The POTS/MCAS/EDS Triad
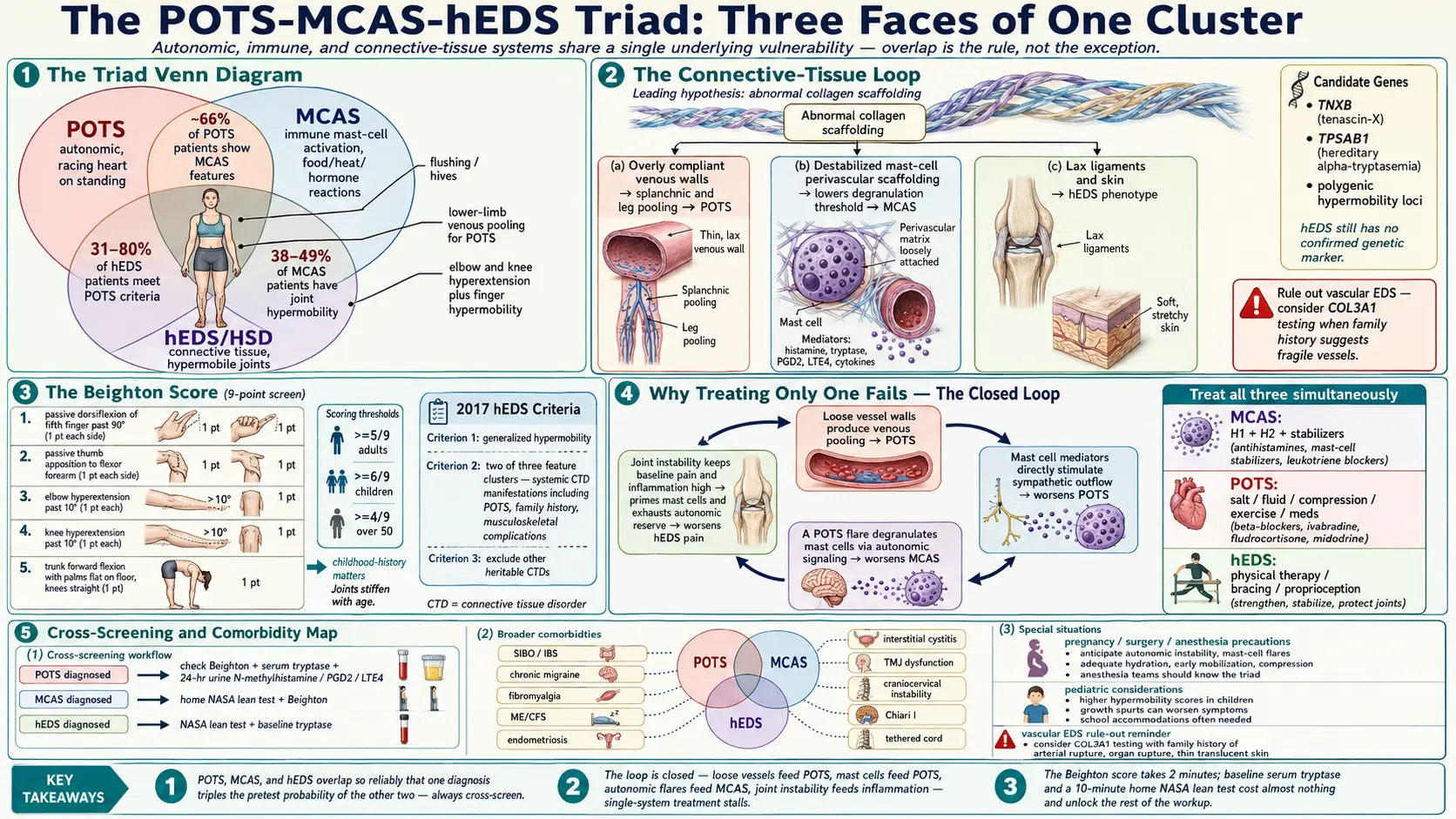
Table of Contents
- The Triad at a Glance
- Why They Cluster
- Hypermobility Spectrum Disorder (HSD) vs hEDS
- The Beighton Score (9-point scale)
- The hEDS 2017 Diagnostic Criteria
- MCAS Within the Triad
- When to Screen for the Other Two
- Shared Comorbidities Beyond the Core 3
- Why Treating Only One Fails
- Integrated Treatment Strategy
- Finding Providers Who Understand the Triad
- Pregnancy, Surgery, and Anesthesia in the Triad
- Children and Adolescents
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
1. The Triad at a Glance
If you’ve been told that your racing heart, your food reactions, and your bendy joints “can’t possibly all be connected,” you have been told something that contradicts twenty years of clinical research. They are connected. They are connected so reliably that specialists now talk about them as a single clustered phenomenon with three faces.
