Low-Dose Naltrexone (LDN) for Fibromyalgia
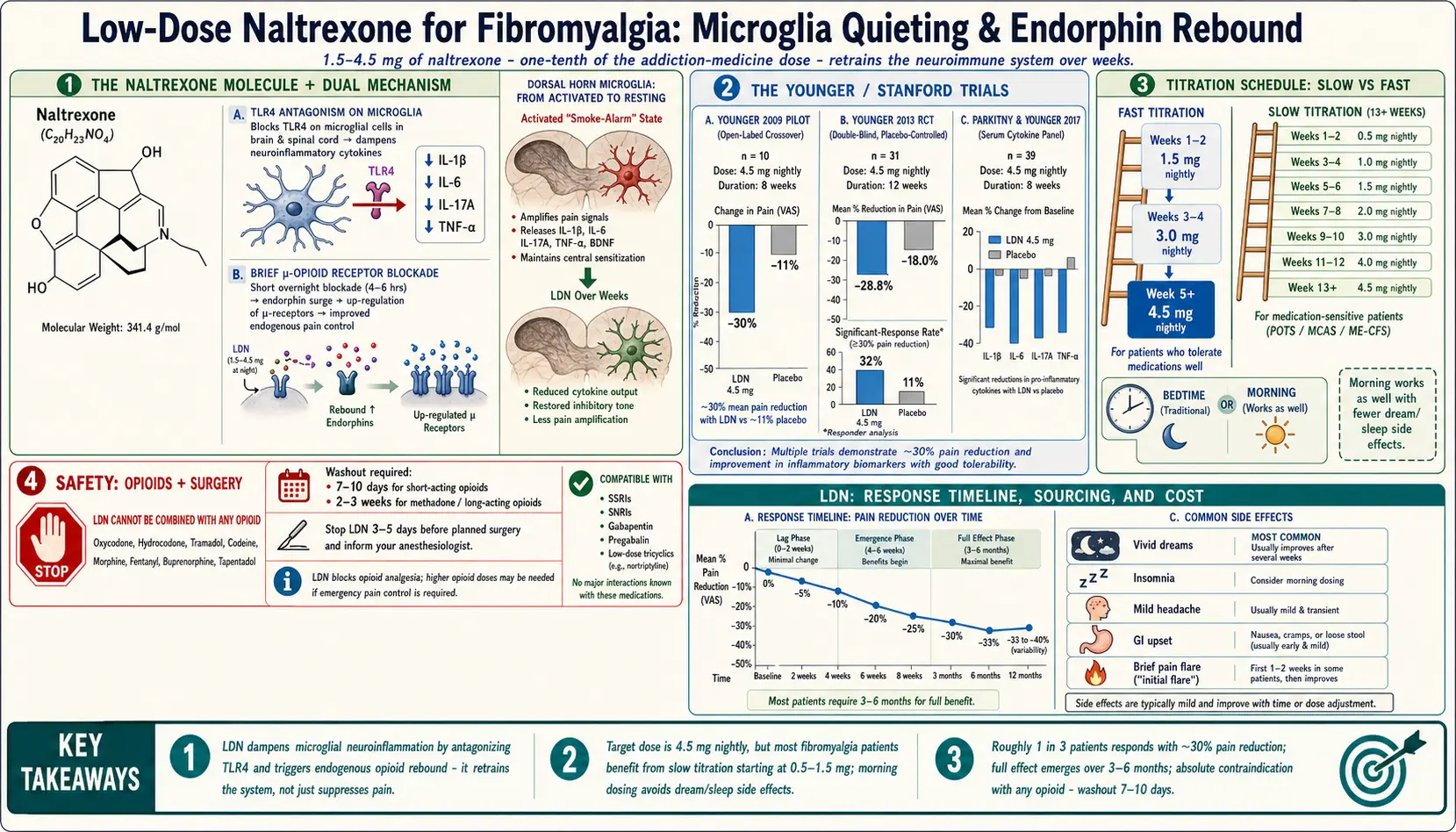
Table of Contents
- What Low-Dose Naltrexone Is
- How LDN Works in Fibromyalgia
- The Younger / Stanford Trials
- Typical Dosing — Where to Start and Where to Land
- Slow vs Fast Titration Schedules
- Compounding Pharmacies, Cost, and Insurance
- Side Effects — What to Expect
- Drug Interactions — Opioids and Surgery
- Morning vs Bedtime — The Timing Debate
- What “Working” Looks Like
- When to Give Up
- Practical Tips for Starting
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
What Low-Dose Naltrexone Is
Naltrexone is an old drug, FDA-approved in 1984 at 50 mg for alcohol and opioid use disorder. At that dose it is a powerful opioid-receptor blocker — it keeps heroin, oxycodone, and the body's own endorphins from docking onto nerve cells for 24 hours at a time.
