Low-Dose Naltrexone (LDN) for POTS
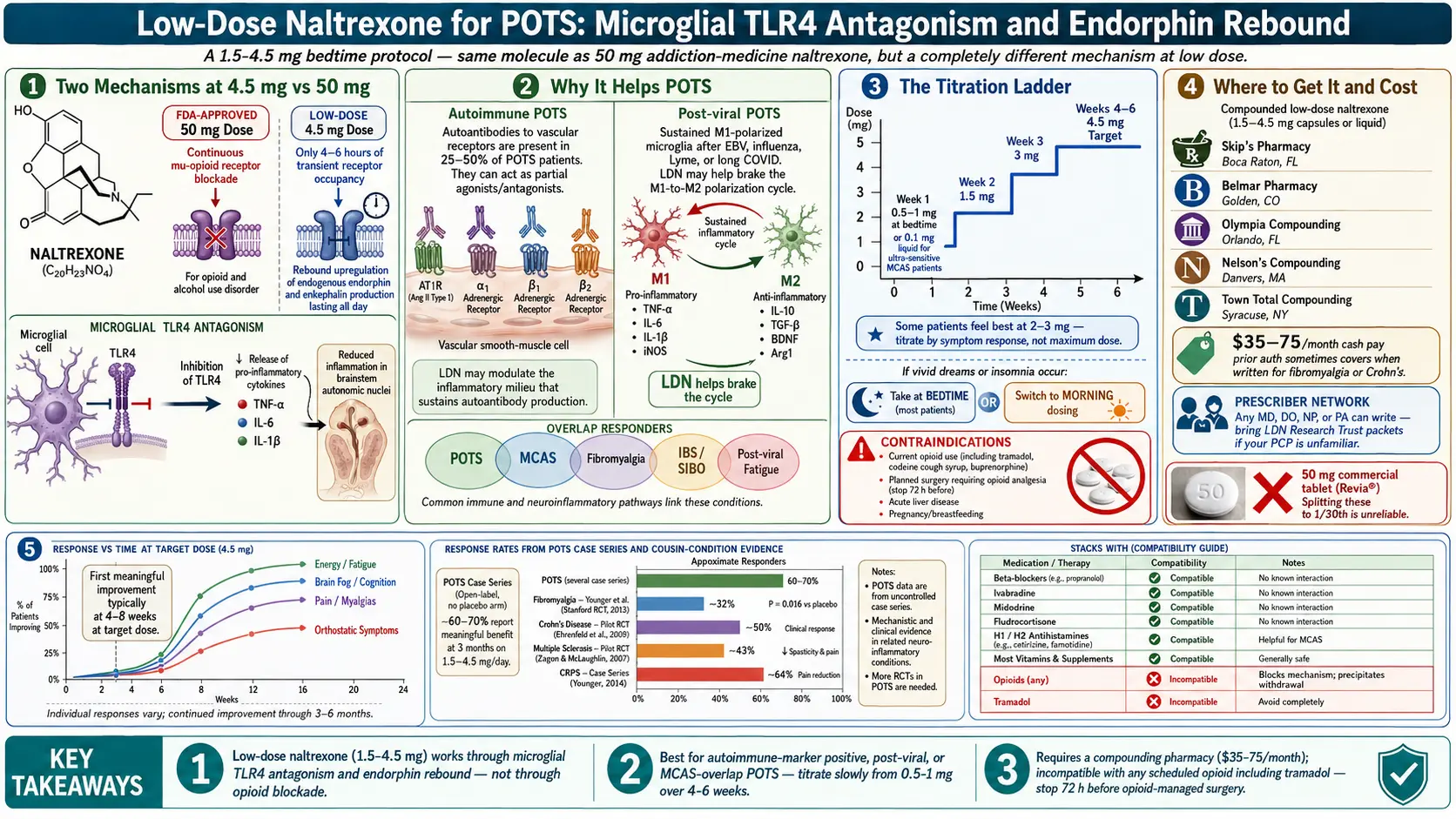
Table of Contents
- What LDN Actually Is
- Why It Helps POTS
- What the Evidence Shows
- Who LDN Is Best For
- Who Should NOT Take LDN
- Starting Protocol — Titration
- Side Effects and How to Manage Them
- Where to Get It — Compounding Pharmacies
- Getting a Prescription When Your Doctor Isn’t Familiar
- Combining LDN with Other POTS Treatments
- Timeline of Expected Effects
- What “Worked” Looks Like
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
1. What LDN Actually Is
Naltrexone is a pure opioid receptor antagonist. At its FDA-approved dose of 50 mg daily, it’s used to treat opioid dependence and alcohol use disorder — it blocks the mu-opioid receptor continuously so that opioids and alcohol-driven endorphin reward produce no high.
At low doses (1.5–4.5 mg), the drug behaves almost nothing like its 50 mg self. Two completely different mechanisms take over:
- Transient opioid receptor blockade. At 4.5 mg, naltrexone occupies opioid receptors for only about 4–6 hours. That brief blockade fools the hypothalamus into thinking endorphins are in short supply, triggering a rebound upregulation of endogenous endorphin and enkephalin production that lasts all day.
