H1 and H2 Blocker Stacks for MCAS
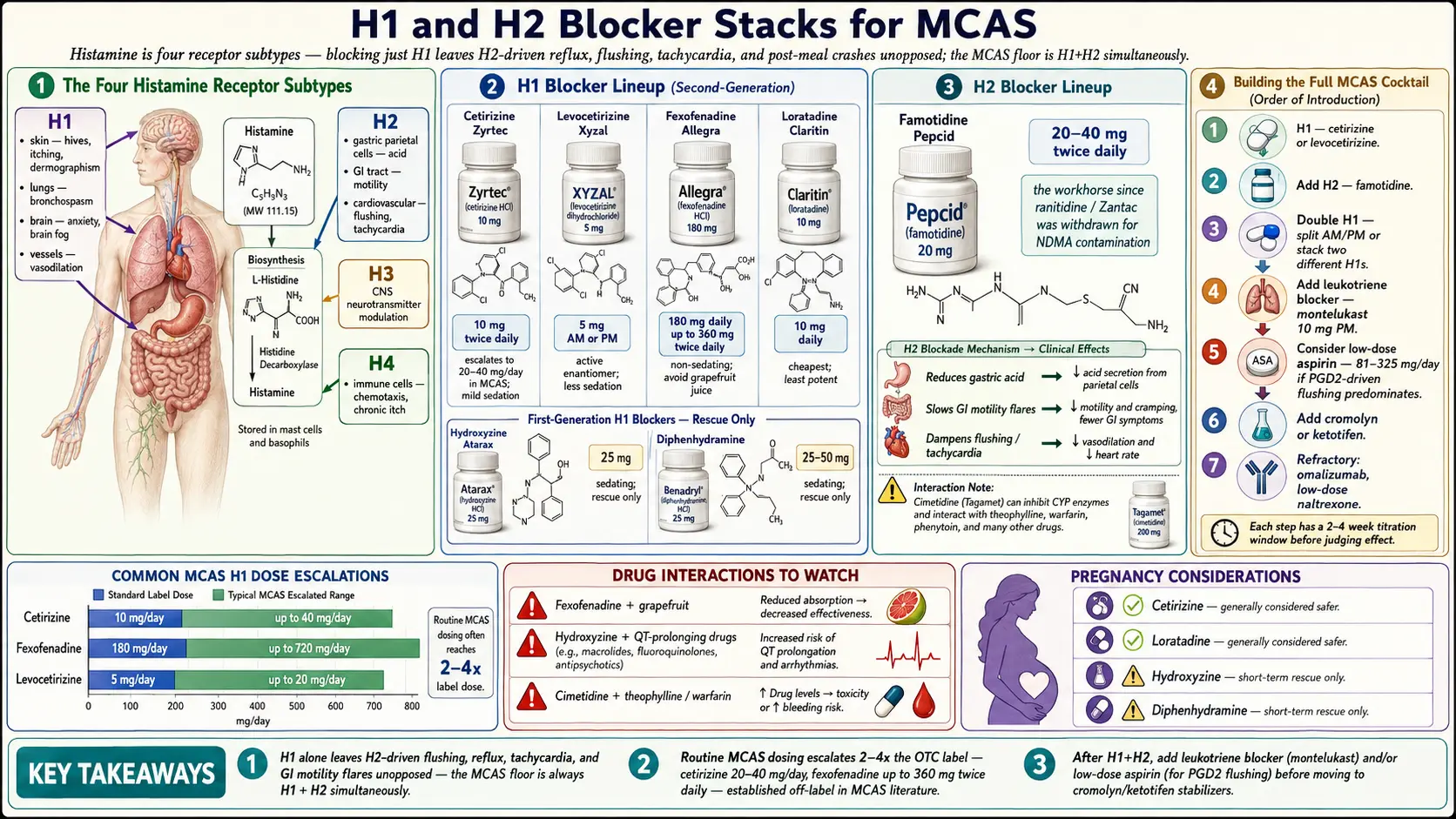
Table of Contents
- Why MCAS Needs More Than One Antihistamine
- H1 Blockers (Skin, Lungs, Brain, Vascular)
- H2 Blockers (GI, Flushing, Cardiovascular)
- Leukotriene Receptor Antagonists
- Aspirin for PGD2-Dominant Disease
- The “MCAS Cocktail” — Order of Introduction
- Morning vs Night Dosing
- Doubling Up Safely
- Drug Interactions to Watch
- Side Effects and How to Manage
- Pregnancy Considerations
- Pediatric Dosing
- Cost & Access
- When Stacks Aren’t Enough
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
1. Why MCAS Needs More Than One Antihistamine
Histamine is not one signal — it’s four. Four different receptor subtypes (H1, H2, H3, H4) sit on different tissues and produce different symptoms. H1 drives itching, hives, bronchospasm, vasodilation, and the brain-fog/anxiety component. H2 drives gastric acid production, GI motility changes, and a major share of flushing and tachycardia. H3 is primarily CNS (neurotransmitter modulation). H4 is concentrated on immune cells and mediates chemotaxis and chronic itch.