Low-dose naltrexone (LDN) is the same molecule given at roughly one-tenth the dose — typically 1.5 to 4.5 mg, taken once a day. At this dose the drug behaves completely differently. Instead of paralyzing the opioid system, it produces a brief blockade lasting four to six hours, then washes out. During that window the body senses low endorphin signaling and ramps up its own production. When the drug is gone, endorphin levels rebound above baseline.
LDN is an off-label prescription in the United States. No drug company owns it (the patent expired decades ago), so no sponsor has paid for the expensive Phase III trials needed for a fibromyalgia label. This is the single biggest reason LDN is still treated as a “fringe” therapy by some physicians — not because the science is weak, but because the profit motive is missing.
How LDN Works in Fibromyalgia
Fibromyalgia is, at its core, a disease of amplified pain signaling in the central nervous system (see central sensitization). The volume knob on pain is turned up, and a big part of that volume is controlled by microglia — the immune cells of the brain and spinal cord.
When microglia are chronically activated, they pump out inflammatory cytokines (IL-6, TNF-alpha, IL-1β) and pro-inflammatory chemicals that sensitize the nerves around them. Imagine a smoke alarm wired to an amplifier: every tiny input becomes a siren. This is why a light touch, a cool breeze, or a sound can register as pain in fibromyalgia.
LDN appears to calm that alarm through two distinct actions:
- TLR4 antagonism on microglia. Naltrexone (in its non-opioid stereoisomer and likely the regular one too) blocks Toll-like receptor 4 (TLR4) on microglial cells. TLR4 is a major “danger signal” receptor. Blocking it tells microglia to stand down and stop pumping out inflammatory cytokines. This is thought to be the main pain-quieting mechanism.
- Transient opioid blockade → endorphin rebound. The short, partial opioid blockade briefly reduces activity at mu-opioid receptors. The brain responds by upregulating both endorphin production and opioid-receptor sensitivity. Over weeks, the body's own pain-control system becomes more responsive.
In short, LDN is not a painkiller the way ibuprofen or an opioid is. It does not suppress pain while you take it. It retrains the neuroimmune system over weeks to generate less pain in the first place.
The Younger / Stanford Trials
The modern LDN story in fibromyalgia was largely built by Jarred Younger, PhD, first at Stanford and now at the University of Alabama at Birmingham. Two small but careful studies put LDN on the map:
Younger 2009 (pilot, single-blind crossover). Ten women with fibromyalgia took LDN 4.5 mg nightly for eight weeks. Average pain dropped by about 30% compared with placebo, and fatigue and stress also improved. The paper is small but carefully done and was the first controlled hint that LDN helps fibromyalgia specifically.
Younger 2013 (randomized, double-blind, placebo-controlled crossover). Thirty-one women completed twelve weeks of LDN versus placebo. LDN reduced daily pain by roughly 28.8%, compared with 18.0% for placebo. Roughly 32% of LDN-treated participants met the threshold for a significant response, versus 11% on placebo. Mood improved. Side effects were mild.
Parkitny & Younger 2017. A follow-up study measured serum cytokines before and after LDN and found reductions in IL-1β, IL-6, IL-17A, TNF-alpha, and others. This gave the first biological fingerprint to back up the clinical results — LDN really was dampening inflammatory signaling, not just producing a placebo response.