- TLR4 antagonism on microglia. Low-dose naltrexone inhibits toll-like receptor 4 on microglia in the central and peripheral nervous system, damping the release of pro-inflammatory cytokines (TNF-alpha, IL-6, IL-1-beta) that drive chronic neuroinflammation.
The opioid-receptor blockade itself does nothing therapeutic — it’s the rebound and the microglial effects that matter. That is why LDN doesn’t feel like anything when you take it; it isn’t working through an acute pharmacologic effect the way a beta-blocker does.
2. Why It Helps POTS
Two strands of POTS research point directly at what LDN does.
Autoimmunity. Roughly 25–50% of POTS patients in screened cohorts carry functional autoantibodies against adrenergic and muscarinic receptors — most notably the AT1R (angiotensin II type 1 receptor), alpha-1 adrenergic receptor, and beta-1 and beta-2 adrenergic receptors. These antibodies act as partial agonists or antagonists and appear to drive at least a subset of the orthostatic dysregulation.
Neuroinflammation. Post-viral POTS (post-EBV, post-influenza, post-Lyme, long COVID) shows features consistent with chronic low-grade neuroinflammation — persistent fatigue, brain fog, post-exertional malaise, sensitivity to light and sound. Microglia in a sustained M1-polarized state amplify autonomic dysregulation in the brainstem nuclei that govern heart rate and vascular tone.
LDN’s microglial TLR4 effect is a plausible brake on that inflammatory loop, and its endorphin-rebound effect modulates pain processing and autonomic balance. Case series and patient-reported outcomes describe improvements in fatigue, brain fog, orthostatic symptoms, pain, and mast cell overlap symptoms.
3. What the Evidence Shows
Honest summary: there are no large randomized controlled trials of LDN for POTS specifically. The evidence base is pieced together from three sources.
- POTS-specific case series from Dysautonomia International collaborators and the Stanford Autonomic Clinic describe meaningful symptom improvement in roughly 60–70% of patients who complete a 3-month trial. Open-label, no placebo arm — interpret accordingly.
- RCT-grade evidence in adjacent conditions. High-quality positive trials exist in fibromyalgia (Younger and colleagues, Stanford), Crohn’s disease, multiple sclerosis, and complex regional pain syndrome. All share mechanistic overlap with POTS: neuroinflammation, autonomic involvement, chronic pain.
- Emerging long-COVID trials — a close cousin of post-viral POTS — are reporting benefit in fatigue, brain fog, and post-exertional malaise.
LDN is not a miracle drug for POTS. Expect modest-to-moderate improvement, most useful when stacked with other POTS treatments rather than used as a standalone therapy.
4. Who LDN Is Best For
Response is not uniform. The subgroups most likely to benefit:
- POTS with autoimmune markers — positive AT1R or beta-1 adrenergic receptor antibodies, elevated ANA, Hashimoto’s or other thyroid autoimmunity
- Post-viral POTS — post-EBV, post-Lyme, post-influenza, long COVID
- POTS with prominent fatigue, brain fog, or post-exertional malaise
- POTS with MCAS overlap (mast cell activation)
- POTS with fibromyalgia overlap
- POTS with IBS or SIBO overlap (LDN doubles as a gentle gut prokinetic at these doses)
5. Who Should NOT Take LDN
- Current opioid use — naltrexone will block your painkillers and can trigger acute withdrawal. Includes tramadol, codeine-containing cough syrups, and buprenorphine.
- Planned surgery requiring opioid pain management — stop LDN at least 72 hours before, restart after opioids are no longer needed.
- Acute liver disease — caution; check LFTs with your prescriber.
- Pregnancy or breastfeeding — insufficient data; most prescribers pause.
6. Starting Protocol — Titration
The classic target dose is 4.5 mg at bedtime, reached by slow titration. Starting at the target dose is the most common reason patients bounce off LDN with vivid dreams or a symptom flare.
- Week 1: 0.5–1 mg at bedtime. Ultra-sensitive patients (common in MCAS and POTS) should start at 0.1 mg — compounding pharmacies can provide a liquid for precise micro-dosing.
- Week 2: 1.5 mg at bedtime if tolerated.
- Week 3: 3 mg at bedtime.
- Weeks 4–6: 4.5 mg at bedtime — the target.
A meaningful minority of patients feel best on 2–3 mg and worse at 4.5 mg. Titrate by symptom response, not by reaching the maximum dose. If vivid dreams or insomnia persist, try morning dosing — many patients tolerate it equally well and some prefer it.
7. Side Effects and How to Manage Them
- Vivid dreams — the most common side effect; typically occurs in the first 1–2 weeks and fades. If persistent or distressing, switch to morning dosing.
- Insomnia or sleep disruption — lower the dose by 50% for a week or move the dose to morning.
- GI upset — take with food. Usually resolves within days.
- Headache — reduce dose by 50% for one week, then re-escalate slowly.
