Central Sensitization Explained
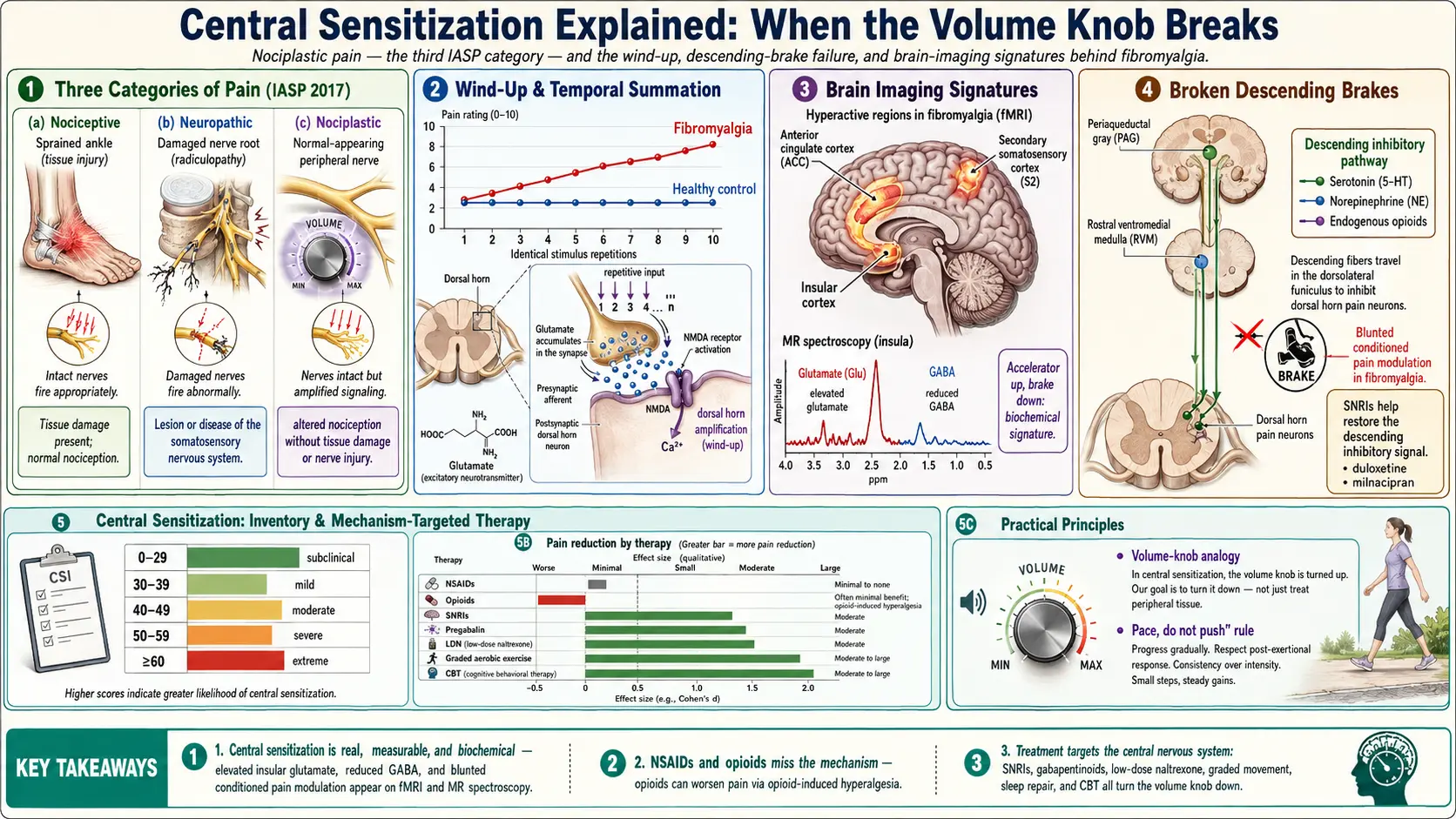
Table of Contents
- What Central Sensitization Is
- Nociplastic Pain — The Third Category
- The Volume-Knob Analogy
- Wind-Up and Temporal Summation
- What Brain Imaging Shows
- Why Your MRI and Blood Work Look Normal
- Broken Brakes: Descending Pain Modulation
- Why NSAIDs and Opioids Often Fail
- What Actually Helps — and Why
- The Central Sensitization Inventory (CSI)
- Daily-Life Strategies That Turn the Volume Down
- Talking to Doctors Who Do Not Know This Language
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
What Central Sensitization Is
Central sensitization is a change in how your central nervous system — your spinal cord and brain — processes signals from the body. In a normally calibrated nervous system, a light touch registers as light touch, a pinch registers as a pinch, and a stubbed toe registers as a short burst of pain that fades in minutes. In a centrally sensitized system, those same inputs get amplified, prolonged, and broadened. A light touch can feel like sandpaper. A minor bump can throb for hours. Clothing seams, tag labels, a bra strap, a handshake, a hug — all of these can cross the threshold from sensation into genuine pain.