If you block only H1 with a single Zyrtec a day, every H2-driven symptom — reflux, bloating, flushing, post-meal crashes — rolls on unopposed. That’s why the standard MCAS floor is H1 + H2 simultaneously, and why patients who stall out on that combination go on to add a leukotriene blocker, a mast cell stabilizer, and often aspirin or natural stabilizers like quercetin on top.
For a full map of which mediators drive which symptoms, see Mediators & Symptom Map.
2. H1 Blockers (for Skin, Lungs, Brain, Vascular Symptoms)
Second-generation H1 antihistamines are the backbone. They cross the blood-brain barrier less than the first-generation drugs (diphenhydramine, hydroxyzine), so they don’t wreck your cognition. In MCAS, they are routinely dosed above the OTC labeling — this is off-label but well-established in the MCAS literature.
Cetirizine (Zyrtec)
Start at 10 mg every morning. Many MCAS patients require 10 mg twice daily (off-label), and clinical practice often escalates to 20–40 mg/day total. Cetirizine has the best balance of potency and tolerability of the second-gen H1s — it quiets skin and airway symptoms aggressively and has real activity on eosinophil recruitment. Minimal but non-zero sedation (~10% of patients).
Levocetirizine (Xyzal)
5 mg AM or PM. This is the active enantiomer of cetirizine and is often better tolerated at equivalent potency — less sedation, less dry mouth. Now OTC in the US. Good first choice if cetirizine makes you drowsy but you want cetirizine-level efficacy.
Fexofenadine (Allegra)
180 mg daily, escalating to 360 mg twice daily in refractory MCAS. Truly non-sedating — fexofenadine barely crosses the blood-brain barrier at all. The trade-off is that it’s less potent than cetirizine for some patients, particularly for skin symptoms. Ideal for patients who drive, work, or cannot tolerate any cognitive dulling. Absorption is reduced by grapefruit and orange juice; take with water.
Loratadine (Claritin)
10 mg daily. The least potent of the common second-gen H1s, but the cheapest and one of the best-tolerated. Acceptable as a maintenance H1 for mild MCAS, or as a second H1 stacked with cetirizine to cover a dip in the middle of the day.
Hydroxyzine (Atarax, Vistaril)
10–50 mg at bedtime. First-generation and frankly sedating — but that’s exactly why it earns a place in MCAS protocols. Hydroxyzine is an outstanding nighttime H1 for patients with nocturnal itching, sleep-destroying flushing, or the anxiety component of MCAS (it has some 5-HT1A activity). Many MCAS patients live on 25–50 mg qhs as their evening anchor. Avoid morning dosing unless you’re home and not driving.
Diphenhydramine (Benadryl)
25–50 mg — for rescue only, not daily maintenance. The 2015 JAMA Internal Medicine analysis linking cumulative anticholinergic burden to dementia risk was persuasive enough that most MCAS clinicians now reserve diphenhydramine for acute reactions, EpiPen-adjacent situations, and episodic flares. It’s fine as a rescue; it is not a daily plan.
Doxepin
10–25 mg at bedtime. Technically a tricyclic antidepressant, but at low doses doxepin is one of the most potent H1 antihistamines ever characterized — reportedly more potent per milligram than diphenhydramine. Reserved for severe MCAS with refractory urticaria or insomnia. Sedating; dry mouth; not compatible with MAOIs and caution-required with SSRIs.
Ketotifen
Dual H1 blocker + mast cell stabilizer; not a standard second-gen antihistamine and covered separately — see Cromolyn & Ketotifen Protocols.
3. H2 Blockers (for GI Symptoms, Flushing, Cardiovascular)
H2 blockers were developed for peptic ulcers, but in MCAS they pull double duty: they block the H2-mediated flushing, tachycardia, and gut-motility symptoms that H1 blockers cannot touch. Every MCAS patient on an H1 should also be on an H2 unless there is a specific contraindication.