- Transient symptom flare in the first week — fatigue, orthostatic worsening, or pain uptick; common and often called a “herx” (a loose analogy, not a literal Herxheimer reaction). Push through if tolerable, drop back a step if severe.
- Rare: liver enzyme elevation — check LFTs at 6 weeks if you have any liver history or unexplained fatigue.
8. Where to Get It — Compounding Pharmacies
LDN is not a stock pharmacy item at these doses. Commercial naltrexone comes as 50 mg tablets; trying to split a tablet into 1/30th is unreliable and potentially unsafe. You need a compounding pharmacy that makes LDN capsules or liquid in the exact dose prescribed (0.5, 1, 1.5, 2, 3, 4.5 mg are standard).
Well-known LDN compounding pharmacies in the US:
- Skip’s Pharmacy (Boca Raton, Florida)
- Belmar Pharmacy (Golden, Colorado)
- Nelson Pharmacy (Massachusetts)
- Olympia Compounding Pharmacy (Orlando, Florida)
- Town Total Health (New York)
Cost: typically $35–75 per month cash pay. Some insurers will cover with prior authorization, especially when the prescribing indication is fibromyalgia, Crohn’s, or another approved off-label use. Any MD, DO, NP, or PA can write the prescription — no specialty licensure required.
9. Getting a Prescription When Your Doctor Isn’t Familiar
Most primary care physicians have never prescribed LDN. You’ll likely need to arrive prepared.
- Print the LDN Research Trust patient information packet and physician information packet. Hand both to your PCP.
- Bring the Dysautonomia International clinician summary on LDN.
- If your PCP declines, many POTS-aware telemedicine clinics (Dysautonomia Consultants, Autonomic Medical Specialists, and similar) prescribe LDN routinely.
- The LDN Research Trust physician directory lists prescribers by state and country.
An initial visit plus a compounding script is usually all it takes. Refills are straightforward once the pharmacy relationship is set up.
10. Combining LDN with Other POTS Treatments
LDN plays well with essentially every standard POTS medication:
- Beta-blockers (propranolol, metoprolol)
- Ivabradine
- Midodrine
- Fludrocortisone
- H1 and H2 antihistamines (for MCAS overlap)
- Most vitamins, minerals, and non-opioid analgesics
The one real-world constraint is opioids — any scheduled opioid analgesic, including tramadol, is incompatible while LDN is onboard. For stacking strategy with other POTS drugs, see the Medications Guide.
11. Timeline of Expected Effects
LDN is slow. Most patients notice the first meaningful shift at 4–8 weeks at target dose. A minority of responders feel it within 1–2 weeks, usually as improved energy or reduced brain fog first.
A fair trial is at least 3 months at target dose before deciding you’re a non-responder. If you stopped titrating at 3 mg because you felt great, stay there — target dose is not the same as optimal dose.
12. What “Worked” Looks Like
Responders typically describe:
- Reduced orthostatic symptoms — less pre-syncope, less heart-pounding on standing
- Less post-exertional malaise after modest activity
- Better cognitive function — quicker word-finding, longer reading stamina
- Fewer MCAS flares or reduced flare intensity
- Improved exercise tolerance (usually in conjunction with a graded program)
The effect is often subtle at first. A weekly symptom diary — standing heart rate, fatigue score, brain fog score, flare count — is the most reliable way to spot whether LDN is earning its place in your stack.
13. Key Research Papers
- Younger J, Parkitny L, McLain D. The use of low-dose naltrexone (LDN) as a novel anti-inflammatory treatment for chronic pain. Journal of Inflammation Research. 2014.
- Toljan K, Vrooman B. Low-Dose Naltrexone (LDN) — Review of Therapeutic Utilization. Biomedicines. 2018;6(3):82.
- Younger J, Noor N, McCue R, Mackey S. Low-dose naltrexone for the treatment of fibromyalgia. Clinical Rheumatology. 2014.
- Low-dose naltrexone and mast-cell mediator-related disorders (review). Journal of Allergy and Clinical Immunology. 2019.
14. Research Papers
Curated PubMed topic searches — each link opens a live query so you always see the most recent literature.
- PubMed: Low-dose naltrexone for POTS
- PubMed: LDN for fibromyalgia
- PubMed: LDN and autoimmune disease
- PubMed: Naltrexone, microglia, and TLR4
- PubMed: LDN for long COVID
- PubMed: Naltrexone and mast cells
- PubMed: LDN for chronic fatigue syndrome
Connections
- POTS Overview
- Medications Guide
- POTS Subtypes
- Compression and Exercise Program
- Salt and Hydration Protocol
- Vagus Nerve and Autonomic Retraining
- Tilt Table Test
- POTS/MCAS/EDS Triad
- MCAS
- Fibromyalgia
- ME/CFS
- SIBO
- Low-Dose Naltrexone (Remedy)
- Post-Viral Triggers: EBV and Others
- Brain Fog
- Fatigue
- Low Dose Naltrexone for Fibromyalgia
- LDN Abilify and Experimental Therapies