Nothing is wrong with your skin, your muscles, or your joints in the ordinary sense. The wiring between tissue and brain has become hyper-reactive. Pain neurons fire more easily, fire longer, and recruit neighboring neurons that would normally stay quiet. The pain is real. It is measurable in laboratory settings. It is visible on specialized brain scans. It is not imagined, not exaggerated, and not a character flaw. It is a neurobiological condition with a name.
Central sensitization is the mechanism that best explains fibromyalgia, but it is not unique to fibromyalgia. It shows up in irritable bowel syndrome, chronic migraine, interstitial cystitis, vulvodynia, temporomandibular joint disorder, tension-type headache, chronic low back pain that persists after tissue has healed, and large subsets of complex regional pain syndrome and post-surgical chronic pain. Many patients carry more than one of these diagnoses simultaneously — because the underlying problem is not in each organ, it is in the shared pain-processing system.
Nociplastic Pain — The Third Category
In 2017 the International Association for the Study of Pain (IASP) formally recognized a third mechanistic category of pain called nociplastic pain. The word combines "noci-" (pain) with "-plastic" (changed, remodeled). It sits alongside the two categories physicians already used for a century:
- Nociceptive pain — pain from actual or threatened tissue damage. A sprained ankle, a burn, a broken bone. The nervous system is working correctly; it is reporting real injury.
- Neuropathic pain — pain from identifiable damage to the nervous system itself. A pinched nerve root, diabetic nerve damage, post-shingles pain. The wires are broken.
- Nociplastic pain — pain arising from altered nociception without clear tissue damage or nerve injury. The hardware looks intact, but the software is miscalibrated.
This third category was not invented to be polite. It was added because the first two categories failed to describe what was happening in millions of patients. A woman with fibromyalgia whose MRI, nerve conduction studies, rheumatology panel, and inflammatory markers all come back clean does not have "nothing." She has nociplastic pain. Giving that experience a name was a significant step, and it shifted how specialists now talk about fibromyalgia, chronic pelvic pain, and a long list of other conditions formerly dismissed as medically unexplained.
Nociplastic pain and central sensitization are closely related but not identical. Central sensitization is the specific mechanism; nociplastic pain is the broader clinical category. In practice, the terms are often used interchangeably in patient-facing materials.
The Volume-Knob Analogy
The single most useful mental image for central sensitization is a volume knob on a stereo. Imagine your nervous system constantly receiving inputs from the body — pressure, temperature, movement, stretch — and piping them through an amplifier before they reach the conscious mind. In most people, the amp sits at a reasonable level. A gentle touch comes through as a gentle touch. A noxious stimulus is clearly louder than a neutral one.
In central sensitization, someone has turned the knob up to eleven and then broken it off. Every input — including inputs that should barely register — comes through the amp at distorted, painful volume. The brain has no easy way to turn it back down. A firm handshake, a warm shower that is slightly too warm, a short walk on a hard floor, the pressure of a seatbelt across the collarbone — all of these can trigger real pain signals that a calibrated nervous system would have dismissed as noise.
