Hereditary Alpha-Tryptasemia (HαT)
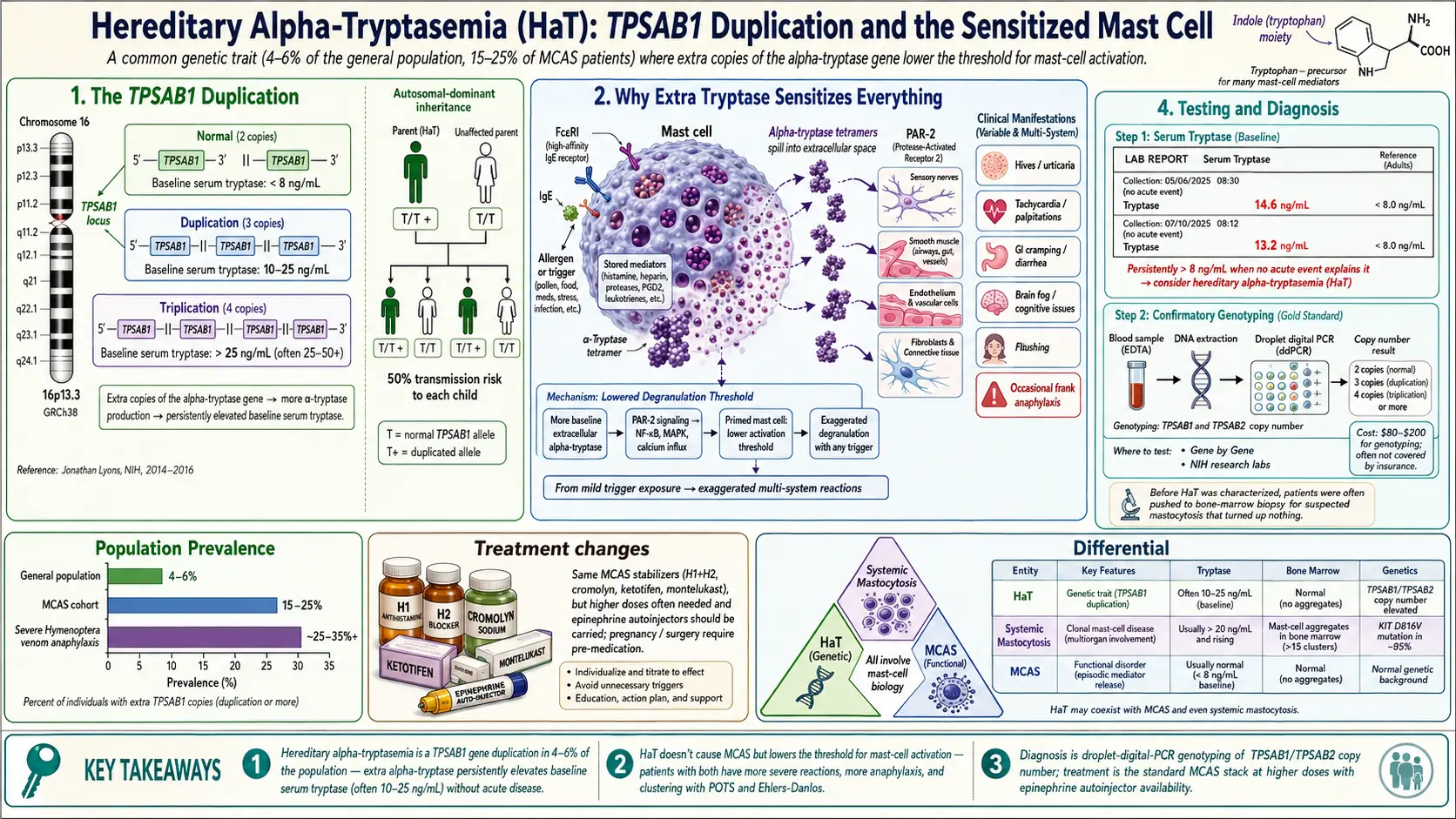
Table of Contents
- What Hereditary Alpha-Tryptasemia Is
- How Common It Is
- Why It Matters for MCAS
- Symptoms Associated with HαT
- Overlap with the POTS/MCAS/EDS Triad
- Testing for HαT
- Inheritance
- What HαT Diagnosis Changes About Treatment
- HαT vs. Systemic Mastocytosis vs. MCAS
- Practical Implications for Life Planning
- Emerging Research
- Resources & Support
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
1. What Hereditary Alpha-Tryptasemia Is
Hereditary alpha-tryptasemia (HαT) is a genetic trait, not a disease in the classical sense. It is caused by a duplication or triplication of the TPSAB1 gene on chromosome 16, the gene that encodes alpha-tryptase. People with HαT carry extra copies of TPSAB1 and therefore make more alpha-tryptase at baseline — even when their mast cells are sitting quietly and not actively degranulating.
