LDN, Abilify & Experimental Therapies for ME/CFS
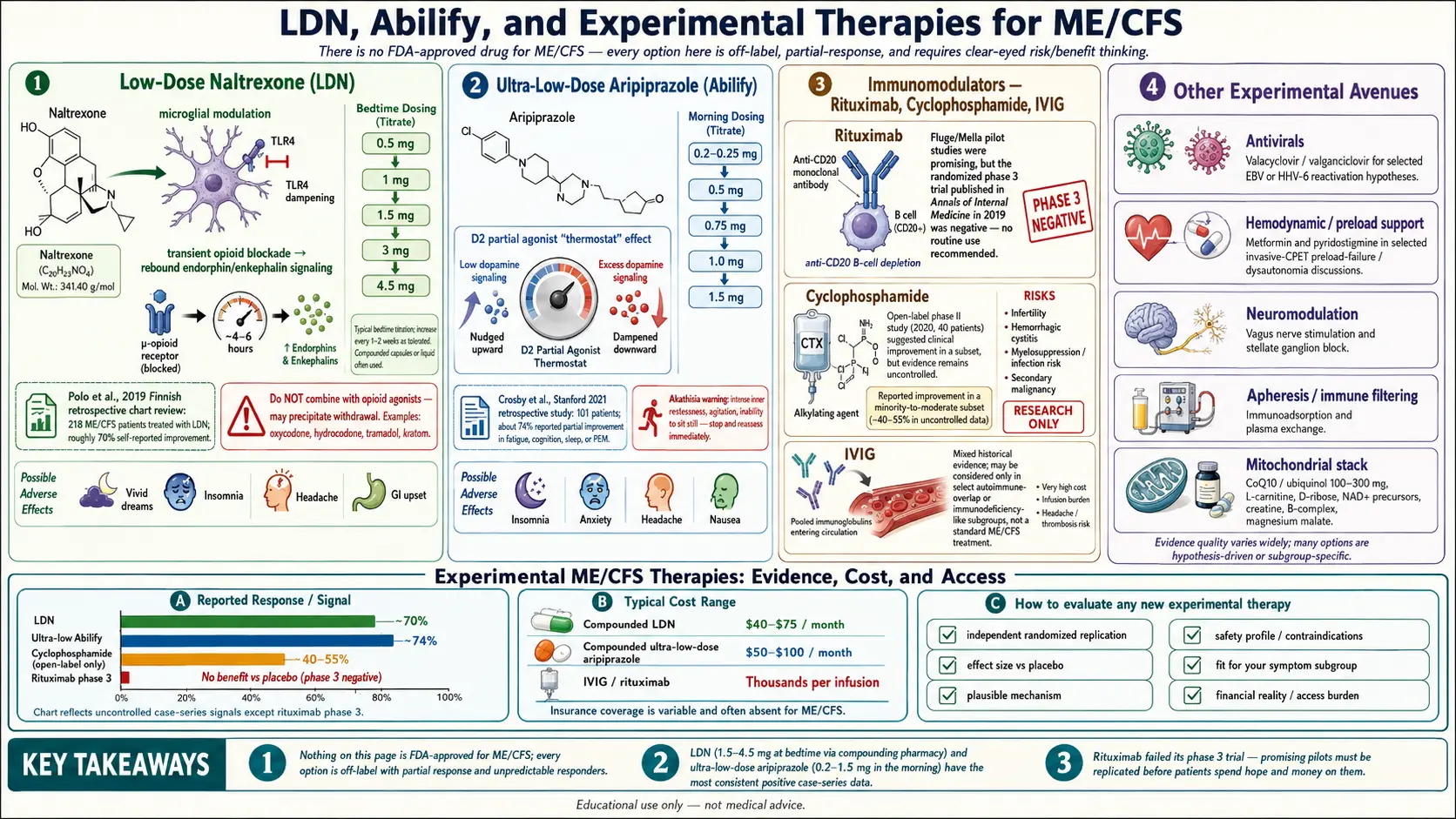
Table of Contents
- The Honest Framing — Nothing Here Is FDA-Approved
- Low-Dose Naltrexone (LDN)
- Ultra-Low-Dose Abilify (Aripiprazole)
- Rituximab — The Norwegian Story
- Cyclophosphamide
- IVIG (Intravenous Immunoglobulin)
- Metformin and Pyridostigmine
- Antivirals — Valacyclovir and Valganciclovir
- Immunoadsorption
- Mitochondrial Support — CoQ10, D-Ribose, NADH
- Vagus Nerve Stimulation and Stellate Ganglion Block
- Plasma Exchange
- How to Evaluate a New Experimental Therapy
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
The Honest Framing — Nothing Here Is FDA-Approved
Before the drug-by-drug rundown, a piece of framing that most articles skip: there is no FDA-approved medication for ME/CFS. Every therapy on this page is off-label, compassionate-use, or strictly experimental. Some have small, encouraging trials behind them. Others have one promising Phase 2 result and one crushing Phase 3 failure. A few rest on case reports and physician experience alone.