This analogy matters for two reasons. First, it explains to skeptical friends, family, and clinicians why you can hurt badly when nothing on imaging looks wrong. Second, it points toward the treatment goal: we are not trying to fix a tissue. We are trying to turn the volume down. Every therapy that works in fibromyalgia works by nudging that knob in the right direction.
Wind-Up and Temporal Summation
Two laboratory phenomena define central sensitization in measurable terms.
Wind-up refers to the progressive increase in the response of spinal cord neurons to repeated, identical inputs. In healthy people, if you tap the skin with the same pressure once per second, the spinal signal stays roughly flat. In a centrally sensitized person, each tap produces a slightly larger signal than the one before. By the tenth tap, the signal is several times the starting value — even though nothing about the stimulus changed. The wiring is learning to fire harder at the same prompt.
Temporal summation is the perceptual counterpart. Researchers apply a series of identical mildly uncomfortable heat pulses to the forearm and ask the patient to rate each one. Healthy controls rate them flat — a 3 out of 10, then another 3, then another 3. Fibromyalgia patients rate them as climbing — 3, then 5, then 7, then 8 — until a stimulus that started as annoying feels frankly painful. Temporal summation can be quantified in the lab and is reliably elevated in fibromyalgia and related conditions.
The practical consequence for daily life is what patients often call "the trap of doing a little too much." A mildly unpleasant activity that would be fine in isolation (one load of laundry, ten minutes of vacuuming, a short shopping trip) stacks up into a full-volume pain flare when repeated across an afternoon. This is not weakness or deconditioning. It is wind-up, running in real time, in your spinal cord.
What Brain Imaging Shows
If you only read mainstream news, you might still think fibromyalgia is a "diagnosis of exclusion" with no objective findings. That is outdated. Functional MRI, positron emission tomography, and magnetic resonance spectroscopy studies over the past two decades have documented consistent, reproducible differences in the brains of fibromyalgia patients compared to controls. The key regions:
- Insular cortex — hyperactive at rest and during mild stimulation. The insula integrates body sensation into emotional meaning.
- Anterior cingulate cortex (ACC) — hyperactive. The ACC is the brain's pain-attention hub; it decides how much cognitive real estate a sensation deserves.
- Secondary somatosensory cortex (S2) — activated by pressures that do not activate it in controls.
- Periaqueductal gray (PAG) and rostral ventromedial medulla (RVM) — nodes of the descending pain-inhibition system that function abnormally.
- Default mode network — abnormal connectivity, consistent with the cognitive "fibro fog" symptom.
Magnetic resonance spectroscopy studies have found elevated glutamate (the main excitatory neurotransmitter) in the insula and posterior cingulate in fibromyalgia patients, with levels correlating to reported pain severity. Other studies find reduced GABA (the main inhibitory neurotransmitter) in related regions. Put bluntly: the accelerator neurotransmitter is up, the brake neurotransmitter is down. That is a biochemical signature, not an imaginary one.
Why Your MRI and Blood Work Look Normal
Every fibromyalgia patient eventually hears some version of "everything came back normal." This sentence lands like a door slamming. It feels like being told the pain is not real. It is worth unpacking exactly what standard testing can and cannot see.
A standard lumbar MRI images the anatomy of your back — discs, bones, nerve roots, ligaments. It is excellent for herniated discs, arthritis, fractures, and tumors. It is completely blind to the function of your spinal cord's pain-processing neurons. A normal lumbar MRI in a fibromyalgia patient is the expected result. It does not rule out the diagnosis; it supports it.
A rheumatology panel (CBC, CMP, CRP, ESR, ANA, rheumatoid factor, CCP antibody, TSH) screens for inflammatory and autoimmune diseases. Fibromyalgia is not an inflammatory disease in the classical sense, so these markers are usually normal or mildly elevated for unrelated reasons. Again, normal results here rule out rheumatoid arthritis, lupus, and thyroid dysfunction — they do not rule out a pain-amplification disorder.
