Potassium – Essential Mineral for Human Health
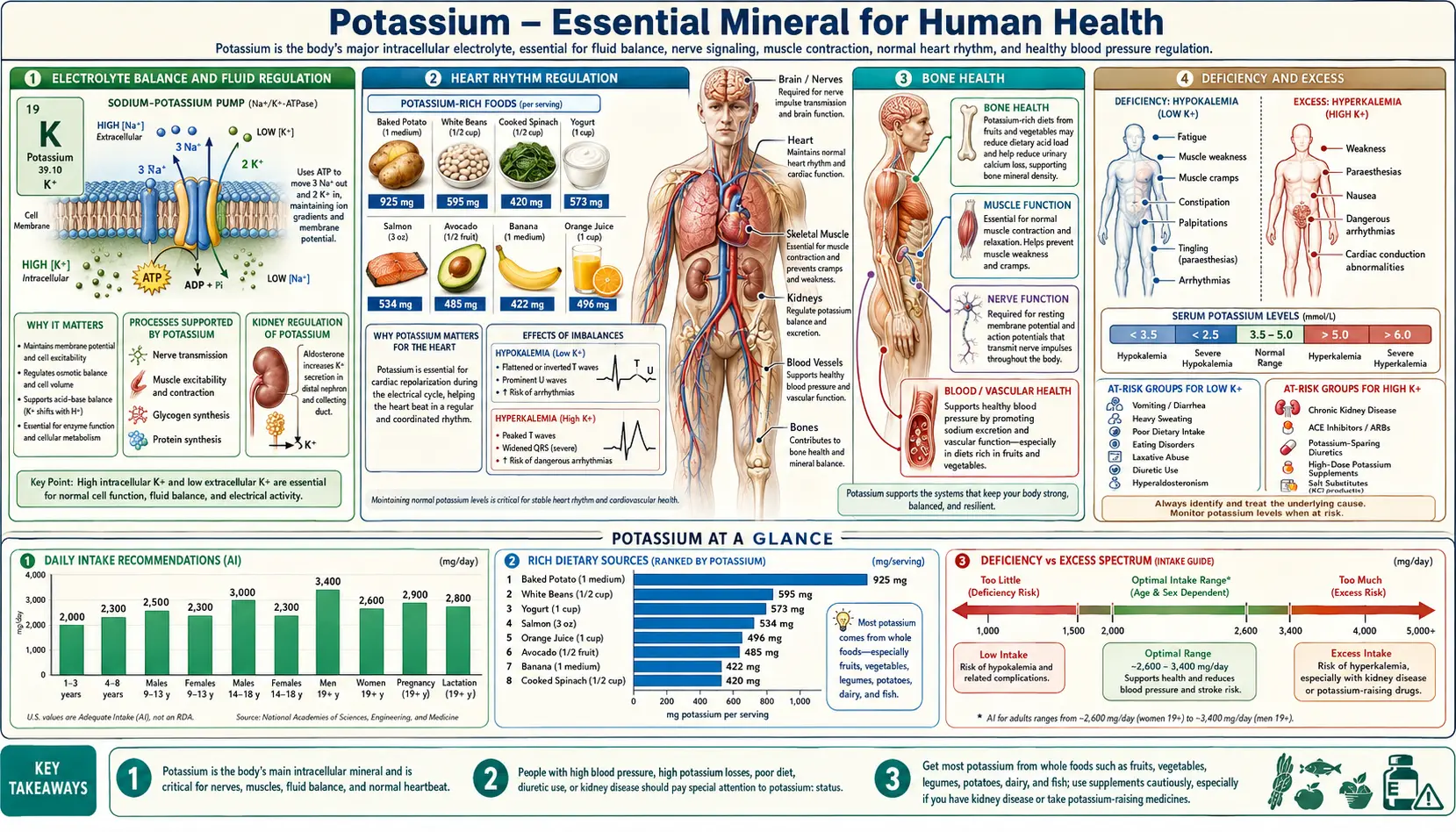
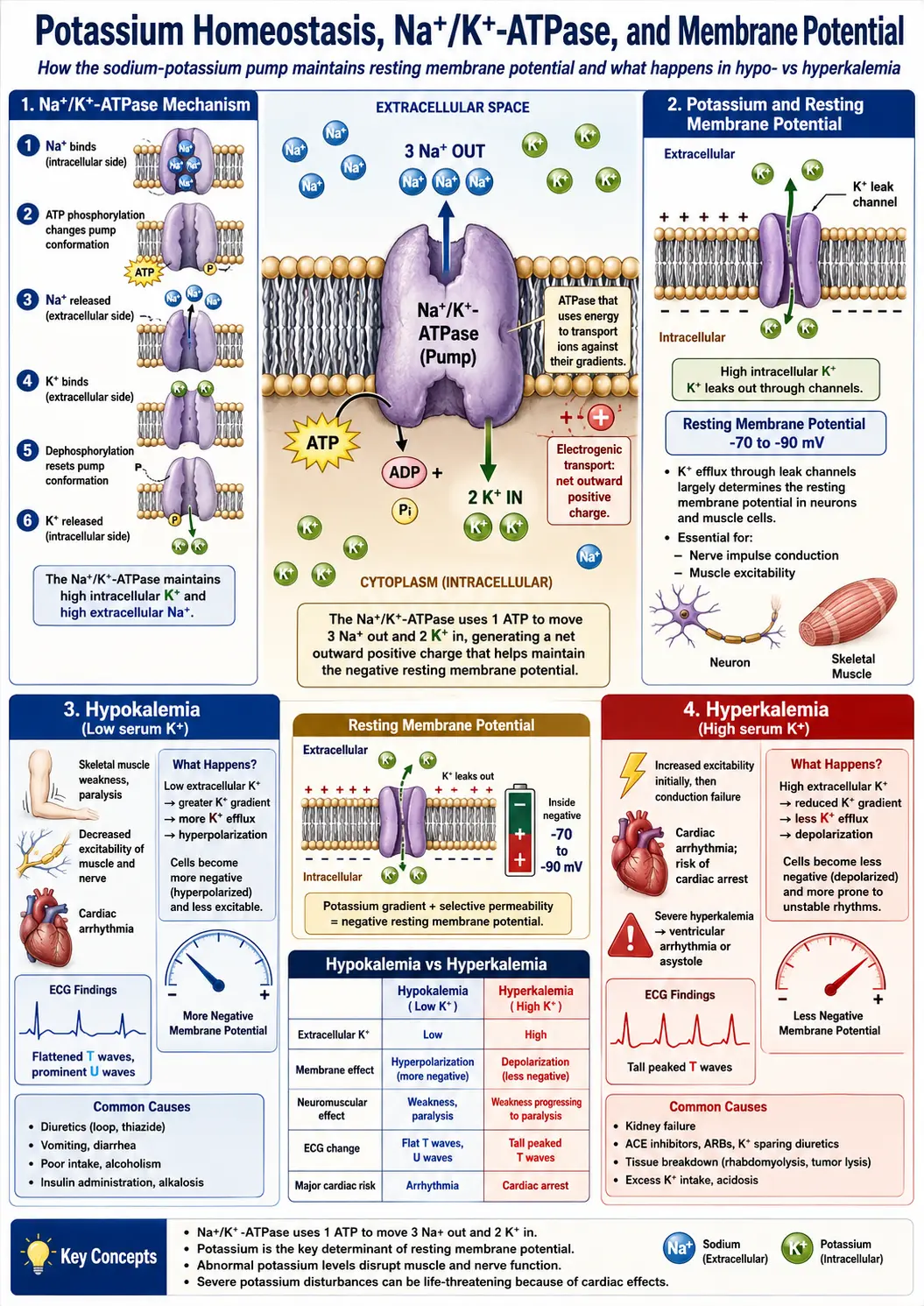
Table of Contents
- Potassium-Rich Foods (Ranked)
- Electrolyte Balance and Fluid Regulation
- Nerve Impulse Transmission and the Sodium-Potassium Pump
- Muscle Contraction
- Heart Rhythm Regulation
- Blood Pressure Control
- Kidney Function
- Acid-Base Balance
- Bone Health
- Metabolic Functions
- Dietary Sources and Recommendations
- Deficiency and Excess
- Connections
Potassium (chemical symbol K, from the Latin kalium) is the most abundant intracellular cation in the human body. Approximately 98% of total body potassium resides inside cells, with the remaining 2% found in the extracellular fluid. This steep concentration gradient across cell membranes is fundamental to cellular function, electrical signaling, and life itself. The adequate intake (AI) for potassium in adults is 2,600 mg/day for women and 3,400 mg/day for men, though many populations fall well short of these recommendations.