That does not mean these options are useless. It means you need to go in with clear eyes: response rates are partial, responders cannot be predicted in advance, side effects matter, and cost and access are real obstacles. A 30–40% response rate looks tiny in a cancer trial but can be life-changing in a disease where the alternative is nothing. What follows is a patient-facing map of the terrain — what each option actually is, what the evidence says, and what it typically costs a real person to try.
None of this replaces a long-term ME-literate physician. Use it to have smarter conversations with them.
Low-Dose Naltrexone (LDN)
What it is. Naltrexone is an opioid-receptor blocker originally approved at 50 mg daily to treat opioid and alcohol dependence. At one-tenth that dose or less — typically 1.5 to 4.5 mg at bedtime — it does something very different. Low-dose naltrexone appears to dampen neuroinflammation rather than block opioids in any meaningful clinical way.
How it works (best current theory). At these tiny doses, naltrexone binds Toll-like receptor 4 (TLR4) on microglia, the immune cells of the brain. In ME/CFS, post-viral syndromes, fibromyalgia, and long-illness states generally, microglia appear to be stuck in a chronically activated, pro-inflammatory mode. Blocking TLR4 helps quiet them. There is also a brief transient opioid blockade during the few hours the drug is in your system, which is thought to trigger a rebound rise in endorphins and enkephalins — a compensatory endorphin surge. Both mechanisms are plausible; neither is fully proven.
The evidence in ME/CFS. The most-cited study is Polo and colleagues (2019), a Finnish retrospective chart review of 218 ME/CFS patients treated with LDN. Roughly 70% reported meaningful improvement in fatigue, cognition, pain, and sleep. Retrospective chart reviews are the weakest form of clinical evidence — there is no placebo arm and recall bias is real — but the signal is consistent with case series from Jarred Younger's group at UAB (originally in fibromyalgia) and decades of anecdotal reports from ME/CFS specialists.
How it is actually used.
- Starting dose: 0.5 mg or 1 mg at bedtime. Most ME/CFS patients are sensitive to medication and crash on full starting doses.
- Titration: increase by 0.5–1 mg every 1–2 weeks, as tolerated, toward a target of 3–4.5 mg. Some patients do best at 1.5 or 2 mg and never go higher.
- Timing: bedtime is traditional, but vivid dreams and insomnia are the single most common reason people quit. If that happens, switch to morning dosing — the mechanism does not appear to depend on timing.
- Trial length: give it at least 3 months at target dose before deciding. Some responders take 4–6 months to bloom.
Compounding and cost. The 50 mg naltrexone tablet cannot be split accurately to 1.5 mg. You need a compounding pharmacy to make capsules or a sublingual liquid. In the U.S., compounded LDN typically runs $40–$75 per month cash. Insurance rarely covers it because the use is off-label, but some pharmacies (Skip's, Belmar, AllianceRx, and many regional compounders) will ship nationwide with a prescription. UK and EU patients often pay less.
Side effects. Most common: vivid dreams, insomnia for the first 1–2 weeks, mild headaches. Less common: GI upset, anxiety spike on the first few doses. Serious side effects are rare at these doses.