The tests that can detect central sensitization — functional MRI, quantitative sensory testing for temporal summation, conditioned pain modulation paradigms, magnetic resonance spectroscopy — are research tools. They exist in academic pain centers but are not billable in a standard clinic. Until they become mainstream, the diagnosis will continue to rest on clinical criteria and a validated questionnaire called the Central Sensitization Inventory.
Broken Brakes: Descending Pain Modulation
Your brain does not just receive pain signals. It actively regulates them. A network of pathways running from the brainstem down the spinal cord — collectively called the descending pain modulation system — acts as a set of brakes. It can suppress incoming pain signals at the spinal level before they ever reach consciousness. This is why a soldier can finish carrying a wounded comrade before noticing their own shrapnel wound, and why a concentrated athlete can play through what would otherwise be disabling discomfort.
The key chemicals running those brakes are serotonin, norepinephrine, and endogenous opioids (your own body's morphine-like molecules). The key brain regions are the periaqueductal gray in the midbrain and the rostral ventromedial medulla in the brainstem.
In fibromyalgia and related nociplastic conditions, the descending brakes do not work well. Researchers measure this with a technique called conditioned pain modulation: they apply a painful stimulus in one place (hand in cold water) while measuring pain threshold in another (pressure on the leg). In healthy people, the brain's brakes kick in and the second stimulus feels less painful — "pain inhibits pain." In fibromyalgia patients, this effect is blunted or absent. The brakes do not engage.
This single finding explains why drugs that raise serotonin and norepinephrine in the spinal cord — the SNRIs duloxetine and milnacipran — help a meaningful fraction of patients. They are not treating depression incidentally. They are trying to restore the chemical signal of the descending brakes.
Why NSAIDs and Opioids Often Fail
This is the single most important practical consequence of understanding central sensitization.
NSAIDs (ibuprofen, naproxen, diclofenac, celecoxib) work by reducing inflammation at the site of tissue injury. They block prostaglandin production in damaged tissue. If your pain is not driven by tissue inflammation — and in pure fibromyalgia it is not — then NSAIDs have almost nothing to act on. Patients often report that a full dose of ibuprofen takes the edge off no more than a placebo would. That is not a failure of the patient. It is an NSAID working correctly on a problem it cannot fix.
Opioids (oxycodone, hydrocodone, tramadol, morphine) are even more problematic. In the short term they blunt pain perception broadly, including nociplastic pain. But across weeks and months they induce opioid-induced hyperalgesia — a paradoxical state in which the nervous system becomes more sensitive to pain, not less. They also downregulate the body's own endogenous opioid system, which is part of the descending brakes. In a fibromyalgia patient, long-term opioids can make the underlying problem measurably worse while creating tolerance, dependence, constipation, cognitive fog, and overdose risk. Current specialty guidelines actively recommend against chronic opioid therapy for fibromyalgia.
This is why well-meaning patients can spend ten years rotating through anti-inflammatories, muscle relaxants, and escalating opioid prescriptions with no durable benefit. They were prescribed the drug classes that match nociceptive pain, while their pain is nociplastic. Switching the target is how treatment finally starts to work.
What Actually Helps — and Why
The therapies with the strongest evidence in fibromyalgia all share one feature: each of them adjusts the central nervous system rather than the peripheral tissue.
SNRI antidepressants — duloxetine (Cymbalta) and milnacipran (Savella). FDA-approved specifically for fibromyalgia. They raise serotonin and norepinephrine in the spinal cord and help restore descending pain inhibition. Effective dose for pain is typically 60–120 mg daily for duloxetine.
Gabapentinoids — pregabalin (Lyrica) and gabapentin (Neurontin). Pregabalin is also FDA-approved for fibromyalgia. They bind the alpha-2-delta subunit of voltage-gated calcium channels on overactive neurons and reduce the release of excitatory neurotransmitters — effectively dampening the wind-up signal at the spinal cord. Common side effects are sedation, dizziness, and weight gain; start low, titrate slowly.