Electrolyte Balance and Fluid Regulation
Potassium is one of the body's principal electrolytes, working in concert with sodium, chloride, and bicarbonate to maintain fluid homeostasis across all tissue compartments.
- Intracellular fluid volume – Potassium is the dominant osmotic force inside cells. Changes in intracellular potassium concentration directly alter cell volume, which in turn affects enzyme activity, protein synthesis, and metabolic rate.
- Extracellular fluid balance – Although extracellular potassium concentrations are low (3.5–5.0 mEq/L), even small deviations can have profound physiological consequences, particularly on cardiac and neuromuscular function.
- Osmotic equilibrium – Potassium and sodium together determine the osmolarity of body fluids. The distribution of water between intracellular and extracellular compartments is governed by the relative concentrations of these two ions.
- Transcellular fluid shifts – Hormones such as insulin and catecholamines, as well as changes in acid-base status, can rapidly shift potassium between intracellular and extracellular compartments, providing a rapid buffering mechanism for serum potassium levels.
Nerve Impulse Transmission and the Sodium-Potassium Pump
The transmission of electrical signals throughout the nervous system depends critically on the potassium gradient across neuronal membranes.
The Na+/K+-ATPase Pump
The sodium-potassium pump (Na+/K+-ATPase) is a membrane-spanning enzyme found in virtually all animal cells. It actively transports three sodium ions out of the cell and two potassium ions into the cell for each molecule of ATP hydrolyzed. This pump consumes approximately 20–40% of the resting energy expenditure of most cells and is responsible for establishing and maintaining the electrochemical gradient that underlies all electrical signaling.
- Resting membrane potential – The activity of the Na+/K+-ATPase, combined with the selective permeability of the cell membrane to potassium through leak channels, establishes a resting membrane potential of approximately −70 to −90 millivolts in most excitable cells.
- Action potential generation – Nerve impulses are generated when sodium channels open, causing depolarization, followed by potassium channel opening for repolarization. Without adequate potassium, repolarization is impaired and neurons cannot reset for subsequent signaling.
- Signal propagation velocity – The speed and reliability of nerve signal transmission depend on the rapid cycling of depolarization and repolarization, both of which require proper potassium gradients.
Neuromuscular Junction
At the neuromuscular junction, potassium plays a role in terminating the action potential at the motor end plate, enabling the muscle fiber to relax and prepare for the next contraction cycle. Disturbances in extracellular potassium levels can alter neuromuscular transmission, leading to weakness, cramping, or paralysis.
Muscle Contraction
Potassium is indispensable for normal function of all three types of muscle tissue: skeletal, smooth, and cardiac.
- Skeletal muscle function – Skeletal muscle contains approximately 70% of total body potassium. During intense exercise, potassium efflux from contracting muscle fibers can transiently raise local extracellular potassium concentrations, contributing to muscle fatigue. Recovery requires reuptake of potassium via the Na+/K+-ATPase.
- Smooth muscle tone – Potassium channels in vascular smooth muscle regulate arterial tone. Opening of potassium channels causes hyperpolarization and vasorelaxation, while closure promotes depolarization and vasoconstriction. This mechanism is central to blood pressure regulation.
- Excitation-contraction coupling – The sequence from electrical excitation to mechanical contraction depends on proper membrane potential, which in turn depends on potassium homeostasis. Both hypokalemia and hyperkalemia disrupt this coupling and can cause muscle weakness or paralysis.
Heart Rhythm Regulation
The heart is exquisitely sensitive to potassium levels. The cardiac action potential, which governs the orderly sequence of atrial and ventricular contraction, depends on precisely regulated potassium currents at multiple phases.