The opioid conflict. This is critical. Naltrexone blocks opioid receptors, so it will precipitate immediate withdrawal in anyone taking prescription opioids (oxycodone, hydrocodone, tramadol, kratom, etc.) and will blunt their pain relief. If you take opioids for any reason, LDN is off the table until you have been opioid-free for at least a week. Tramadol is a particular problem because many patients forget it counts. Non-opioid pain medications (NSAIDs, gabapentin, duloxetine, low-dose amitriptyline) are fine to combine with LDN.
For a deeper dive on dosing, sourcing, and protocols, see the main Low-Dose Naltrexone article. Many ME/CFS patients with comorbid fibromyalgia or POTS also find the disease-specific pages useful: LDN for Fibromyalgia and LDN for POTS.
Ultra-Low-Dose Abilify (Aripiprazole)
What it is. Aripiprazole (brand name Abilify) is a second-generation antipsychotic approved at 10–30 mg daily for schizophrenia and bipolar disorder. The ME/CFS application uses ultra-low doses of 0.2 to 2 mg — roughly one-tenth to one-twentieth the psychiatric dose.
How it works (theory). Aripiprazole is a partial agonist at the dopamine D2 receptor. Depending on ambient dopamine tone, it either nudges the receptor up or dampens it down — it behaves like a thermostat rather than a switch. At ultra-low doses, the hypothesis is that it modestly increases dopamine signaling in a brain that has become hypo-dopaminergic from chronic neuroinflammation. There may also be microglial effects shared with other antipsychotics. Nobody fully understands why it would help ME/CFS, which is part of why most rheumatologists and neurologists have never heard of this use.
The evidence. The key publication is a Stanford case series by Crosby and colleagues (2021), reporting on 101 ME/CFS patients treated with ultra-low-dose aripiprazole by a single clinician. Roughly 74% reported at least partial improvement, with benefits most pronounced for cognitive fog and fatigue. This is one uncontrolled case series from one clinic. Placebo and selection bias are unaccounted for, and no randomized trial has yet replicated the finding.
How it is used.
- Starting dose: 0.2–0.25 mg in the morning. This requires compounding (commercial Abilify comes in 2, 5, 10 mg tablets).
- Titration: adjust by 0.25 mg every 1–2 weeks. Most responders settle in the 0.5–1.5 mg range. Going higher often worsens symptoms — this is unusual for a drug and part of why ultra-low dosing matters.
- Timing: morning. Aripiprazole has a long half-life (~75 hours), which also means side effects build over weeks.
Compounded cost. $50–$100 per month cash, similar to LDN.
Side effects and risks. The main risk is akathisia — an intensely unpleasant inner restlessness, a need to keep moving, often misidentified as anxiety. It can appear at any dose and is the most common reason people stop. Other antipsychotic-class warnings (weight gain, metabolic syndrome, tardive dyskinesia) are vanishingly rare at these doses and short durations, but not zero. Long-term safety data at ultra-low doses does not exist. Anyone starting aripiprazole should know what akathisia feels like and stop immediately if it appears.
Rituximab — The Norwegian Story
What it is. Rituximab is a monoclonal antibody that depletes CD20-positive B cells. It is FDA-approved for non-Hodgkin lymphoma, rheumatoid arthritis, and several autoimmune conditions. The ME/CFS story started by accident.
The origin. In the mid-2000s, Norwegian oncologists Øystein Fluge and Olav Mella noticed that ME/CFS patients who happened to be treated with rituximab for lymphoma sometimes had their ME/CFS symptoms dramatically improve. They followed up with small controlled trials.
The evidence arc.
- Fluge & Mella, PLoS ONE 2011. Randomized, placebo-controlled pilot in 30 patients. Roughly two-thirds of the rituximab arm reported meaningful improvement, often delayed by months. The ME community was electrified.
- Open-label phase 2 (2015). Extended dosing confirmed the signal in 29 patients with response rates around 64%.
- Phase 3 (Fluge, Annals of Internal Medicine, 2019). Large multicenter RCT, 151 patients, proper blinding. Negative. Rituximab did not outperform placebo. The field deflated.