Famotidine (Pepcid)
20–40 mg twice daily. This is the default H2 blocker in 2024—2026 practice. Ranitidine (Zantac) was pulled from the market in 2020 after the FDA found NDMA contamination in manufactured lots, and nobody trusts the replacement formulations. Famotidine is OTC, cheap, essentially no CYP interactions, and well-tolerated. Many MCAS patients do best on 40 mg BID or even TID.
Nizatidine (Axid)
150–300 mg twice daily. An alternative when famotidine is insufficient or unavailable. Equivalent efficacy in most head-to-head data. Useful as a rotation if famotidine seems to lose effect (uncommon).
Cimetidine (Tagamet)
400 mg twice daily. The original H2 blocker. It works, but it is a strong CYP450 inhibitor (1A2, 2C9, 2D6, 3A4) and thus a drug-interaction minefield — warfarin, theophylline, phenytoin, many SSRIs, tricyclics, beta-blockers. Cimetidine also has weak anti-androgen effects. Generally avoid as first-line in favor of famotidine.
Do not use PPIs as substitutes for H2 blockers. Omeprazole, pantoprazole, and esomeprazole shut down gastric acid production entirely but do nothing to H2-receptor histamine signaling on cardiovascular, enteric, or dermal tissue. Chronic PPI use also drives small intestinal bacterial overgrowth (see SIBO), which in turn aggravates MCAS symptoms in a painful loop. Reflux in MCAS is frequently histamine-driven; treat it with famotidine, not a PPI, whenever possible.
4. Leukotriene Receptor Antagonists (for Respiratory + Some GI)
Mast cells release not just histamine but also leukotrienes (LTC4, LTD4, LTE4) — lipid mediators 1000× more potent than histamine at causing bronchoconstriction and airway edema. If your MCAS has a strong respiratory, sinus, or asthma-overlap component, an H1+H2 stack will not reach those symptoms.
Montelukast (Singulair)
10 mg daily, typically at bedtime. Blocks the CysLT1 receptor for LTD4 and LTE4. Highly effective for chronic rhinosinusitis, aspirin-exacerbated respiratory disease, and mast-cell-driven asthma overlap. In 2020 the FDA applied a boxed warning for neuropsychiatric adverse effects (depression, suicidality, sleep disturbance, vivid dreams) — these are uncommon but real, and they tend to appear within the first weeks. Warn patients, and stop at the first sign of mood change.
Zafirlukast (Accolate)
20 mg twice daily. A less-used alternative. Requires empty-stomach dosing (food reduces absorption ~40%). Rare hepatotoxicity; monitor LFTs if used long-term. Reasonable backup when montelukast is not tolerated.
5. Aspirin (for PGD2-Dominant Disease)
Mast cells also release prostaglandin D2 (PGD2), which is the dominant mediator in flushing-dominant MCAS and in systemic mastocytosis. If your flushing, tachycardia, and vascular symptoms ignore a fully-optimized H1+H2 stack, PGD2 is the likely culprit.
Low-dose aspirin 81–325 mg daily inhibits COX-1 and blocks PGD2 synthesis. Many flushing-dominant MCAS patients report a dramatic response — dramatic enough that aspirin deserves a formal trial in this subgroup before escalating further. Start at 81 mg/day with food, titrate upward over weeks as tolerated.
The catch: roughly 15% of MCAS patients cannot take aspirin. Some have aspirin-exacerbated respiratory disease (Samter’s triad — asthma, nasal polyps, aspirin sensitivity); some have true aspirin-triggered mast cell reactions. The first dose should ideally be observed, and in high-suspicion patients a formal desensitization protocol in an allergist’s office is the safest path.
6. The “MCAS Cocktail” — Order of Introduction
The single biggest mistake in MCAS management is starting everything at once. You’ll never know which drug did what, and if you flare you won’t know what to stop. Stage the introduction one layer at a time, roughly every 1–2 weeks:
- Step 1 (Week 1–2): Cetirizine 10 mg AM + famotidine 20 mg BID. This is the MCAS floor.
- Step 2 (Week 3–4): If insufficient: increase cetirizine to 20 mg AM (or split 10 mg BID) + add hydroxyzine 10–25 mg qhs for sleep and nocturnal itching.
- Step 3 (Week 5–6): Add montelukast 10 mg qhs, especially if respiratory, sinus, or asthma-overlap symptoms are prominent. Monitor mood.
- Step 4 (Week 7–8): Consider an aspirin trial (81 mg/day, titrate) if flushing or vascular symptoms dominate and persist despite the stack.