- Pacemaker activity – Specialized pacemaker cells in the sinoatrial node rely on potassium currents (particularly the "funny current" and delayed rectifier currents) to generate the rhythmic depolarizations that set the heart rate.
- Repolarization – Potassium efflux through voltage-gated potassium channels (IKr, IKs, IK1) is responsible for repolarizing the cardiac myocyte after each contraction, resetting the cell for the next beat.
- Arrhythmia risk – Hypokalemia (serum K+ below 3.5 mEq/L) prolongs repolarization, predisposing to dangerous arrhythmias including torsades de pointes and ventricular fibrillation. Hyperkalemia (serum K+ above 5.0 mEq/L) can cause conduction delays, bradycardia, and ultimately cardiac arrest.
For more detailed discussion, see Potassium and Heart Rhythm.
Blood Pressure Control
Potassium plays a central role in blood pressure regulation through multiple complementary mechanisms.
- Natriuresis – Potassium promotes renal excretion of sodium. Higher potassium intake reduces sodium reabsorption in the renal tubules, thereby lowering extracellular fluid volume and blood pressure.
- Vasodilation – Potassium directly relaxes vascular smooth muscle by activating potassium channels and stimulating the sodium-potassium pump in endothelial cells, promoting the release of nitric oxide.
- Sympathetic nervous system modulation – Adequate potassium intake attenuates sympathetic nervous system activity, reducing catecholamine-mediated vasoconstriction and cardiac stimulation.
- Epidemiological evidence – Population studies consistently demonstrate an inverse relationship between dietary potassium intake and blood pressure. The DASH (Dietary Approaches to Stop Hypertension) diet, which is rich in potassium from fruits and vegetables, has been shown to lower systolic blood pressure by 8–14 mmHg.
For more detailed discussion, see Potassium and Blood Pressure Regulation.
Kidney Function
The kidneys are the primary regulators of potassium homeostasis, adjusting urinary potassium excretion to match dietary intake and maintain serum levels within the narrow physiological range.
- Glomerular filtration – Potassium is freely filtered at the glomerulus. Approximately 600–700 mEq of potassium is filtered daily, of which about 90% is reabsorbed in the proximal tubule and loop of Henle.
- Distal secretion – Fine-tuning of potassium excretion occurs in the distal convoluted tubule and cortical collecting duct, where principal cells secrete potassium into the tubular lumen under the regulation of aldosterone.
- Aldosterone regulation – Rising serum potassium stimulates aldosterone release from the adrenal cortex. Aldosterone increases the number and activity of Na+/K+-ATPase pumps and epithelial sodium channels (ENaC) in the collecting duct, promoting potassium secretion and sodium reabsorption.
- Renal compensation – In chronic kidney disease, the kidneys' ability to excrete potassium diminishes, making patients increasingly susceptible to hyperkalemia, particularly when consuming potassium-rich foods or taking potassium-sparing medications.
Acid-Base Balance
Potassium and hydrogen ion (H+) homeostasis are intimately linked, and disturbances in one frequently produce disturbances in the other.
- Transcellular exchange – In acidosis, excess hydrogen ions move into cells, displacing potassium into the extracellular fluid. In alkalosis, the reverse occurs: hydrogen ions leave cells, and potassium moves inward. As a general rule, a 0.1-unit change in arterial pH produces an opposite change in serum potassium of approximately 0.6 mEq/L.
- Renal acid excretion – In the distal nephron, potassium secretion and hydrogen ion secretion are partially competitive processes. Hypokalemia stimulates renal hydrogen ion secretion and ammoniagenesis, contributing to metabolic alkalosis. Conversely, hyperkalemia inhibits ammoniagenesis and can worsen metabolic acidosis.
- Clinical significance – The interplay between potassium and acid-base status has important clinical implications. For example, treating hypokalemia is often essential for correcting concurrent metabolic alkalosis, and correction of diabetic ketoacidosis requires careful potassium monitoring and replacement.
Bone Health
Emerging evidence suggests that potassium contributes to the maintenance of skeletal health through several mechanisms.
