POTS Subtypes: Hyperadrenergic, Neuropathic, and Hypovolemic
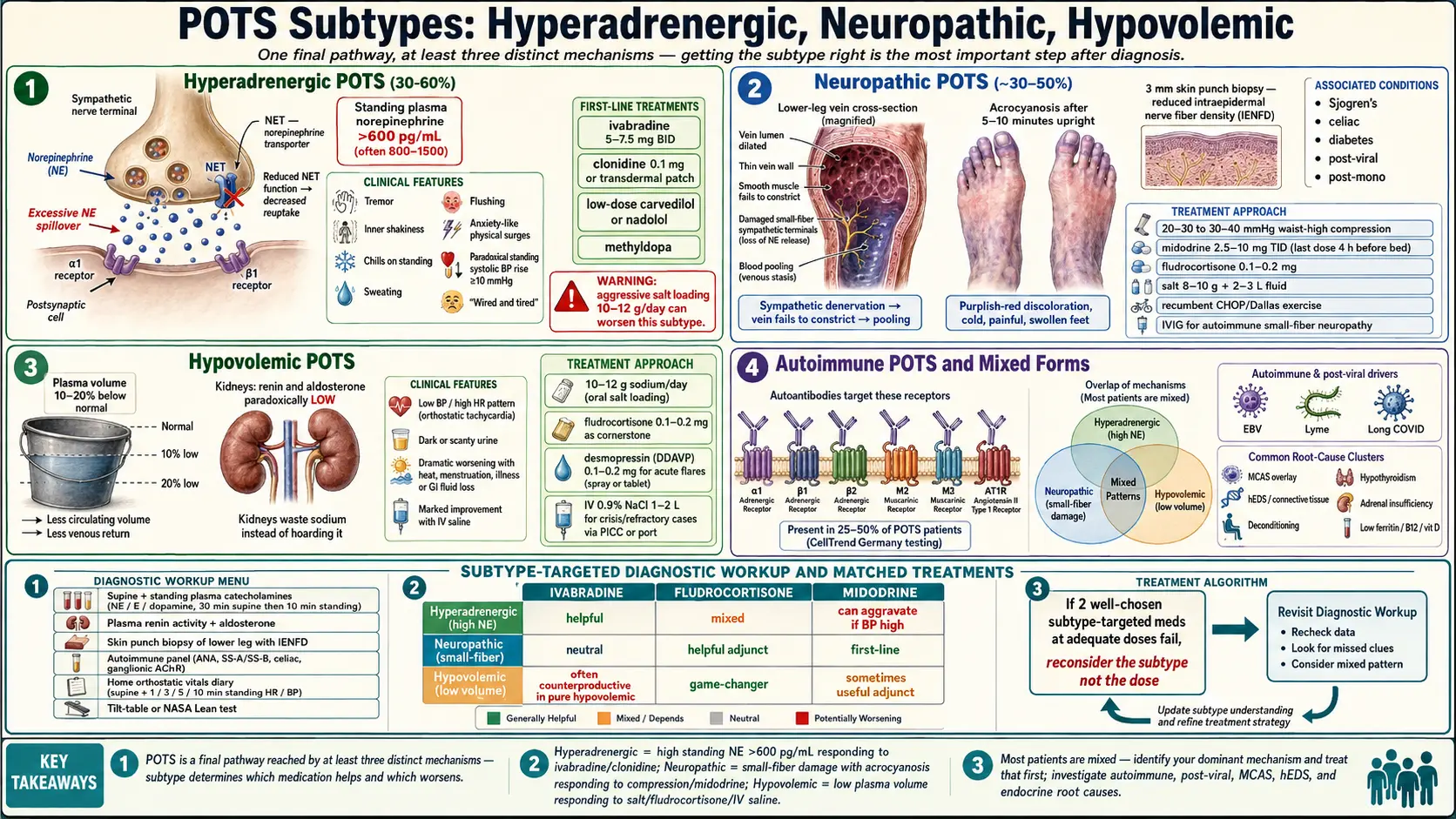
Table of Contents
- Why Subtypes Matter
- Hyperadrenergic POTS
- Neuropathic POTS
- Hypovolemic POTS
- Overlap and Mixed Forms
- How to Tell Which Subtype You Have
- Why the Treatment Difference Matters
- Underlying Drivers to Investigate for Each Subtype
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
1. Why Subtypes Matter
If you’ve been diagnosed with POTS and handed the same generic advice as everyone else in the clinic — drink more water, add salt, wear compression socks, take a beta blocker — and it’s not working, you are not failing. You may simply have been matched to the wrong protocol for your subtype.