These trials are small by pharma standards — tens of patients, not thousands. A large, definitive Phase III trial has never been done because no sponsor would profit from it. What we have instead is a growing number of open-label case series, a handful of replications, and tens of thousands of patient reports showing a consistent signal: roughly one in three fibromyalgia patients gets meaningful relief, the drug is cheap, and the side-effect profile is mild.
Typical Dosing — Where to Start and Where to Land
The standard target dose in fibromyalgia is 4.5 mg once daily. Almost every published trial and almost every experienced LDN prescriber aims there. Some patients do better at slightly lower (3.0 mg) or slightly higher (up to 6 mg) doses, but 4.5 mg is the anchor.
You do not start at 4.5 mg. Jumping straight to the target is the single biggest reason patients quit LDN in the first two weeks — vivid dreams, insomnia, and headaches are far worse if you skip titration.
A typical starting dose is 0.5 mg to 1.5 mg. From there you climb over several weeks toward 4.5 mg, pausing or dropping back if side effects flare.
LDN comes as a capsule, a sublingual tablet, or a liquid from a compounding pharmacy. The liquid is the most flexible — you can draw up 0.5 mg increments — and is popular with sensitive patients. Capsules are most common and perfectly fine for most people.
Slow vs Fast Titration Schedules
Two mainstream schedules exist. Neither is “right” — the choice depends on how sensitive you are to new medications.
Fast titration (for most people).
- Weeks 1–2: 1.5 mg nightly.
- Weeks 3–4: 3.0 mg nightly.
- Week 5 onward: 4.5 mg nightly.
Slow titration (for medication-sensitive patients, which includes many fibromyalgia patients and anyone with coexisting POTS, MCAS, or ME/CFS).
- Weeks 1–2: 0.5 mg nightly.
- Weeks 3–4: 1.0 mg nightly.
- Weeks 5–6: 1.5 mg nightly.
- Weeks 7–8: 2.0 mg nightly.
- Weeks 9–10: 3.0 mg nightly.
- Weeks 11–12: 4.0 mg nightly.
- Week 13 onward: 4.5 mg nightly.
If a dose increase causes a flare — worse pain, worse sleep, new headaches — drop back to the previous dose for another two weeks, then try again. There is no prize for getting to 4.5 mg quickly. The target matters; the timeline does not.
Compounding Pharmacies, Cost, and Insurance
Low-dose naltrexone is not manufactured commercially. The only commercial naltrexone product is the 50 mg tablet, which is impractical to break into 4.5 mg pieces. Instead, your prescriber sends the prescription to a compounding pharmacy, which measures out a tiny amount of pure naltrexone and mixes it into a capsule base (typically avicel or cellulose — ask for a filler without lactose or calcium carbonate, which can blunt absorption).
Well-known U.S. LDN compounders include Skip's Pharmacy (Florida), Belmar Pharmacy (Colorado), and The Compounder (Illinois). Most will ship nationwide. Typical cost is $35–$70 per month out of pocket.
Insurance rarely covers LDN. Because it is off-label and compounded, most U.S. insurance plans deny it outright. A few plans reimburse a percentage under “compounded medications” riders. HSA and FSA accounts usually do cover it with a prescription — use them if you have them.
Ask the compounding pharmacy whether they offer a cash-pay discount for a three-month supply. Many do. Ask if they have a preferred capsule filler for sensitive patients — avicel (microcrystalline cellulose) and sucrose are usually the safest choices.
Side Effects — What to Expect
Compared with most pain medications, LDN is remarkably well tolerated. Most side effects appear in the first two to four weeks and fade as the body adapts.
- Vivid dreams. By far the most common. Dreams become louder, longer, stranger — sometimes interesting, sometimes unpleasant. Usually fades within two to four weeks. If dreams are disturbing, switch from bedtime to morning dosing (see timing debate).
- Insomnia or fragmented sleep. Trouble falling or staying asleep during the first week or two of each dose increase. Usually transient. If persistent, move to morning dosing.