What to make of this. The Phase 3 was well-run and reasonably sized. The honest reading is that rituximab is not a broadly effective ME/CFS drug. A minority of clinicians still argue there may be a B-cell-driven subgroup that responded in the earlier trials and got diluted out of the bigger one — but that remains hypothesis, not evidence. Rituximab should be considered closed for routine ME/CFS use. It is worth knowing about because the story illustrates why single positive trials must be replicated before patients spend hope and money on them.
Cyclophosphamide
What it is. A broadly immunosuppressive chemotherapy drug, used for aggressive autoimmune disease (lupus nephritis, vasculitis) and some cancers.
The evidence. The same Fluge/Mella group reported an open-label Phase 2 study in 40 ME/CFS patients (Fluge et al., 2020), with about 40% showing sustained improvement at follow-up. No placebo arm. No large RCT has followed.
Why it is rarely used. Cyclophosphamide is genuinely toxic: infertility risk, bone marrow suppression, hemorrhagic cystitis, long-term secondary cancer risk. The risk-benefit math is hard to justify outside a trial, especially when we cannot yet predict responders. It remains of biological interest because it suggests an immune-driven subgroup exists, but it is not an appropriate off-label option.
IVIG (Intravenous Immunoglobulin)
What it is. Pooled human antibodies from thousands of donors, infused over several hours. Used in primary immunodeficiency, autoimmune neurologic disease (CIDP, myasthenia gravis), and some vasculitides.
The evidence in ME/CFS. Mixed and mostly old. Rowe (1997) reported encouraging open-label results in adolescent ME/CFS. Later controlled adult trials were negative or inconsistent. Today IVIG is most commonly used in ME/CFS patients who have documented immune abnormalities — low IgG subclasses, specific antibody deficiency, or overlapping small-fiber neuropathy or autoimmune dysautonomia. In those subgroups it sometimes helps substantially.
Access reality. IVIG is expensive ($5,000–$10,000 per infusion) and insurance approval requires one of the recognized FDA-approved indications. Getting it for "ME/CFS" alone is nearly impossible; getting it for common variable immune deficiency or biopsy-proven small-fiber neuropathy is sometimes feasible. If IVIG is on your radar, the first step is a full immunology workup: quantitative immunoglobulins, IgG subclasses, pneumococcal and tetanus titers before and after vaccination.
Metformin and Pyridostigmine
Metformin. The old diabetes drug has attracted interest because it targets AMPK and mitochondrial function — both implicated in ME/CFS. Small pilots are underway; the case for trying 500–1500 mg daily in ME/CFS patients who also have insulin resistance or metabolic syndrome is stronger than in lean patients. Cheap, well-tolerated, and widely available, so the risk of a trial is low. Main side effect: GI upset and B12 depletion with long-term use (supplement B12 if you stay on it more than a year).
Pyridostigmine (Mestinon). An acetylcholinesterase inhibitor originally approved for myasthenia gravis. At 30–60 mg three times daily, it boosts acetylcholine signaling and appears to improve orthostatic intolerance, cognitive fog, and exercise tolerance in a subset of ME/CFS patients, particularly those with overlapping POTS. Joseph and colleagues (2022) from Harvard invasive CPET studies showed improved cardiac preload and oxygen extraction on pyridostigmine. It is used widely by ME and POTS specialists. Side effects are predictable cholinergic ones: cramping, diarrhea, increased saliva, muscle twitching — all dose-dependent and reversible.
Neither drug is ME/CFS-specific, but both are cheap, generic, well-understood, and reasonable additions to a comprehensive plan under a knowledgeable physician.
Antivirals — Valacyclovir and Valganciclovir
The rationale. A meaningful fraction of ME/CFS is triggered by herpes-family viruses — Epstein-Barr virus (EBV) and human herpesvirus 6 (HHV-6) most often. See the sibling article on post-viral triggers. The theory is that in some patients these viruses do not fully clear; they smolder in a low-grade, partially-reactivated state that keeps the immune system and brain chronically inflamed. Long-term antivirals, in theory, suppress the smolder.
