
- Flank pain: suggestive of obstruction, renal vein thrombosis, or papillary necrosis
Signs by Etiology
- Prerenal: orthostatic hypotension, tachycardia, dry mucous membranes, reduced skin turgor, elevated BUN:creatinine ratio >20:1
- Intrinsic (ATN): muddy brown granular casts on urinalysis, isosthenuria
- Glomerulonephritis: hematuria (dysmorphic red cells, RBC casts), proteinuria, hypertension
- Acute interstitial nephritis: fever, rash, eosinophilia, WBC casts (classic triad present in <10% of cases)
- Postrenal: suprapubic fullness, costovertebral angle tenderness, distended bladder
6. Diagnosis
Laboratory Evaluation
- Serum creatinine and BUN: Serial measurements to confirm AKI and assess trajectory; baseline creatinine estimation using CKD-EPI equation if unavailable
- Estimated GFR (eGFR): Calculated using CKD-EPI 2021 creatinine equation; unreliable in acute setting due to non-steady state
- Urinalysis with microscopy: Essential to differentiate AKI etiology — muddy brown granular casts (ATN), RBC casts (GN), WBC casts (AIN), waxy/broad casts (advanced CKD)
- Urine electrolytes: Fractional excretion of sodium (FENa) <1% suggests prerenal; >2% suggests intrinsic ATN; FEUrea <35% is more reliable in patients receiving diuretics
- Urine osmolality: >500 mOsm/kg in prerenal; isosthenuria (250–300 mOsm/kg) in ATN
- Complete metabolic panel: Potassium (hyperkalemia), bicarbonate (metabolic acidosis), phosphate, calcium, uric acid
- CBC: Evaluate for hemolytic anemia (microangiopathy), eosinophilia (AIN), thrombocytopenia (TTP/HUS)
- Novel biomarkers: NGAL (neutrophil gelatinase-associated lipocalin), KIM-1 (kidney injury molecule-1), TIMP-2 × IGFBP7 ([Nephrocheck]) — detect tubular injury earlier than creatinine
Immunological Workup (when intrinsic GN/vasculitis suspected)
- Anti-GBM antibodies, ANCA (MPO-ANCA, PR3-ANCA), ANA, anti-dsDNA, complement (C3, C4, CH50)
- Serum protein electrophoresis, free light chains (myeloma-associated AKI)
- Cryoglobulins, hepatitis B and C serology, HIV
Imaging
- Renal ultrasound: First-line imaging; excludes obstruction (hydronephrosis), assesses kidney size and echogenicity; small echogenic kidneys suggest pre-existing CKD
- CT urography (without contrast if possible) for urolithiasis, retroperitoneal pathology
- Doppler ultrasound for renal vascular assessment
Kidney Biopsy
Indicated when the etiology remains unclear after clinical evaluation, when intrinsic GN, vasculitis, or AIN is suspected, or when RRT is required without an identifiable cause. Contraindicated in coagulopathy, solitary kidney, or uncontrolled hypertension.
7. Treatment
General Principles
- Identify and treat the underlying cause (antibiotics for sepsis, relief of obstruction, cessation of nephrotoxins)
- Optimize hemodynamic status and renal perfusion
- Monitor fluid balance, electrolytes, and acid-base status closely
- Avoid or dose-adjust nephrotoxic medications; monitor aminoglycoside and vancomycin levels
Fluid Management
Judicious fluid resuscitation is critical. Isotonic crystalloids (normal saline or balanced solutions such as Lactated Ringer's or Plasma-Lyte) are preferred over colloids for volume expansion. Balanced crystalloids are associated with lower rates of AKI and hyperchloremic acidosis compared to 0.9% saline (SMART and SALT-ED trials). Albumin infusion is indicated in hepatorenal syndrome and spontaneous bacterial peritonitis. Fluid overload independently worsens outcomes; targeted decongestion with diuretics is appropriate once euvolemia is achieved.
Vasoactive Agents
- Norepinephrine is the vasopressor of choice in septic shock to maintain MAP ≥65 mmHg
- Vasopressin analogues (terlipressin) for hepatorenal syndrome type 1
- Low-dose dopamine is not recommended for AKI prevention or treatment
Electrolyte and Acid-Base Management
- Hyperkalemia: Calcium gluconate (membrane stabilization), insulin/dextrose, sodium bicarbonate, cation exchange resins (patiromer, sodium zirconium cyclosilicate), emergency dialysis for K⁺ >6.5 mEq/L with ECG changes
- Metabolic acidosis: Sodium bicarbonate for pH <7.15 or bicarbonate <15 mEq/L; RRT for severe refractory acidosis
- Hyperphosphatemia: Dietary restriction, phosphate binders (calcium carbonate, sevelamer)
- Fluid overload: Loop diuretics (furosemide) at high doses; may prevent progression but do not reduce mortality
Renal Replacement Therapy (RRT)
Indications for urgent RRT (the "AEIOU" mnemonic):
- Acidosis: refractory metabolic acidosis pH <7.1
- Electrolytes: severe hyperkalemia (>6.5 mEq/L) refractory to medical management
- Ingestion: life-threatening drug or toxin (lithium, salicylates, methanol)
- Overload: refractory pulmonary edema unresponsive to diuretics
- Uremia: uremic encephalopathy, pericarditis, bleeding diathesis
RRT modalities include intermittent hemodialysis (IHD), sustained low-efficiency dialysis (SLED), and continuous renal replacement therapy (CRRT). CRRT is preferred in hemodynamically unstable patients. The STARRT-AKI trial found no mortality benefit from accelerated versus standard RRT initiation strategy, supporting a watchful-waiting approach unless urgent indications are present.