The three faces are:
- POTS — Postural Orthostatic Tachycardia Syndrome. A disorder of the autonomic nervous system in which the heart rate spikes 30+ beats per minute on standing, often with lightheadedness, brain fog, and exercise intolerance.
- MCAS — Mast Cell Activation Syndrome. A disorder of the immune system’s mast cells, which release histamine, tryptase, and other mediators inappropriately in response to food, heat, stress, hormones, or nothing identifiable at all.
- hEDS / HSD — Hypermobile Ehlers-Danlos Syndrome and Hypermobility Spectrum Disorder. Heritable disorders of connective tissue in which collagen-rich structures (joints, ligaments, skin, blood vessel walls, gut wall) are structurally fragile and overly compliant.
The overlap is not rare. It is the rule.
- 31–80% of hEDS patients meet criteria for POTS.
- ~66% of POTS patients have some MCAS features.
- 38–49% of MCAS patients have joint hypermobility.
Draw a Venn diagram of the three and the shared middle is enormous. If you have one confirmed, the pretest probability of having another is far above baseline — and high enough that screening is worth your time.
2. Why They Cluster
No one has proven a single unifying cause, but the leading theory is mechanistically satisfying. A connective tissue defect affects blood vessel walls — the veins of the lower body become abnormally compliant, blood pools on standing, cardiac return drops, and the sympathetic nervous system compensates with a tachycardia (POTS). The same connective tissue abnormality is thought to disturb the scaffolding around mast cells at every tissue interface, lowering the threshold for degranulation (MCAS). And the hypermobile joints are the most visible expression of the underlying collagen problem.
Genetic hypotheses under active study include TNXB variants (tenascin-X deficiency produces an EDS-like phenotype), TPSAB1 duplications (hereditary alpha-tryptasemia, associated with elevated baseline tryptase and a striking overlap with connective tissue symptoms), and a set of polygenic hypermobility loci. No single confirmed gene explains the whole triad, and hEDS itself remains a clinical diagnosis without a confirmed genetic marker.
The takeaway for patients: the mechanism is real, it is being actively researched, and the absence of a single smoking-gun gene does not make your cluster imaginary.
3. Hypermobility Spectrum Disorder (HSD) vs hEDS
Two labels, one spectrum.
HSD describes people who have symptomatic joint hypermobility — pain, subluxations, fatigue — but who do not meet every box of the 2017 hEDS criteria. hEDS is the label for those who do meet all three criterion sets. Functionally, the two groups look and behave very similarly, and many clinicians treat them identically. The label matters for insurance, for research enrollment, and for family counseling, but it does not meaningfully change management.
A crucial caveat: other EDS subtypes exist and are not part of this triad in the same way. Classical EDS (COL5A1/COL5A2), vascular EDS (COL3A1), dermatosparaxis, kyphoscoliotic, and rarer subtypes each have a confirmed genetic test and a different management picture. Vascular EDS in particular is life-threatening and must be ruled out when family history or physical findings suggest it. If anything about your presentation is atypical — translucent skin, fragile vessels, a parent who died young of arterial rupture — insist on a full genetic panel before settling on an hEDS label.