The evidence. A. Martin Lerner (Michigan) and Jose Montoya (Stanford) each published open-label case series in ME/CFS subgroups with elevated EBV or HHV-6 antibody titers. Valacyclovir (primarily for EBV) and valganciclovir (broader, including HHV-6 and CMV) produced partial response rates of roughly 30–50% after 6–12 months of treatment. Subsequent placebo-controlled trials have been smaller and more mixed. Montoya's 2013 valganciclovir RCT showed improvement in cognitive and fatigue outcomes versus placebo in an antibody-selected subgroup.
How it is used. Valacyclovir 1 g two to four times daily, or valganciclovir 450–900 mg daily, typically for 6–12 months. Valganciclovir is the more potent option but carries real bone-marrow toxicity and requires frequent lab monitoring (CBC every 2–4 weeks). Valacyclovir is much safer but may be too narrow for HHV-6 dominant cases. Finding a physician who will prescribe these off-label for ME/CFS is the hard part — usually an infectious disease specialist who works with ME/CFS patients or one of the handful of ME specialty clinics.
Immunoadsorption
What it is. A procedure like dialysis in which your blood is circulated through a column that selectively removes autoantibodies — particularly those against adrenergic and muscarinic receptors, which have been repeatedly implicated in ME/CFS and POTS by German researchers (Scheibenbogen group, Charité Berlin).
The evidence. Small open-label trials from Berlin show meaningful improvement in a subgroup of ME/CFS patients with elevated β2-adrenergic and M3/M4 muscarinic receptor autoantibodies. Benefits last months to a year or more in some responders. No large RCT has been completed.
Access. Available mainly in Germany and a few European centers, generally on self-pay basis (cost often $15,000–$25,000 for a course of sessions). Not available in the U.S. for this indication.
Mitochondrial Support — CoQ10, D-Ribose, NADH
Multiple studies show disturbed energy metabolism in ME/CFS — impaired oxidative phosphorylation, reduced ATP production, increased oxidative stress. Three supplements have accumulated small-trial evidence:
- Coenzyme Q10 (CoQ10 / ubiquinol) at 200–400 mg daily, often combined with NADH. The Castro-Marrero group (Spain, 2015 and 2016) published randomized controlled trials showing reductions in fatigue, improved sleep, and reduced heart-rate recovery abnormalities versus placebo. Effect sizes were modest but real.
- D-ribose at 5 g three times daily. Open-label Teitelbaum studies reported broad improvements; placebo-controlled data are weaker. Cheap and well-tolerated, but expect at most a modest effect.
- NADH at 10–20 mg daily. Used in combination with CoQ10 in the Castro-Marrero protocol.
None of these are curative. They sit in the category of cheap, safe, evidence-supported additions that a minority of patients notice clearly, that a larger fraction notice subtly, and that some do not notice at all. A 3-month trial is reasonable.
Vagus Nerve Stimulation and Stellate Ganglion Block
Vagus nerve stimulation (VNS). Non-invasive devices (transcutaneous auricular VNS, gammaCore handheld devices) target the vagus nerve through the ear or the neck. The mechanism is anti-inflammatory: vagal stimulation dampens systemic cytokine production via the cholinergic anti-inflammatory pathway. Small pilot studies in ME/CFS, long-illness populations, and fibromyalgia show signal; RCT evidence is preliminary. Devices range from cheap clip-on ear stimulators (~$100) to prescription handhelds (several hundred dollars). Risk is minimal; expectation should be modest.
Stellate ganglion block (SGB). An anesthetic injection into the sympathetic ganglion in the neck, originally used for complex regional pain syndrome and PTSD. Case reports and small series suggest benefit in dysautonomia and some post-viral fatigue states. It is genuinely experimental for ME/CFS — no RCT exists, mechanism is speculative (sympathetic reset), and the procedure costs $1,000–$2,500 per side, usually repeated. Only consider under an anesthesiologist experienced with the procedure and after honest discussion of the limited evidence.