POTS is not one disease. It’s a final common pathway — an abnormally large heart-rate response to standing — produced by at least three distinct mechanisms. Hyperadrenergic POTS is driven by excess sympathetic nerve output. Neuropathic POTS is driven by small-fiber nerve damage in the legs, causing blood to pool downward. Hypovolemic POTS is driven by low circulating plasma volume and broken volume regulation.
Each mechanism responds best to a different medication and a different lifestyle emphasis. A drug that calms one subtype can actively worsen another. That is why the cookie-cutter approach fails so many patients — and why figuring out your dominant subtype is arguably the most important step after diagnosis.
2. Hyperadrenergic POTS
Roughly 30–60% of POTS patients have a hyperadrenergic component. The defining lab feature: standing plasma norepinephrine above 600 pg/mL (often 800–1500+), compared with a normal supine baseline. The nervous system behaves as if it’s in chronic fight-or-flight.
Typical clinical picture:
- Tremor, inner shakiness, and chills on standing
- Sweating and flushing, sometimes with migraines
- Anxiety-like surges that are physical, not psychological, in origin
- Paradoxical hypertension on standing — systolic BP rises ≥10 mmHg upright (most POTS patients drop)
- Heart-pounding awareness, palpitations, feeling “wired and tired”
Underlying mechanisms include norepinephrine transporter (NET) deficiency, autoimmune activation of adrenergic receptors, and central sympathetic dysregulation. Many patients trace symptom onset to a viral illness, head injury, or significant stressor.
Best-Responding Medications
- Ivabradine (5–7.5 mg twice daily) — slows sinus node firing without touching blood pressure. First choice for many hyperadrenergic patients.
- Clonidine (0.1 mg once or twice daily, or 0.1 mg transdermal patch weekly) — central alpha-2 agonist, dials down sympathetic outflow directly. Also helpful for sleep.
- Low-dose beta blockers with vasodilating action — carvedilol (3.125–6.25 mg twice daily) or nadolol (10–20 mg daily). Better tolerated than propranolol for this subtype.
- Methyldopa in a subset whose daytime BP runs high.
What Often Backfires
Aggressive salt loading (10–12 g/day) and fludrocortisone — the standard playbook for the other two subtypes — can push standing blood pressure higher and intensify symptoms in hyperadrenergic patients. Start low and watch your standing BP before assuming salt is your friend.
3. Neuropathic POTS
In neuropathic POTS, the peripheral sympathetic nerves that normally squeeze the leg veins on standing are partially damaged. Blood pools in the lower body. Venous return drops. The heart compensates by racing.
Typical picture:
- Purplish-red discoloration of the feet and lower legs after a few minutes standing (acrocyanosis)
- Legs feel heavy and swollen; cold feet are common
- Symptoms much better when recumbent; worse in heat and after meals
- Heart rate rises sharply but blood pressure often stays near normal
- Often preceded by a viral infection, mononucleosis, or diagnosed alongside Sjögren’s, celiac disease, or diabetes
The gold-standard confirmation is a skin punch biopsy from the lower leg, examined for intraepidermal nerve fiber density. Reduced density confirms small-fiber neuropathy (SFN), which is present in an estimated 30–50% of POTS patients and is the signature of this subtype.
Best-Responding Medications and Interventions
- High-grade compression — 20–30 or 30–40 mmHg, waist-high or at minimum thigh-high. Knee-high alone is inadequate for true neuropathic pooling.
- Recumbent exercise (rowing, recumbent bike, swimming) — the Levine/Dallas protocol rebuilds cardiac and lower-body muscle pump capacity without triggering symptoms. Progress to upright work only after 2–3 months.
- Midodrine (2.5–10 mg up to three times daily, last dose no later than 4 hours before bedtime) — peripheral alpha-1 agonist, directly constricts the leaky leg vasculature.