4. The Beighton Score (9-point scale)
The Beighton score is a fast bedside screen for generalized joint hypermobility. You can self-administer it in about two minutes, or have a partner help. Score one point for each of the following:
- Passive dorsiflexion of the 5th finger past 90° (1 point each side — max 2)
- Passive apposition of the thumb to the flexor forearm (1 point each side — max 2)
- Hyperextension of the elbow past 10° (1 point each side — max 2)
- Hyperextension of the knee past 10° (1 point each side — max 2)
- Forward flexion of the trunk, palms flat on the floor with knees straight (1 point — max 1)
Scoring: ≥5/9 in adults, ≥6/9 in children, and ≥4/9 in adults over 50 suggests generalized joint hypermobility. Scores tend to decrease with age as tissues stiffen, so childhood history matters. Patients whose joints were dramatically loose at age 10 but have stiffened in adulthood still carry the connective tissue pattern.
A Beighton score alone does not diagnose hEDS — it is one of the three criterion sets (see below). But a positive Beighton in a patient with POTS or MCAS is one of the strongest reasons to pursue the full workup.
5. The hEDS 2017 Diagnostic Criteria (simplified)
Formal hEDS diagnosis requires all three of the following criteria:
Criterion 1 — Generalized joint hypermobility
Beighton score as above, or historical evidence of hypermobility when the score was higher.
Criterion 2 — Two of three feature clusters
- Feature A: Systemic manifestations of a generalized connective tissue disorder. Soft or velvety skin, mild skin hyperextensibility, unexplained striae, bilateral piezogenic papules, recurrent or multiple abdominal hernias, atrophic scarring, pelvic floor/rectal/uterine prolapse without known risk factors, dental crowding and high/narrow palate, arachnodactyly, arm span-to-height ratio ≥1.05, mitral valve prolapse, aortic root dilation, and — crucially for the triad — POTS.
- Feature B: Positive family history — one or more first-degree relatives independently meeting the criteria.
- Feature C: Musculoskeletal complications — chronic musculoskeletal pain in two or more limbs for at least 3 months, chronic widespread pain for at least 3 months, or recurrent joint dislocations/subluxations or frank joint instability without trauma.
At least two of the three feature clusters must be present.
Criterion 3 — Exclusion of alternative diagnoses
Other EDS subtypes (classical, vascular, dermatosparaxis, kyphoscoliotic, etc.), Marfan syndrome, Loeys-Dietz, osteogenesis imperfecta, and other heritable connective tissue disorders must be ruled out — by clinical features and, when indicated, a genetic panel.
6. MCAS Within the Triad
Mast cells sit at every tissue interface where the body meets the outside world — skin, gut lining, airways, genitourinary tract — and they communicate constantly with autonomic nerves and connective tissue. When the connective tissue scaffolding is abnormal and the autonomic nervous system is dysregulated, mast cells appear to lose their usual regulatory restraint.
Common MCAS symptoms in the triad:
- Flushing, especially after meals, heat, stress, or exercise
- Hives, dermatographism, chronic itch
- GI reactions — nausea, cramping, diarrhea, bloating after specific foods or “random” triggers
- Anaphylaxis-like episodes without a classic IgE-mediated allergen
- Food-dependent exercise-induced anaphylaxis — fine at rest, fine eating alone, but a reaction when the two combine
- Chronic sinus congestion, throat clearing, reactive airways
- Menstrual-cycle-linked symptom flares (estrogen primes mast cells)
Screening tests
- Serum tryptase — drawn at baseline and, ideally, during a symptomatic flare. A rise of 20% + 2 ng/mL above baseline during a flare is one of the validated MCAS criteria.
- 24-hour urine N-methylhistamine, prostaglandin D2 (PGD2), and leukotriene E4 (LTE4) — chilled collection, with the patient off antihistamines where safe.
Cross-link: MCAS Testing Guide.
7. When to Screen for the Other Two
Diagnosed with POTS?
Check your own Beighton score in the next two minutes — it’s free and takes less time than making coffee. If you score ≥5, request a referral to an EDS-aware geneticist or rheumatologist. Also screen for MCAS: baseline serum tryptase, and a 24-hr urine N-methylhistamine (plus PGD2 and LTE4 if available).
Diagnosed with MCAS?