Specific Therapies
- Contrast-induced AKI: IV hydration with isotonic saline before and after contrast; N-acetylcysteine is no longer recommended (PRESERVE trial); use iso-osmolar or low-osmolar contrast; minimize contrast volume
- Rhabdomyolysis-induced AKI: Aggressive IV fluid resuscitation (target urine output 200–300 mL/h); urinary alkalinization controversial; treat compartment syndrome
- ANCA vasculitis/anti-GBM: High-dose corticosteroids + cyclophosphamide or rituximab; plasma exchange for anti-GBM disease (PEXIVAS trial for ANCA)
- Acute interstitial nephritis: Discontinue offending drug; corticosteroids (prednisone 1 mg/kg/day) may accelerate recovery if initiated early
- Hepatorenal syndrome: Terlipressin + albumin (CONFIRM trial); liver transplantation is definitive therapy
8. Complications
- Fluid and electrolyte disorders: Hyperkalemia (life-threatening arrhythmias), hyponatremia, hyperphosphatemia, hypocalcemia, hypermagnesemia
- Acid-base disturbances: High anion gap metabolic acidosis (uremic acids), mixed disorders
- Cardiovascular: Pulmonary edema, arrhythmias, hypertension, pericarditis
- Hematological: Normocytic anemia (erythropoietin deficiency, hemodilution), platelet dysfunction, bleeding risk
- Infectious: Increased susceptibility to infection due to uremia-associated immunosuppression
- Nutritional: Catabolism, malnutrition; protein requirements increase with RRT
- Long-term: Progression to CKD (25%), ESKD (8%), increased cardiovascular mortality
- Distant organ effects: AKI-to-lung injury cross-talk (ARDS), hepatic dysfunction, myocardial depression
9. Prognosis
Prognosis in AKI is determined by the severity of AKI (KDIGO stage), underlying etiology, comorbidities, and the presence of multi-organ failure. Community-acquired AKI carries a 13% in-hospital mortality, compared to 23% for hospital-acquired AKI and 50–70% for ICU-associated AKI requiring RRT. Non-oliguric AKI generally carries a better prognosis than oliguric AKI.
Renal recovery is defined as return to within 25% of baseline creatinine. Complete recovery occurs in approximately 60% of survivors. Predictors of non-recovery include older age, pre-existing CKD, higher AKI stage, prolonged duration of AKI, and sepsis as the underlying cause. The AKI-to-CKD transition is now recognized as a major pathway to ESKD globally.
10. Prevention
- Hemodynamic optimization: Goal-directed therapy in high-risk surgical patients; early recognition and treatment of sepsis (Surviving Sepsis Campaign guidelines)
- Nephrotoxin avoidance: Avoid concurrent nephrotoxic agents; use non-nephrotoxic alternatives when possible; therapeutic drug monitoring for aminoglycosides and vancomycin
- Contrast media precautions: Pre-procedure hydration; use lowest effective contrast dose; avoid high-osmolar agents; consider alternative imaging modalities in high-risk patients
- Medication review: Hold ACE inhibitors/ARBs, diuretics, and NSAIDs in acutely ill patients at risk for AKI (sick-day rules)
- Glycemic control: Moderate glycemic control (target 140–180 mg/dL) in ICU patients
- Remote ischemic preconditioning: May reduce AKI in cardiac surgery (RIPC trials)
- Clinical decision support: Electronic AKI alerts integrated in EHR systems improve early recognition and response
11. Recent Research and Advances
- SGLT2 inhibitors: Emerging evidence supports nephroprotective effects via tubuloglomerular feedback restoration and reduced hyperfiltration; trials ongoing for AKI prevention in high-risk hospitalized patients
- Biomarker-guided care: TIMP-2 × IGFBP7 (NephroCheck) validated for predicting AKI within 12 hours; emerging panels combining NGAL, KIM-1, and IL-18 for precision diagnostics
- Machine learning: Deep learning models (e.g., DeepMind AKI model) demonstrated ability to predict AKI up to 48 hours before laboratory diagnosis using routine clinical data
- Mitochondrial-targeted therapies: Mitochondrial dysfunction is central to ischemic ATN; SS-31 peptide (elamipretide) and other mitochondrial protective agents are under investigation
- RRT optimization: STARRT-AKI established no benefit to accelerated RRT initiation; optimal membrane type and anticoagulation strategies (citrate vs. heparin in CRRT) remain active research areas
- AKI-CKD transition: Understanding of maladaptive repair mechanisms (persistent G2/M cell cycle arrest, epigenetic reprogramming, pericyte-to-myofibroblast transition) driving fibrosis is guiding novel therapeutic targets
- Sepsis-associated AKI: Persistent research focus on the distinct hemodynamic, inflammatory, and microcirculatory mechanisms driving AKI in septic shock; experimental therapies target endothelial glycocalyx protection, selective cytokine removal, and angiotensin II replacement
12. References
- KDIGO Clinical Practice Guideline for Acute Kidney Injury. Kidney International Supplements. 2012;2(1):1–138.