- Step 5: If the pharmacologic stack is still inadequate, add cromolyn sodium or ketotifen — true mast cell stabilizers. See Cromolyn & Ketotifen Protocols.
- Step 6: Layer in natural stabilizers: quercetin, luteolin, vitamin C, and others — see Natural Mast Cell Stabilizers.
Give each step a real trial — 10 to 14 days minimum — before judging it. Keep a simple symptom log (1–10 scale, morning and evening) so you can see signal through the noise.
7. Morning vs Night Dosing
A well-constructed MCAS stack is not “once a day.” It is a 24-hour coverage plan with a morning side and an evening side:
- AM: non-sedating H1 (cetirizine 10–20 mg, or fexofenadine 180 mg) + first H2 dose (famotidine 20–40 mg) + aspirin (if on it).
- Noon / mid-afternoon: second H1 dose if on BID cetirizine; a mid-day famotidine if patient is on TID; this is also when most patients hit a symptom trough and benefit from quercetin.
- Evening: second H1 dose (hydroxyzine 25 mg or doxepin 10 mg) for nocturnal itching and sleep + third famotidine if needed + montelukast (the published trials used evening dosing; most benefit appears to occur overnight).
Histamine peaks cyclically; many patients notice a 2–4 AM flare window. An evening hydroxyzine or ketotifen dose specifically targets this.
8. Doubling Up Safely
Most OTC antihistamines are labeled “once daily” because the extended-release formulation maintains therapeutic serum levels for 24 hours in most people. MCAS patients are not most people. Mast cell degranulation is not a 24-hour smooth release; it’s bursty, provoked, and overwhelms steady-state receptor blockade.
The published MCAS dosing literature (Afrin, Molderings, Valent) explicitly supports dosing second-generation H1 and H2 blockers at 2–4× the OTC recommendation:
- Cetirizine: OTC 10 mg/day → MCAS 20–40 mg/day.
- Fexofenadine: OTC 180 mg/day → MCAS up to 360 mg BID.
- Famotidine: OTC 20 mg BID → MCAS 40 mg BID or TID.
This is off-label but evidence-based, standard among MCAS-literate clinicians, and not new. If your prescriber flinches, point them at the Afrin 2020 consensus-2 paper cited below.
9. Drug Interactions to Watch
- Cimetidine — CYP450 inhibitor across 1A2, 2C9, 2D6, 3A4. Interacts with warfarin, phenytoin, theophylline, many antidepressants, beta-blockers. Choose famotidine unless you have a specific reason not to.
- Hydroxyzine — prolongs the QT interval. Avoid stacking with other QT-prolonging drugs (citalopram >20 mg, ondansetron, macrolides, fluoroquinolones, methadone). An EKG is reasonable before chronic dosing in older patients.
- Doxepin — contraindicated with MAOIs; serotonin-syndrome risk with SSRIs and SNRIs (low at 10–25 mg but real). Anticholinergic load adds to any other anticholinergic in the regimen.
- Diphenhydramine — additive anticholinergic cognitive effects with tricyclics, oxybutynin, benztropine, and hydroxyzine. Cumulative anticholinergic burden is a legitimate long-term concern.
- Montelukast — induced by phenytoin, rifampin, and phenobarbital (levels drop); neuropsychiatric interactions with anything that affects mood.
10. Side Effects and How to Manage
- Drowsiness: switch the morning H1 to fexofenadine 180 mg, and confine sedating H1s (hydroxyzine, doxepin, diphenhydramine) to bedtime only.
- Dry mouth: common with hydroxyzine, doxepin, and diphenhydramine. First-generation H1s are worst. Hydration and sugar-free gum help; if disabling, reduce the dose or shift toward fexofenadine / levocetirizine.
- Urinary retention: older men with benign prostatic hyperplasia plus first-generation H1s are a predictable problem. Use second-gen H1s preferentially in this group.
- Weight gain: cetirizine produces mild appetite stimulation in some patients. Doxepin and mirtazapine produce substantially more. Track weight monthly on any new regimen.
- Neuropsychiatric effects on montelukast: depression, suicidal ideation, vivid dreams, insomnia, irritability. Boxed warning. Stop the drug at the first sign — do not try to push through.
- Rebound on abrupt cetirizine withdrawal: a recognized pruritus-rebound syndrome appears in a minority of long-term users. Taper over 1–2 weeks rather than stopping cold.