Low-dose naltrexone (LDN). At 1.5–4.5 mg at bedtime — a fraction of the addiction-medicine dose — naltrexone acts on microglia (the immune cells of the central nervous system) and appears to reduce neuroinflammation that contributes to central sensitization. Several small trials show meaningful pain reduction. Inexpensive and generally well-tolerated. See the dedicated LDN article.
Graded aerobic exercise. Regular, gentle, progressively built-up aerobic activity (walking, swimming, recumbent biking, water aerobics) is one of the most effective interventions. It raises endogenous opioids and BDNF, improves descending pain modulation, and retrains the autonomic nervous system. The catch is pacing — too much too fast triggers a flare and teaches the patient to avoid exercise entirely. See exercise pacing.
Cognitive behavioral therapy (CBT) and pain neuroscience education. CBT does not treat pain by convincing you it is imaginary. It retrains the brain's pain-attention circuits, reduces catastrophizing, and rebuilds a sense of agency. Pain neuroscience education — simply learning the material on this page — has been shown in randomized trials to reduce pain intensity by itself.
Sleep repair. Non-restorative sleep is both a symptom and a driver of central sensitization. Restoring deep slow-wave sleep measurably lowers next-day pain sensitivity. See sleep and non-restorative sleep.
No single therapy is curative. A combination of a central-acting medication, graded movement, sleep repair, and some form of cognitive or mind-body work produces the durable results.
The Central Sensitization Inventory (CSI)
The Central Sensitization Inventory is a free, validated, 25-item self-report questionnaire developed by Mayer, Neblett, and colleagues in 2012. It asks how often you experience symptoms that cluster with central sensitization — widespread pain, unrefreshing sleep, cognitive fog, sensitivity to light and sound, irritable bowel symptoms, bladder symptoms, jaw pain, stress-triggered symptoms. Each item is scored 0–4 for a total of 0–100.
Score interpretation:
- 0–29 — subclinical. Central sensitization is unlikely to be a significant driver.
- 30–39 — mild.
- 40–49 — moderate.
- 50–59 — severe.
- 60 and above — extreme. Central sensitization is very likely the dominant mechanism.
A score above 40 in a patient with widespread pain of unclear origin is a strong signal that nociplastic pain is present and that treatment should be directed at the central nervous system. The CSI is not diagnostic by itself — no questionnaire is — but it reliably identifies which patients are likely to respond to central-acting therapies. Taking it on your own and sharing the result with your physician is a reasonable way to open the conversation.
Daily-Life Strategies That Turn the Volume Down
Daily life is where central sensitization is won or lost. Medication sets the ceiling; habits determine where you actually live inside that ceiling.
- Pace, do not push. Break tasks into 20–30 minute blocks with real rests in between. Stop before the pain rises, not after. The nervous system learns from what you do, and pushing past a wind-up threshold teaches it to wind up faster next time.
- Sleep hygiene as medicine. Same bedtime seven nights a week. Cool dark bedroom. No screens the last hour. Deep, consolidated sleep measurably lowers next-day pain thresholds.
- Move every day. A ten-minute walk counts. Stillness deconditions the descending brakes; daily rhythmic movement strengthens them.
- Warm water. Warm pool exercise, warm baths, hot showers before bed. Warmth reduces muscle guarding and downregulates sympathetic tone.
- Mind-body practice. Slow nasal breathing, body-scan meditation, tai chi, yin yoga. The evidence for tai chi in fibromyalgia is substantial and well-replicated. Twenty minutes a day is enough.
- Limit sensory overload. Noise-canceling headphones, sunglasses indoors under fluorescent lights, soft fabrics, tag-free clothing. These are not luxuries; they reduce the incoming load on an already-amplified system.
- Eat regularly. Blood sugar swings and skipped meals amplify pain. Three steady meals, protein at breakfast.
- Track flares, not only pain scores. After each bad week, write down the two or three triggers you can identify — poor sleep, a social overload, a weather change, a skipped workout, a stressful phone call. Over a few months, patterns emerge and become controllable.