Tryptase is the enzyme mast cells release when they activate, and it is measured in the blood as a proxy for mast cell burden. Most of the population has two copies of TPSAB1 (one from each parent) and a baseline serum tryptase below 8 ng/mL. HαT patients, because of gene dosage, run baseline tryptase values that are persistently elevated — often 10 to 25 ng/mL — without any acute allergic event to explain it.
The trait was first characterized by Jonathan Lyons and colleagues at the NIH between 2014 and 2016. Before that work, patients with unexplained elevated tryptase were often labeled as having “idiopathic” tryptasemia or pushed toward bone marrow biopsies for suspected mastocytosis that turned up nothing. The discovery of the TPSAB1 duplication gave a name and a mechanism to a population that had been invisible to medicine.
2. How Common It Is
HαT is remarkably common. Population-level studies estimate 4–6% of the general population carries the duplication. Among patients with a clinical picture of MCAS, the rate rises to 15–25%. Among patients with severe insect venom anaphylaxis or unexplained systemic reactions, it climbs higher still.
That makes HαT one of the most common genetic conditions that almost nobody — including most primary care physicians and many allergists — has heard of. If your tryptase is high and your family has always been “the allergic one” or “the flushing one” or “the bendy one,” HαT deserves a look.
3. Why It Matters for MCAS
HαT does not directly cause MCAS. You can have HαT and be completely asymptomatic. But when HαT is present, it lowers the threshold at which mast cells activate and amplifies the downstream symptoms when they do. Patients with HαT + MCAS tend to have a more severe clinical course than patients with MCAS alone: more anaphylaxis, more flushing, more GI involvement, more medication reactions.
The working model is that extra baseline alpha-tryptase — because of its activity on PAR-2 (protease-activated receptor 2) on sensory nerves, smooth muscle, and connective tissue — sensitizes the whole downstream signaling environment. Mast cells don’t need as large a trigger to produce a clinically significant reaction.
For a patient struggling to control MCAS, a positive HαT result is actually good news in one sense: it explains why “standard” doses of antihistamines feel underpowered and why your reactions are more severe than textbook MCAS cases predict.
4. Symptoms Associated with HαT
Not every HαT carrier is symptomatic — probably half are silent. Among those who do have symptoms, the common features include:
- Easy flushing — face, neck, and chest, often with alcohol, heat, or stress
- Headache and migraine
- Itching, hives, and dermographism (the ability to “draw” raised red lines on the skin with light pressure)
- GI symptoms — IBS-like bloating, reflux, abdominal pain, altered bowel habits
- Bone and joint pain — often unexplained, sometimes migratory
- Autonomic symptoms — lightheadedness on standing, palpitations, temperature dysregulation (overlap with POTS)
- Joint hypermobility — clear overlap with Ehlers-Danlos and hypermobility spectrum disorder
- Anaphylaxis — especially venom anaphylaxis. HαT is substantially overrepresented in patients with severe bee, wasp, and hornet sting reactions
- Anxiety and depression — thought to be mediated in part through mast cell activation in the brain and meninges
No single symptom is specific to HαT; the pattern is what matters. A patient with flushing + bone pain + anxiety + hypermobility + a persistently elevated tryptase is a textbook picture.
5. Overlap with the POTS/MCAS/EDS Triad
HαT clusters strongly with the POTS/MCAS/hypermobile EDS triad. The three conditions share enough patients that many specialists now treat them as a single neuroimmune-connective-tissue syndrome rather than three separate diagnoses.