- Kellum JA, Romagnani P, Ashuntantang G, et al. Acute kidney injury. Nature Reviews Disease Primers. 2021;7(1):52.
- Hoste EAJ, Bagshaw SM, Bellomo R, et al. Epidemiology of acute kidney injury in critically ill patients: the multinational AKI-EPI study. Intensive Care Medicine. 2015;41(8):1411–1423.
- Semler MW, Self WH, Wanderer JP, et al. Balanced crystalloids versus saline in critically ill adults (SMART trial). New England Journal of Medicine. 2018;378(9):829–839.
- STARRT-AKI Investigators. Timing of initiation of renal replacement therapy in acute kidney injury. New England Journal of Medicine. 2020;383(3):240–251.
- Legrand M, Bagshaw SM, Bhagat V, et al. Terlipressin vs norepinephrine in patients with septic shock and acute kidney injury. JAMA. 2023 (ATHOS-3 and subsequent meta-analyses).
- Kashani K, Al-Khafaji A, Ardiles T, et al. Discovery and validation of cell cycle arrest biomarkers in human acute kidney injury. Critical Care. 2013;17(1):R25.
- Rajasekaran A, Julian BA, Rizk DV. IgA nephropathy: an interesting autoimmune kidney disease. American Journal of the Medical Sciences. 2021;361(2):176–194.
- Perazella MA, Rosner MH. Drug-induced acute kidney injury. Clinical Journal of the American Society of Nephrology. 2022;17(8):1220–1233.
- Ronco C, Bellomo R, Kellum JA. Acute kidney injury. Lancet. 2019;394(10212):1949–1964.
- Chawla LS, Eggers PW, Star RA, Kimmel PL. Acute kidney injury and chronic kidney disease as interconnected syndromes. New England Journal of Medicine. 2014;371(1):58–66.
- Investigators of the Randomized Evaluation of Normal vs. Augmented Level of Replacement Therapy (RENAL) study. Intensity of continuous renal replacement therapy in critically ill patients. New England Journal of Medicine. 2009;361(17):1627–1638.
- Gaudry S, Hajage D, Schortgen F, et al. Initiation strategies for renal replacement therapy in the intensive care unit (AKIKI). New England Journal of Medicine. 2016;375(2):122–133.
- Kidney Disease: Improving Global Outcomes (KDIGO). KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney International. 2024;105(4S):S117–S314.
- Tomasev N, Glorot X, Rae JW, et al. A clinically applicable approach to continuous prediction of future acute kidney injury. Nature. 2019;572(7767):116–119.
- Endre ZH, Walker RJ, Pickering JW, et al. Early intervention with erythropoietin does not affect the outcome of acute kidney injury. Journal of the American Society of Nephrology. 2010;21(8):1397–1408.
Research Papers
Explore current literature on acute kidney injury via PubMed topic searches. These links open live PubMed searches for the listed keywords — results update as new studies are indexed.
- Clinical trials on acute kidney injury — PubMed search
- Clinical trials on AKI biomarkers (NGAL, KIM-1) — PubMed search
- Clinical trials on contrast-induced nephropathy — PubMed search
- Clinical trials on sepsis-associated AKI — PubMed search
- Clinical trials on renal replacement therapy in AKI — PubMed search
- Clinical trials on AKI to CKD transition — PubMed search
- Clinical trials on KDIGO AKI guidelines — PubMed search
Connections
- Kidney Disease
- Glomerulonephritis
- Hypertension
- Sepsis
- Cirrhosis
- Heart Failure
- Diabetes
- Nephrotic Syndrome
- Creatinine
- Blood Urea Nitrogen
- eGFR
- Kidney Function Tests
- Urinalysis
- Methylene Blue
- Edema
- Lead
- Preeclampsia
- Disseminated Intravascular Coagulation