- Fludrocortisone (0.1–0.2 mg daily) — expands plasma volume, increases vascular sensitivity to norepinephrine.
- Salt and fluid loading — 8–10 g sodium and 2–3 L water per day typically well-tolerated in this subtype.
If autoimmunity is driving the small-fiber damage (biopsy plus positive antibodies), some centers use IVIG in refractory cases.
4. Hypovolemic POTS
Here the problem is plumbing volume, not nerves or adrenaline. Formal testing (radiolabeled albumin or red-cell mass) often shows plasma volume deficits of 10–20%. The renin-aldosterone axis, which should correct the shortfall, paradoxically runs low instead of high — so the kidneys waste salt when they should be hoarding it.
Typical picture:
- Classic low blood pressure, high heart rate orthostatic pattern
- Dark or scanty urine despite reasonable fluid intake
- Marked worsening with heat, menstruation, illness, or any GI fluid loss
- Dramatic, immediate improvement from IV saline — a key clinical clue
- Salt cravings that feel physiological, not habit
Best-Responding Medications and Interventions
- High-dose sodium: 10–12 g/day through food, salt tablets, or electrolyte mixes (LMNT, Salt Stick, Vitassium).
- Fludrocortisone (0.1–0.2 mg daily) — the cornerstone pharmacologic treatment. Pair with potassium monitoring.
- Desmopressin (DDAVP 0.1–0.2 mg orally) for acute flares; reduces free-water loss and blunts orthostatic tachycardia for 4–6 hours. Watch sodium on chronic use.
- IV saline (1–2 L of 0.9% normal saline) for crisis, severe flares, or pre-event support. Some patients with refractory disease run weekly or twice-weekly infusions through a PICC or port — a last-resort measure but genuinely life-restoring for a subset.
Midodrine and beta blockers can be layered on but are rarely sufficient alone in this subtype. The priority is filling the tank.
5. Overlap and Mixed Forms
Here is the honest reality nobody puts on the intake sheet: most POTS patients have features of more than one subtype. A patient can have mild small-fiber neuropathy in the legs and a norepinephrine spike on standing. Another can be hypovolemic and have secondary hyperadrenergic compensation.
This is why rigid subtype-labeling is less useful than identifying your dominant mechanism — the one contributing most to your worst symptoms — and treating that first. Lab tests and orthostatic vitals point you toward a starting point. Your response to the first medication trial refines the picture.
A reasonable rule: if two well-chosen subtype-targeted medications at adequate doses don’t help, reconsider the subtype, not the dose. You may be treating the wrong mechanism.
6. How to Tell Which Subtype You Have
No single test nails it. The constellation does. These are the evaluations worth asking for:
- Supine and standing plasma catecholamines (norepinephrine, epinephrine, dopamine) — 30 minutes supine, then 10 minutes standing, drawn at each timepoint. A standing norepinephrine >600 pg/mL strongly supports hyperadrenergic POTS.
- Plasma renin activity and aldosterone — inappropriately low values despite orthostasis support hypovolemic POTS.
- Skin punch biopsy (lower leg, 3 mm) with intraepidermal nerve fiber density analysis — confirms or rules out small-fiber neuropathy and points toward neuropathic POTS.
- Comprehensive autoimmune panel — ANA, Sjögren’s SS-A/SS-B, celiac antibodies, ganglionic AChR antibody, and (where available) adrenergic and muscarinic receptor antibodies.
- Home orthostatic vitals diary — supine and 1-, 3-, 5-, and 10-minute standing HR and BP, morning and afternoon, for two weeks. Patterns emerge that single in-office readings miss.
- Formal tilt-table or NASA Lean Test — documents the orthostatic response in a standardized way. See Tilt-Table and NASA Lean Test.
Not every clinic will order all of these. Autonomic specialty centers (Mayo, Vanderbilt, Cleveland Clinic, Johns Hopkins, Dysautonomia International referral network) are the most likely to complete the full workup.
7. Why the Treatment Difference Matters
Concrete examples of why mismatched treatment fails:
- Ivabradine is often transformative in hyperadrenergic POTS because it directly slows the overactive sinus node. In pure neuropathic POTS it helps less — the tachycardia is a compensation for pooled blood, not a primary driver, and slowing the heart without addressing the pooling can worsen presyncope.