- Stay socially connected. Isolation amplifies pain perception in every study that has looked. A weekly coffee with one friend matters more than it sounds.
- Explain it once. To spouse, parent, boss — use the volume-knob analogy. It is faster and more credible than any list of symptoms.
Talking to Doctors Who Do Not Know This Language
Not every primary care physician has kept up with the 2017 IASP framework. If your clinician shrugs at "fibromyalgia" or suggests the pain is stress, you have options that do not require a fight.
First, bring the vocabulary. Say: "I think I have a nociplastic pain condition with central sensitization as the main mechanism. My CSI score is [number]." That sentence signals you have read the current literature and are asking for an appropriate workup, not a sympathetic nod.
Second, ask for a referral to a rheumatologist (to rule out inflammatory disease), a pain medicine specialist familiar with central sensitization, or a physiatrist who works with chronic pain. A multidisciplinary pain center is the best setting if one is available in your region.
Third, ask specifically about SNRIs, gabapentinoids, and LDN as first-line options, and ask to be referred to a physical therapist who understands graded aerobic pacing. If opioids are suggested for chronic management, push back gently: current specialty guidance is against them.
A positive side effect of using precise language is that it shifts the conversation from "are you really in pain" to "which mechanism-targeted therapy should we try first." That is a far more productive place to start.
Key Research Papers
- Woolf CJ. Central sensitization: implications for the diagnosis and treatment of pain. Pain. 2011;152(3 Suppl):S2–S15.
- Clauw DJ. Fibromyalgia: a clinical review. JAMA. 2014;311(15):1547–1555.
- Kosek E, et al. Chronic nociplastic pain affecting the musculoskeletal system: clinical criteria and grading system. Pain. 2021;162(11):2629–2634.
- Fitzcharles MA, Cohen SP, Clauw DJ, et al. Nociplastic pain: towards an understanding of prevalent pain conditions. Lancet. 2021;397(10289):2098–2110.
- Neblett R, Cohen H, Choi Y, et al. The Central Sensitization Inventory (CSI): establishing clinically significant values for identifying central sensitivity syndromes. Pain Pract. 2013;13(4):258–265.
- Harris RE, Sundgren PC, Craig AD, et al. Elevated insular glutamate in fibromyalgia is associated with experimental pain. Arthritis Rheum. 2009;60(10):3146–3152.
- Gracely RH, Petzke F, Wolf JM, Clauw DJ. Functional magnetic resonance imaging evidence of augmented pain processing in fibromyalgia. Arthritis Rheum. 2002;46(5):1333–1343.
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on central sensitization, nociplastic pain, and mechanism-targeted fibromyalgia therapies:
- Central sensitization and fibromyalgia
- Nociplastic pain — IASP framework
- Temporal summation and wind-up in fibromyalgia
- Conditioned pain modulation and descending inhibition
- Fibromyalgia functional MRI and the insula
- Glutamate, GABA, and magnetic resonance spectroscopy in fibromyalgia
- Central Sensitization Inventory validation studies
- SNRIs (duloxetine, milnacipran) in fibromyalgia
- Pregabalin and gabapentin in fibromyalgia
- Low-dose naltrexone in fibromyalgia
- Opioid-induced hyperalgesia
- Pain neuroscience education in chronic pain
Connections
- Fibromyalgia Overview
- Irritable Bowel Syndrome
- Low-Dose Naltrexone for Fibromyalgia
- Sleep and Non-Restorative Sleep
- ACR 2016 Diagnostic Criteria
- Exercise Pacing and Graded Movement
- Fibromyalgia and ME/CFS Overlap
- Nutrient Gaps in Fibromyalgia
- Small-Fiber Neuropathy Overlap
- Chronic Pain
- Migraine
- MCAS
- Chronic Fatigue Syndrome
- POTS
- Low-Dose Naltrexone
- Arthritis
- Central Sensitization and Nociplastic Pain
- Sleep Hygiene