- Headache. Usually mild, front-of-head, resolves with hydration and a slower titration.
- GI upset. Mild nausea, loose stools, or bloating. Taking the dose with a small amount of food (a cracker or a spoonful of yogurt) helps.
- Temporary pain or fatigue flare. Some fibromyalgia patients feel worse for the first one to two weeks before improving. If the flare is tolerable, push through. If it is severe, drop back one dose step.
Serious adverse events are rare. LDN does not cause weight gain, sexual dysfunction, dependence, tolerance, or withdrawal. Long-term use (five-plus years) has not produced a signal for organ toxicity in the published cohorts.
Drug Interactions — Opioids and Surgery
This is the most important safety section in the article. Read it twice.
LDN and opioids do not mix. If you are taking any opioid medication — oxycodone, hydrocodone, tramadol, codeine, morphine, fentanyl, buprenorphine, tapentadol — you cannot start LDN. Naltrexone will displace the opioid from its receptors and can trigger an abrupt, severe withdrawal reaction. This includes over-the-counter cough syrups containing codeine and some diarrhea medicines (loperamide in high doses, diphenoxylate).
If you are on an opioid and want to try LDN, you must taper off the opioid first, under supervision of the prescriber who started it. A typical safe gap between the last opioid dose and the first LDN dose is 7–10 days for short-acting opioids and 2–3 weeks for methadone or long-acting formulations. Your prescriber will individualize this.
Surgery planning. Because LDN blocks opioid receptors for several hours after each dose, surgical anesthesiologists cannot give you normal amounts of opioid pain medication during or after an operation. Stop LDN at least 3–5 days before a planned surgery, and tell the anesthesiologist you have been taking it. After surgery, restart LDN only once you are fully off post-operative opioids — typically one to two weeks later, starting again at a low dose.
For emergency surgery, tell the team immediately. Anesthesia can be managed with non-opioid options (ketamine, regional blocks, NSAIDs) and higher-than-usual opioid doses if required.
Other drug interactions are minimal. LDN is compatible with SSRIs, SNRIs, gabapentin, pregabalin, low-dose tricyclics (amitriptyline, nortriptyline), muscle relaxants, and all of the common fibromyalgia medications except opioids.
Morning vs Bedtime — The Timing Debate
For two decades, LDN was dosed almost exclusively at bedtime. The theory was that endorphin release peaks around 2–4 a.m., so a brief opioid blockade late at night would maximize the rebound.
In practice, the bedtime dose is the reason many fibromyalgia patients quit. Vivid dreams, sleep fragmentation, and restless-legs sensations are all more likely at night. Because fibromyalgia patients already have non-restorative sleep (see sleep article), anything that worsens sleep is a dealbreaker.
The modern consensus — supported by open-label experience — is that morning dosing works just as well for pain reduction, with far fewer sleep side effects. Many LDN-experienced prescribers now start patients on morning dosing by default.
Practical guidance:
- If sleep is already fragile, start with morning dosing.
- If you start at bedtime and develop dream or insomnia problems, switch to morning for two weeks before giving up on LDN entirely.
- If both timings cause side effects, try split dosing: half the dose in the morning, half at noon.
What “Working” Looks Like
LDN is slow. It is not a pill you take and feel an hour later. Realistic expectations are critical to not giving up before it has had a chance.
- Weeks 1–2: side effects (vivid dreams, insomnia) often outweigh benefit. Some patients feel slightly worse.
- Weeks 4–6: side effects fade. Subtle improvements: a little less morning stiffness, slightly more stamina, dropping one nap, sleeping thirty minutes longer.
- Weeks 8–12: responders notice a real drop in baseline pain, usually 20–40%. Fewer bad-pain days. Flares are less intense and shorter.
- Months 3–6: full effect. Some patients continue improving through month six.
In the Younger trials, responders reduced pain by an average of roughly 30%. That is not a cure. It is a meaningful improvement — the difference between housebound and functional, between disabled and part-time working. Combined with sleep work, pacing, and nutrient repletion (see nutrient gaps), the total quality-of-life improvement can be substantial.