- Fludrocortisone is a game-changer in hypovolemic POTS and useful in neuropathic. In hyperadrenergic POTS it can push an already-elevated standing blood pressure into headache, chest tightness, and worse symptoms.
- Midodrine shines for neuropathic pooling and is reasonable in hypovolemic POTS as add-on. In hyperadrenergic POTS with supine hypertension it can trigger dangerous overnight BP spikes.
- Propranolol at low doses is often well-tolerated in hyperadrenergic POTS. In neuropathic POTS with already-marginal cardiac output on standing, even a small beta blockade can worsen fatigue.
- High-dose salt rescues hypovolemic patients and helps neuropathic patients; it aggravates a subset of hyperadrenergic patients.
For a deeper dive into individual drugs, doses, and titration, see the Medications Guide.
8. Underlying Drivers to Investigate for Each Subtype
Subtype identification isn’t the end of the workup. Each subtype has its own short list of root causes that, if found and treated, can shrink or resolve the POTS itself.
- Autoimmune drivers — adrenergic alpha-1, beta-1, beta-2 receptor antibodies; muscarinic M2, M3 antibodies; angiotensin II type 1 receptor (AT1R) antibodies. Strongest association with hyperadrenergic and some neuropathic cases. Testing is specialty-lab only (e.g., CellTrend in Germany). Positive results can justify IVIG or plasmapheresis trials.
- Post-infectious triggers — Epstein-Barr virus reactivation, Lyme and co-infections, post-viral syndromes (including long-haul post-viral illness after SARS-CoV-2 infection). Common prelude to neuropathic and mixed subtypes.
- Mast Cell Activation Syndrome (MCAS) — flushing, food/medication reactivity, and unexplained anaphylactoid episodes suggest MCAS overlay, especially in hyperadrenergic presentations. See the POTS-MCAS-EDS Triad.
- Ehlers-Danlos syndrome / hypermobility spectrum disorder — connective-tissue laxity means veins don’t hold their tone. Strongly associated with neuropathic and mixed POTS.
- Deconditioning — prolonged bedrest from any illness reduces plasma volume and cardiac chamber size. Contributes to all subtypes and is almost always partially reversible with graded recumbent exercise.
- Endocrine contributors — hypothyroidism, adrenal insufficiency, low ferritin, B12 deficiency, vitamin D deficiency. Cheap to screen, sometimes dramatically impactful to correct.
9. Key Research Papers
- Raj SR, et al. Postural Tachycardia Syndrome (POTS). Circulation. 2013.
- Sheldon RS, et al. 2015 Heart Rhythm Society expert consensus statement on the diagnosis and treatment of postural tachycardia syndrome, inappropriate sinus tachycardia, and vasovagal syncope. Heart Rhythm. 2015;12(6):e41-e63.
- Miller AJ, Raj SR. Pharmacotherapy for postural tachycardia syndrome. Auton Neurosci. 2018.
- Arnold AC, et al. Postural tachycardia syndrome: A brief review of etiology, diagnosis, and management. J Intern Med. 2019.
10. Research Papers
Curated PubMed topic searches — each link opens a live query so you always see the most recent literature.
- PubMed: Hyperadrenergic POTS and norepinephrine
- PubMed: Neuropathic POTS and small-fiber neuropathy
- PubMed: Hypovolemic POTS and plasma volume
- PubMed: POTS autoimmune antibodies
- PubMed: POTS, clonidine, and ivabradine
- PubMed: POTS subtype classification
- PubMed: POTS, fludrocortisone, and midodrine
- PubMed: POTS plasma volume and renin-aldosterone
Connections
- POTS Overview
- Medications Guide
- POTS/MCAS/EDS Triad
- Tilt Table Test
- Compression and Exercise Program
- Salt and Hydration Protocol
- Vagus Nerve and Autonomic Retraining
- LDN for POTS
- MCAS
- SIBO
- ME/CFS
- Fibromyalgia
- Ehlers-Danlos Syndrome
- Potassium
- Magnesium
- Fatigue
- Brain Fog
- Vestibular Migraine