Do a home NASA lean test (see Tilt Table and NASA Lean Test). A heart-rate rise of ≥30 bpm within 10 minutes of standing is a strong POTS signal and justifies formal workup. Self-check your Beighton score the same day.
Diagnosed with hEDS or HSD?
Run a NASA lean test at home. If you have any MCAS-type symptoms — flushing, food reactions, chronic rashes, migraines, unexplained anaphylaxis — pursue the MCAS workup above. Even asymptomatic hEDS patients are worth a baseline tryptase because it establishes your individual reference point for the future.
8. Shared Comorbidities Beyond the Core 3
The triad is often the center of a larger constellation. Conditions that cluster with one or more of the three at rates well above baseline include:
- SIBO and IBS — autonomic dysmotility plus connective tissue laxity of the gut wall. See SIBO.
- Chronic migraine and new daily persistent headache
- Fibromyalgia and chronic widespread pain
- ME/CFS (myalgic encephalomyelitis / chronic fatigue syndrome)
- Endometriosis and adenomyosis
- Interstitial cystitis / bladder pain syndrome
- TMJ dysfunction and jaw subluxation
- Cervicocranial instability and craniocervical junction disorders
- Tethered cord syndrome
- Chiari I malformation
Not every patient has every one. But if your chart is already three of these, the clinician who keeps calling them coincidences is not reading the pattern.
9. Why Treating Only One Fails
If you have all three and your care team addresses only one, you will stall.
- Treat only POTS and you miss the mast cell activation that is triggering autonomic flares every time you eat, menstruate, or catch a virus.
- Treat only MCAS and you still crash every time you stand up, because the hemodynamic problem never got addressed.
- Manage only hypermobility with PT and bracing and you leave the autonomic and immune layers untouched, so every PT session leaves you horizontal for three days.
The three feed each other in a closed loop. Loose vessel walls produce venous pooling, which produces POTS. Mast cell mediators directly stimulate sympathetic outflow, worsening POTS. A POTS flare degranulates mast cells via autonomic signaling, which worsens MCAS. Mechanical joint instability keeps baseline pain and inflammation high, which primes mast cells and exhausts the autonomic reserve. None of the loops close unless all three are being addressed.
10. Integrated Treatment Strategy
Clinicians experienced in the triad tend to sequence interventions roughly in this order:
Step 1 — Stabilize mast cells first
Reducing the baseline trigger load pays dividends on everything else. An H1 + H2 blocker stack (a second-generation H1 such as cetirizine or fexofenadine plus an H2 such as famotidine), often with a mast cell stabilizer (cromolyn sodium before meals, quercetin, or luteolin), lowers the background hum enough that the POTS interventions actually work.
Step 2 — Fluid and salt loading
2–3 liters of fluid and 8–10 g of sodium daily for most POTS patients (supervised). See Salt and Hydration Protocol.
Step 3 — Compression and recumbent exercise
Waist-high 20–30 mmHg compression during upright time. Recumbent and water-based reconditioning (the Levine / Dallas protocol). See Compression and Exercise Program.
Step 4 — Subtype-targeted medications
Beta blockers, ivabradine, midodrine, fludrocortisone, pyridostigmine — selection depends on the POTS subtype (hyperadrenergic, neuropathic, hypovolemic). See Medications Guide and Subtypes.
Step 5 — Joint protection
PT focused on proprioception and joint stabilization, not on stretching. Bracing during flares. Avoid end-range hyperextension. Isometric and closed-chain strengthening build the ligamentous support the collagen cannot provide on its own.
Step 6 — Address the gut overlap
Triad patients with bloating, reflux, or post-prandial symptoms should get a SIBO workup. See SIBO.
11. Finding Providers Who Understand the Triad
There are not enough of them. Plan accordingly.
- The Ehlers-Danlos Society Healthcare Provider Directory — the most comprehensive international listing of EDS-aware clinicians.
- Dysautonomia International physician list — POTS and autonomic-focused providers, searchable by state.
- The Mast Cell Disease Society — directory of allergists and immunologists familiar with MCAS.
- The Ehlers-Danlos Society “zebra network” — patient-maintained peer recommendations.