The current mechanistic theory: elevated alpha-tryptase cleaves PAR-2 on autonomic nerve endings and on the fibroblasts that lay down collagen. Chronic PAR-2 activation contributes to autonomic dysregulation (the POTS phenotype) and to connective tissue fragility (the EDS/HSD phenotype). That is why fixing the mast cell side of the equation often improves both dizziness and joint instability. For a deeper dive, see the POTS/MCAS/EDS Triad.
6. Testing for HαT
Step 1: Baseline Serum Tryptase
Start with a simple, cheap blood test: baseline serum tryptase, drawn at least 24 hours after any suspected flare. The standard lab reference range tops out at 11.4 ng/mL, but the threshold for clinical suspicion is lower:
- Tryptase > 8 ng/mL — consider HαT testing, especially with matching symptoms
- Tryptase > 11.4 ng/mL — definitely test
- Tryptase > 20 ng/mL — test for HαT and rule out systemic mastocytosis
Step 2: TPSAB1 Copy Number Variant Testing
The definitive test is TPSAB1 copy number variant (CNV) testing, done by droplet digital PCR. This is not a standard commercial lab offering — blood has to be sent to a specialty laboratory. Options include:
- Gene by Gene (a FamilyTreeDNA subsidiary) — direct-to-consumer, roughly $350–500, no physician order required
- Genetic Testing Laboratories — research-based, generally requires a physician order
- Academic centers — the NIH and Brigham & Women’s research groups offer testing for enrolled study participants
Interpreting Results
- 2 copies — normal, one from each parent, no HαT
- 3 copies — duplication on one allele, heterozygous HαT (most common positive result)
- 4 copies — homozygous duplication or heterozygous triplication, typically more severe phenotype
- 5 or more copies — rare, tends to produce the most severe clinical picture
Total cost, depending on route, runs $350–700 out of pocket. Insurance coverage is improving as awareness of the trait spreads, but many patients still pay cash.
7. Inheritance
HαT is inherited in an autosomal dominant pattern. A single copy of the duplicated allele is sufficient to produce the biochemical phenotype of elevated baseline tryptase. Children of a parent with HαT have a 50% chance of inheriting the duplication.
Because penetrance for symptoms is variable — some carriers are asymptomatic, others are severely affected — a positive result in one family member should prompt cascade screening of first-degree relatives, particularly if there is any family history of anaphylaxis, unexplained flushing, hypermobility, or MCAS-like symptoms. Silent carriers often have subtle findings they had written off as personal quirks for decades.
8. What HαT Diagnosis Changes About Treatment
Confirming HαT rarely changes the core medication stack for MCAS, but it changes several important things around the edges:
- MCAS treatment stays the same. The H1 + H2 + stabilizer backbone remains the foundation — see H1 and H2 Blocker Stacks and Cromolyn and Ketotifen Protocols.
- Venom immunotherapy is indicated for anyone with a documented insect sting reaction. The long-term risk of recurrence is high enough in HαT that desensitization is usually worth it.
- Epinephrine auto-injectors should be prescribed generously. HαT patients carry a higher lifetime anaphylaxis risk than baseline MCAS patients. Two EpiPens at all times is the standard.
- Pre-medication before procedures becomes more important. Anesthesia, IV contrast, and first-dose antibiotics are higher-risk events; a standard pre-med (H1 + H2 + steroid ± hydroxyzine) should be written into the chart.
- Family cascade screening. Test first-degree relatives and counsel the family together.
- Redefine the patient’s “normal” tryptase. An HαT patient’s baseline may be 15–25 ng/mL. The diagnostic criterion for an acute mast cell event is still the 20%-plus-2-ng/mL rise above baseline — but the baseline has to be known in advance for that rule to be useful.
9. HαT vs. Systemic Mastocytosis vs. MCAS
Three conditions present with elevated tryptase, and distinguishing them is essential.
- Systemic mastocytosis (SM). Increased number of mast cells, often clonal. Confirmed by bone marrow biopsy, which shows characteristic aggregates. The KIT D816V somatic mutation is usually present. Tryptase is typically > 20 ng/mL and rises over time.
- MCAS (non-clonal). Normal mast cell numbers, but dysregulated activation. No KIT mutation. Tryptase may be normal, elevated, or fluctuate around a normal baseline.
- HαT. A genetic trait. Normal mast cell numbers. Baseline tryptase is elevated because of TPSAB1 gene dosage, not because of mast cell pathology. Can coexist with either SM or MCAS.