Plasma Exchange
Therapeutic plasma exchange (TPE) physically removes plasma and replaces it with donor plasma or albumin, clearing circulating antibodies and inflammatory proteins. It is established therapy for several neuroimmune diseases (myasthenia crisis, Guillain-Barré, TTP). In ME/CFS and overlapping long-illness cohorts, plasma exchange is strictly research-stage: a handful of small European series, mostly overlapping with the immunoadsorption work. No U.S. clinical pathway exists. Listed here for completeness, not as a realistic option.
How to Evaluate a New Experimental Therapy
New ME/CFS therapies will appear in the press regularly. A practical checklist before you spend money, hope, or your fragile energy budget:
- Peer-reviewed or preprint? Preprints (medRxiv, bioRxiv, Research Square) have not been vetted. Treat anything preprint-only as provisional.
- How big was the trial? A case series of 5 patients is a hypothesis; 30 is a pilot; 150 with a placebo arm is a real signal.
- Placebo or open-label? In a disease with strong day-to-day variability and a large subjective component, placebo effects of 20–40% are routine. An open-label 50% response rate may collapse to 10% versus placebo.
- Who funded and who ran it? A trial run by the clinic that sells the treatment is not dispositive.
- Your baseline. If you are severely ill and bedbound, a therapy that causes a PEM crash can cost you months of recovery. The calculus is very different from mild-moderate illness.
- Reversibility. Supplements and LDN are easily stopped. Chemotherapy agents, surgical procedures, and injectable antibodies are not.
- Stacking. Starting three things at once means you will never know which one worked or which one crashed you. Change one variable at a time.
The most common mistake ME/CFS patients make is not trying experimental therapies — it is trying too many, too fast, without baselines, and then losing track of what helped. A simple weekly symptom diary (fatigue, PEM frequency, cognition, sleep, orthostatic symptoms, each 0–10) before and during any trial pays off enormously.
For the structural framework you are working within, see pacing and the energy envelope. No drug replaces pacing; drugs, at best, widen the envelope.
Key Research Papers
- Polo O, et al. Low-dose naltrexone in the treatment of myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). Curr Neuropharmacol. 2019.
- Crosby LD, et al. Off label use of aripiprazole shows promise as a treatment for myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS): a retrospective case series. J Transl Autoimmun. 2021.
- Fluge Ø, et al. B-lymphocyte depletion in patients with myalgic encephalomyelitis/chronic fatigue syndrome: a randomized, double-blind, placebo-controlled trial. Ann Intern Med. 2019.
- Fluge Ø, et al. Benefit from B-lymphocyte depletion using the anti-CD20 antibody rituximab in chronic fatigue syndrome: a double-blind and placebo-controlled study. PLoS ONE. 2011.
- Castro-Marrero J, et al. Does oral coenzyme Q10 plus NADH supplementation improve fatigue and biochemical parameters in chronic fatigue syndrome? Antioxid Redox Signal. 2015.
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on pharmacologic and experimental approaches to ME/CFS:
- Low-dose naltrexone and ME/CFS
- Aripiprazole (Abilify) in ME/CFS
- Rituximab and ME/CFS — the Fluge/Mella trials
- Cyclophosphamide in ME/CFS
- Valganciclovir and herpesvirus-triggered ME/CFS
- Pyridostigmine in ME/CFS and orthostatic intolerance
- Immunoadsorption and autoantibody removal in ME/CFS
- CoQ10 and NADH in ME/CFS
- Vagus nerve stimulation for fatigue
- IVIG in ME/CFS
Connections
- ME/CFS Overview
- Post-Viral Triggers: EBV and Others
- Post-Exertional Malaise
- Pacing and the Energy Envelope
- 2-Day CPET and Objective Testing
- ME/CFS Diagnostic Criteria
- Orthostatic Intolerance in ME/CFS
- Severe and Very Severe ME
- Mitochondrial Dysfunction and Energy Metabolism
- Low-Dose Naltrexone
- LDN for Fibromyalgia
- LDN for POTS
- POTS
- MCAS
- Fibromyalgia
- Chronic Pain
- Fatigue