Track your response with a simple daily 0–10 pain score, logged in a notebook or phone app, starting two weeks before you begin LDN and continuing for three months. Without a written record, it is nearly impossible to tell whether a slow, steady shift is real.
When to Give Up
LDN does not work for everyone. The trial data suggest roughly one in three patients gets a meaningful response. The other two-thirds notice little change.
The honest decision point is three months at 4.5 mg. If you have taken the full target dose consistently for twelve weeks, have tracked your pain and fatigue daily, and see no pattern of improvement, LDN is unlikely to help you. Stopping is straightforward — no taper is required. Simply discontinue.
Before quitting, make sure you have honestly tested:
- The full 4.5 mg target dose (not stuck at 3.0 mg because of side effects).
- Both morning and bedtime timings.
- A cellulose-based capsule (some patients fail on lactose or calcium carbonate fillers and succeed after switching).
- At least twelve weeks at target dose.
A small subset of patients respond only at doses slightly above 4.5 mg (up to 6 mg). If you are a partial responder at 4.5 mg, it is reasonable to ask your prescriber about a one-month trial at 6 mg before stopping.
Practical Tips for Starting
- Find a prescriber who knows LDN. Many rheumatologists and pain specialists are comfortable with it. Functional medicine and integrative clinics almost always are. The LDN Research Trust keeps a searchable list of LDN-experienced prescribers worldwide.
- Use a compounding pharmacy with a good LDN reputation and ask for an avicel (microcrystalline cellulose) filler.
- Start low. Do not let a prescriber push you to 4.5 mg in week one just because that is the target dose.
- Track pain, sleep, and fatigue daily — a simple 0–10 score for each.
- Do not start LDN during a fibromyalgia flare. The first two weeks are noisy enough; adding it on top of a flare makes side effects hard to interpret.
- Do not start LDN the same week as a new SSRI, a new diet, or a new supplement stack. Change one thing at a time so you can tell what is doing what.
- Carry a medical-alert card noting that you take naltrexone, in case of emergency surgery or ER opioid administration.
Key Research Papers
- Younger J, Noor N, McCue R, Mackey S. Low-dose naltrexone for the treatment of fibromyalgia: findings of a small, randomized, double-blind, placebo-controlled, counterbalanced, crossover trial assessing daily pain levels. Arthritis Rheum. 2013;65(2):529–538.
- Younger J, Mackey S. Fibromyalgia symptoms are reduced by low-dose naltrexone: a pilot study. Pain Med. 2009;10(4):663–672.
- Parkitny L, Younger J. Reduced pro-inflammatory cytokines after eight weeks of low-dose naltrexone for fibromyalgia. Biomedicines. 2017;5(2):16.
- Younger J, Parkitny L, McLain D. The use of low-dose naltrexone (LDN) as a novel anti-inflammatory treatment for chronic pain. Clin Rheumatol. 2014;33(4):451–459.
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on LDN in fibromyalgia and related neuroimmune pain conditions:
- Low-dose naltrexone and fibromyalgia
- Naltrexone, TLR4, and microglial activation
- Low-dose naltrexone for chronic pain
- Naltrexone and inflammatory cytokines
- Fibromyalgia and neuroinflammation
- Naltrexone and endorphin rebound
- LDN dosing and titration
- LDN in ME/CFS and overlapping conditions
Connections
- Nutrient Gaps in Fibromyalgia
- POTS
- Sleep and Non-Restorative Sleep
- Central Sensitization Explained
- Chronic Fatigue Syndrome
- Fibromyalgia Overview
- Chronic Pain
- MCAS
- Fibromyalgia and ME/CFS Overlap
- ACR 2016 Diagnostic Criteria
- Small-Fiber Neuropathy Overlap
- Exercise Pacing and Graded Movement
- Low-Dose Naltrexone for POTS
- Low-Dose Naltrexone
- Crohn's Disease
- Insomnia
- Fatigue
- Cannabis and CBD for Fibromyalgia