11. Pregnancy Considerations
MCAS frequently worsens or improves dramatically during pregnancy, and the usual stack needs revisiting. General guidance (pre-2015 FDA category system, still widely cited):
- Cetirizine — Category B, generally preferred.
- Loratadine — Category B.
- Diphenhydramine — Category B, acceptable for rescue.
- Famotidine — Category B.
- Montelukast — Category B.
- Hydroxyzine — contraindicated in early pregnancy; caution later.
- Doxepin — avoid in pregnancy.
- Aspirin — low-dose acceptable in some obstetric contexts but coordinate with OB.
12. Pediatric Dosing
- Cetirizine: 2.5 mg age 2–5; 5 mg age 6+; 10 mg adolescents.
- Fexofenadine: 30 mg BID (children’s formulation); 180 mg daily from ~12 yrs.
- Hydroxyzine: 0.5 mg/kg per dose; excellent pediatric data, long track record.
- Famotidine: 0.5 mg/kg BID; well tolerated.
- Montelukast: 4 mg age 2–5, 5 mg age 6–14, 10 mg 15+. Boxed warning applies; monitor behavior closely in children.
13. Cost & Access
This is one of the rare chronic conditions where the first-line medication stack is genuinely cheap. Almost nothing in the core MCAS regimen is patent-protected.
- OTC generics: cetirizine, loratadine, fexofenadine, levocetirizine, famotidine, diphenhydramine, aspirin — typically $5–$15/month each.
- Prescription generics: hydroxyzine, doxepin, montelukast, nizatidine — $5–$15/month each with GoodRx or Cost Plus Drugs.
- Xyzal (levocetirizine) is now OTC. Atarax/Vistaril (hydroxyzine) remains prescription-only.
- Total cost of a complete H1 + H2 + montelukast + aspirin stack: typically $20–$60/month even cash-pay.
The expensive pieces of MCAS management are the next layer — cromolyn sodium (Gastrocrom), ketotifen (compounded in the US), and specialty biologics like omalizumab. The antihistamine stack is not the budget problem.
14. When Stacks Aren’t Enough
If you’ve run an optimized H1 + H2 + leukotriene + aspirin stack at maximum tolerated doses for 6–8 weeks and symptoms persist, you’ve hit the ceiling of what receptor blockade alone can do. Released histamine, PGD2, and leukotrienes saturate their receptors faster than the blockers can compete.
The next layer targets mast cells themselves:
- Cromolyn sodium (Gastrocrom): inhaled or oral; prevents mast cell degranulation in the first place.
- Ketotifen: dual H1 antagonist + mast cell stabilizer; compounded in the US; widely used in Europe.
Dosing, titration, and the common GI-solution tricks live in Cromolyn & Ketotifen Protocols. Beyond that: omalizumab (Xolair), LDN, low-dose naltrexone, and in refractory cases tyrosine kinase inhibitors (imatinib, midostaurin) for biopsy-confirmed mastocytosis — these are specialist-managed.
15. Key Research Papers
- Valent P, et al. Proposed diagnostic algorithm for patients with suspected mast cell activation syndrome. Journal of Allergy and Clinical Immunology. 2019.
- Afrin LB, et al. Diagnosis of mast cell activation syndrome: a global “consensus-2.” Diagnosis. 2020.
- Molderings GJ, et al. Pharmacological treatment options for mast cell activation disease. Journal of Allergy and Clinical Immunology. 2016.
- Vitte J, Amadei L, Gouitaa M, et al. Paired acute-baseline serum tryptase levels in perioperative anaphylaxis: an observational study. Allergy. 2019;74(6):1157–1165.
16. Research Papers
Curated PubMed topic searches — each link opens a live query so you always see the most recent literature.
- PubMed: H1 + H2 antihistamines in MCAS
- PubMed: Cetirizine and mast cell activity
- PubMed: Famotidine in MCAS
- PubMed: Hydroxyzine for chronic urticaria
- PubMed: Montelukast and mast cell disease
- PubMed: Aspirin for flushing in mastocytosis
- PubMed: Doxepin for chronic urticaria
- PubMed: High-dose fexofenadine in urticaria
Connections
- MCAS Overview
- Natural Mast Cell Stabilizers
- Mediators and Symptom Map
- Cromolyn and Ketotifen Protocols
- Triggers and Flare Management
- MCAS Testing Guide
- Hereditary Alpha-Tryptasemia
- Low-Histamine Diet
- Hormone Sensitivity and Menstrual Flares
- POTS
- Allergies
- SIBO
- Asthma
- Quercetin
- Vitamin C
- Bloating
- Inflammatory Markers