Virtual consults are common given specialist scarcity; many of the leading clinicians see patients from across the country by telehealth. Expect waitlists of 3 to 12 months for top-tier specialists. In the meantime, a well-informed primary care doctor paired with PT, a dietitian, and a sympathetic allergist can cover most of the early management.
12. Pregnancy, Surgery, and Anesthesia in the Triad
This is the section to read twice and save.
Pregnancy
Hormonal shifts, plasma volume changes, and mechanical loading can trigger or dramatically worsen all three conditions. POTS frequently unmasks in pregnancy or postpartum. MCAS can flare spectacularly with estrogen surges. Joint instability worsens with relaxin-driven ligament softening. Pregnancies in triad patients are best co-managed with a maternal-fetal medicine specialist who knows the pattern.
Surgery
hEDS tissue holds sutures poorly and bleeds more than expected. POTS worsens post-operatively from bed rest, fluid shifts, and pain. MCAS patients can react to anesthetic agents, preservatives, latex, adhesives, and IV contrast.
Anesthesia
Agents generally to avoid or use with caution: succinylcholine, morphine, codeine, meperidine, atracurium, and many neuromuscular blockers with histamine-releasing profiles. Agents generally better tolerated: fentanyl, propofol, rocuronium, midazolam, and dexmedetomidine. Local anesthetics often need to be dosed higher in hEDS because they appear to be cleared or distributed differently in hypermobile tissue — bring this up with the anesthesiologist preoperatively.
Pre-op letters to the anesthesia team are critical. The EDS Society publishes template pre-op letters that list the patient’s diagnoses, known triggers, safer agent lists, and positioning precautions. Bring one to every surgical consultation.
13. Children and Adolescents
In children, hypermobility typically shows up first — often years before the autonomic and mast cell pieces declare themselves. Parents and pediatricians should track:
- Chronic joint pain, especially knees and ankles, that comes and goes
- Easy bruising without a bleeding disorder workup abnormality
- Chronic headaches, especially orthostatic or exertional
- Fainting, near-fainting, or dizziness on standing in hot showers, in line, or first thing in the morning
- “Picky eating” that turns out to be food reactions — flushing, tummy aches, throat tickling, hives
- Recurrent hernias, rectal prolapse, or dental crowding in a flexible child
Early identification lets families front-load joint protection, proprioceptive strengthening, salt and hydration habits, and trigger awareness before the crash years hit — which for many patients is the late teens and early twenties.
14. Key Research Papers
- Malfait F, et al. The 2017 international classification of the Ehlers-Danlos syndromes. Am J Med Genet. 2017;175C(1):8–26.
- Hakim A, et al. Cardiovascular autonomic dysfunction in Ehlers-Danlos syndrome / hypermobility spectrum disorders. Am J Med Genet. 2017.
- Seneviratne SL, et al. Mast cell disorders in Ehlers-Danlos syndrome. Am J Med Genet. 2017.
- Arnold AC, et al. Postural tachycardia syndrome — diagnosis, physiology, and prognosis. J Intern Med. 2019.
15. Research Papers
Curated PubMed topic searches for the triad. Each link opens a live PubMed query so you always see the most current studies.
- PubMed: POTS and EDS hypermobility
- PubMed: MCAS and EDS
- PubMed: Hypermobility and autonomic dysfunction
- PubMed: Beighton score and hypermobility
- PubMed: Mast cell activation and hypermobility
- PubMed: POTS, MCAS, and Ehlers-Danlos
- PubMed: Hereditary alpha-tryptasemia and TPSAB1
- PubMed: hEDS 2017 diagnostic criteria
Connections
- POTS Overview
- MCAS
- SIBO
- POTS Subtypes
- Medications Guide
- Compression and Exercise
- Tilt Table Test
- Salt and Hydration
- Vagus Nerve and Autonomic Retraining
- LDN for POTS
- Ehlers-Danlos Syndrome
- Fibromyalgia
- ME/CFS
- Alpha-Gal Syndrome
- MCAS Testing Guide
- Brain Fog
- Lightheadedness
- Fibromyalgia and ME CFS Overlap