- Acid-base buffering and calcium conservation – Diets high in potassium-rich fruits and vegetables generate alkaline precursors (bicarbonate and citrate) that buffer metabolic acid. This reduces the need for bone to serve as an alkaline reservoir, thereby limiting calcium mobilization from the skeleton.
- Reduced urinary calcium loss – Higher potassium intake is associated with lower urinary calcium excretion. This effect is partly mediated by the alkaline load provided by potassium salts and partly by direct effects on renal calcium handling.
- Epidemiological associations – Several observational studies have reported positive associations between dietary potassium intake and bone mineral density, particularly in postmenopausal women. Intervention trials with potassium citrate supplementation have shown reductions in markers of bone resorption.
Metabolic Functions
Beyond its roles in electrical signaling and fluid balance, potassium participates in a range of metabolic processes essential for cellular health.
Carbohydrate Metabolism
- Insulin secretion – Potassium is required for normal insulin release from pancreatic beta cells. Hypokalemia impairs insulin secretion and has been associated with glucose intolerance.
- Glycogen synthesis – Potassium is co-transported with glucose into cells during insulin-mediated glucose uptake and is incorporated into glycogen stores. Each gram of glycogen is stored with approximately 0.36 mEq of potassium.
Protein Synthesis
- Intracellular environment – Adequate intracellular potassium concentration is necessary for ribosomal function and protein synthesis. Potassium depletion reduces the rate of protein synthesis and promotes protein catabolism.
- Cell growth – Potassium influx is one of the early events in the cell cycle following mitogenic stimulation. Potassium channel activity has been linked to cell proliferation in both normal and neoplastic tissues.
Enzymatic Reactions
- Cofactor role – Potassium serves as a cofactor for pyruvate kinase, a key enzyme in glycolysis. It is also required for optimal activity of several other enzymes involved in energy metabolism and biosynthetic pathways.
Dietary Sources and Recommendations
Potassium is widely distributed in foods, with the richest sources being fruits, vegetables, legumes, dairy products, and fish. Notable high-potassium foods include bananas, oranges, potatoes, sweet potatoes, spinach, tomatoes, avocados, beans, lentils, yogurt, and salmon. Despite the wide availability of potassium in the food supply, many people consume less than the adequate intake, largely due to diets low in fruits and vegetables and high in processed foods, which tend to be high in sodium and low in potassium.
For a ranked, side-by-side comparison of common potassium-rich whole foods (mg per 100 g and calories per 100 g, excluding leafy vegetables, edamame, baked white potato, and avocado), see Potassium-Rich Foods.
Deficiency and Excess
Hypokalemia
Hypokalemia (serum potassium below 3.5 mEq/L) can result from inadequate dietary intake, gastrointestinal losses (vomiting, diarrhea), renal losses (diuretic use, hyperaldosteronism), or transcellular shifts. Symptoms range from mild fatigue and muscle cramps to severe weakness, ileus, rhabdomyolysis, and life-threatening cardiac arrhythmias.
Hyperkalemia
Hyperkalemia (serum potassium above 5.0 mEq/L) most commonly occurs in the setting of renal insufficiency, potassium-sparing diuretics, ACE inhibitors, angiotensin receptor blockers, or excessive supplementation. Mild hyperkalemia may be asymptomatic, but severe elevations can cause muscle weakness, paresthesias, and cardiac conduction abnormalities that may progress to ventricular fibrillation and cardiac arrest.
Connections
- Potassium Benefits Deep Dive
- Potassium-Rich Foods
- Potassium and Heart Rhythm
- Potassium and Blood Pressure
- Potassium and Muscle Function
- Magnesium
- Calcium
- Calcium and Muscle Function — excitation-contraction coupling, cramps, tetany, and the calcium-magnesium balance for cardiac and skeletal muscle.
- Hypertension
- Arrhythmia
- Heart Failure
- Kidney Disease
- Bananas
- Avocado
- Celery Juice and Blood Pressure
- Phosphorus
- Spinach
- Sweet Potatoes
- Salt
- Comprehensive Metabolic Panel
- Stroke