The most common combination in clinical practice is HαT + MCAS, and the two together produce the severe, hard-to-control phenotype many patients experience. HαT is also significantly more common in patients with systemic mastocytosis than in the general population — suggesting the extra tryptase copies may contribute to the clinical expression of both conditions.
10. Practical Implications for Life Planning
Pregnancy
Pregnancy-associated hormonal shifts can trigger more severe mast cell flares in HαT mothers. Plan pre-medication protocols in advance, identify a labor-and-delivery team aware of the diagnosis, and build anesthesia choices (fentanyl and propofol are usually well tolerated; morphine and succinylcholine are avoided) before labor begins.
Children with HαT
Cascade testing often identifies children early. Pediatric counseling should cover anaphylaxis risk, school epinephrine plans, and awareness of venom sensitivity. Many symptomatic children improve markedly on a simple H1 + H2 regimen.
Surgery
A written pre-op protocol is essential: H1 + H2 + steroid ± hydroxyzine the night before and morning of surgery. Avoid succinylcholine, morphine, and NSAIDs; prefer fentanyl and propofol. Have epinephrine drawn up and immediately accessible.
Vaccination
Vaccines are generally safe. Pre-medicate with an H1 + H2 dose two hours before, space multi-component vaccines into separate visits when possible, and observe for 30 minutes after injection.
Travel
Carry at least two epinephrine auto-injectors, a written allergy action plan, a medication list with doses, and copies of key lab results. For international travel, a letter from your physician explaining the diagnosis clears most customs and medical-supply issues.
11. Emerging Research
- HαT and mastocytosis overlap. HαT is substantially more common in systemic mastocytosis cohorts than in the general population, suggesting the duplication may contribute to disease expression.
- Basophil activation test (BAT). Emerging as an adjunct diagnostic for mediator release, particularly for drug and food reactions that are hard to provoke safely in a clinical setting.
- Anti-tryptase biologics. Experimental monoclonal antibodies that bind and neutralize tryptase directly are in early-phase trials.
- Mepolizumab (anti-IL-5) is under investigation for HαT-associated MCAS with eosinophilic features.
12. Resources & Support
- The Mast Cell Disease Society (TMS) — patient advocacy organization, physician directory, patient education materials
- Dysautonomia International — overlap resources for the POTS/HαT/MCAS population
- NIH HαT Registry — Dr. Jonathan Lyons’s research group at the NIH enrolls patients and family members for research testing and natural-history studies
- MastAttack.org — clinician-reviewed deep-dive patient education, heavily cited, widely used by the mast cell community
13. Key Research Papers
- Lyons JJ, et al. Elevated basal serum tryptase identifies a multisystem disorder associated with increased TPSAB1 copy number. Nat Genet. 2016;48(12):1564–1569.
- Lyons JJ, et al. Hereditary alpha tryptasemia: genotyping and associated clinical features. J Allergy Clin Immunol. 2020.
- Akin C. How to evaluate the patient with a suspected mast cell disorder. J Allergy Clin Immunol. 2014.
- Afrin LB, et al. Diagnosis of mast cell activation syndrome: a global “consensus-2.” Diagnosis. 2020.
14. Research Papers
Curated PubMed topic searches for HαT and adjacent mast cell biology. Each link opens a live PubMed query so you always see current studies.
- PubMed: Hereditary alpha tryptasemia
- PubMed: TPSAB1 copy number variant
- PubMed: Tryptase, MCAS, and mastocytosis
- PubMed: Lyons — hereditary tryptasemia
- PubMed: Alpha tryptase and anaphylaxis
- PubMed: Venom anaphylaxis and tryptase
- PubMed: PAR-2, mast cells, and tryptase
- PubMed: Hypermobility and mast cell activation
Connections
- MCAS Overview
- H1 and H2 Blocker Stacks
- Cromolyn and Ketotifen Protocols
- Mediators and Symptom Map
- MCAS Testing Guide
- Low-Histamine Diet
- Triggers and Flare Management
- Natural Mast Cell Stabilizers
- Hormone Sensitivity and Menstrual Flares
- Alpha-Gal Syndrome
- Alpha-Gal Natural History and Tolerance
- POTS
- POTS-MCAS-EDS Triad
- Allergies
- SIBO
- Quercetin
- Vitamin C
- Testing and Diagnosis