MCAS Hormone Sensitivity: Estrogen & Menstrual Flares
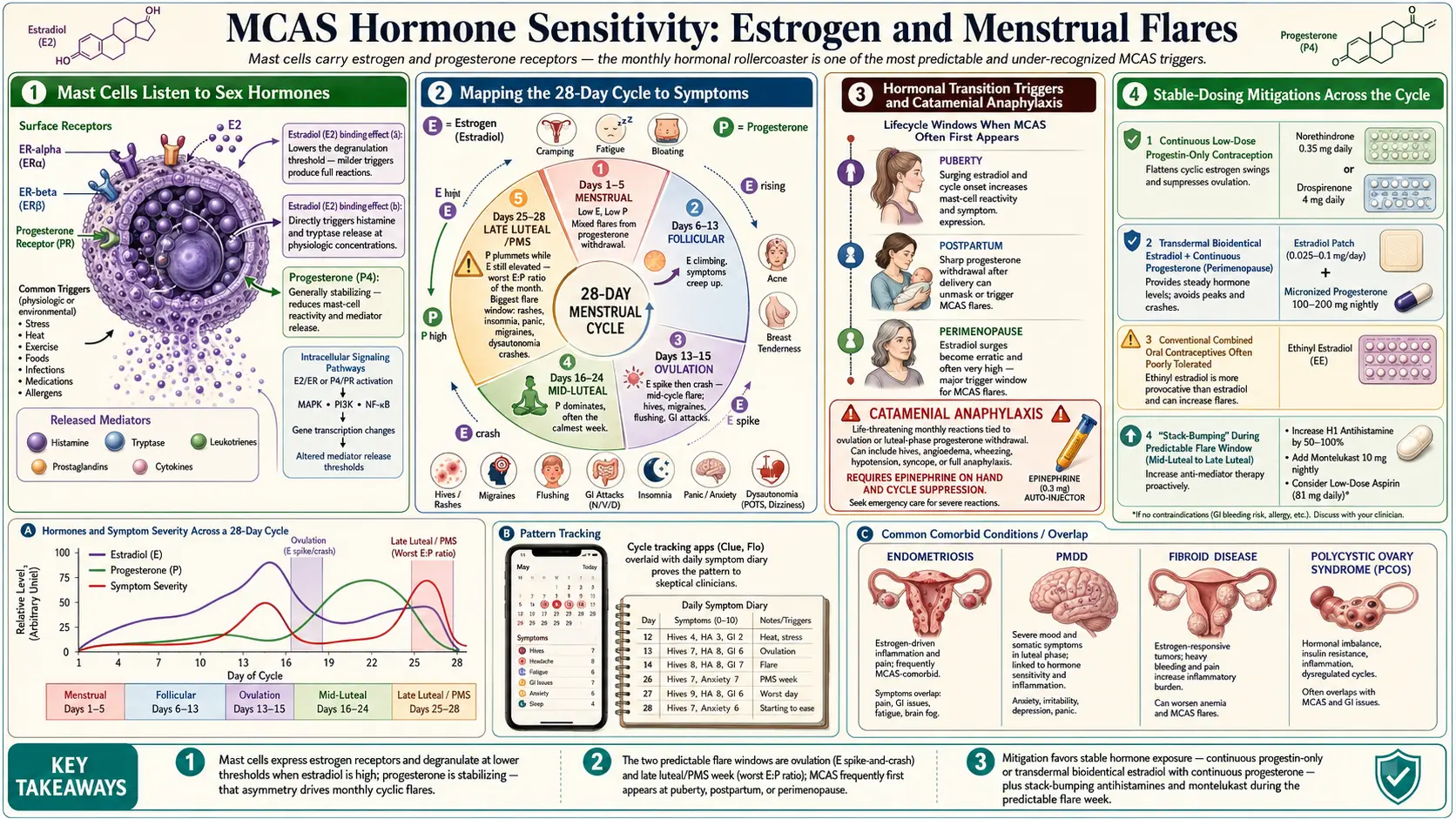
If your MCAS symptoms seem to have a rhythm — worse the week before your period, calmer during pregnancy, suddenly explosive in perimenopause — you are not imagining it. Mast cells are exquisitely tuned to sex hormones, and the monthly estrogen rollercoaster is one of the most common, most predictable, and most under-recognized triggers in mast cell disease. This page explains why, how to track it, and what actually helps.
Table of Contents
- Mast Cells Have Estrogen & Progesterone Receptors
- Mapping the Cycle to Your Symptoms
- Why MCAS Often Appears at Puberty, Postpartum, or Perimenopause
- Catamenial Anaphylaxis — When Your Period Is Life-Threatening
- Cycle Tracking: How to Prove It to Yourself (and Your Doctor)
- Contraception When You Have MCAS
- Pregnancy, Postpartum & MCAS
- Perimenopause, Menopause & MCAS-Friendly HRT
- Endometriosis & PMDD Overlap
- Practical Mitigation: Stable Dosing Across the Cycle
- Key Research Papers
- PubMed Topic Searches
- Connections
- Featured Videos
Mast Cells Have Estrogen & Progesterone Receptors
Mast cells are not passive bystanders of your hormones — they listen closely. On their surface and inside their cytoplasm they express estrogen receptors (ER-alpha and ER-beta) and progesterone receptors. When estradiol binds, it does two things that matter to you:
- It lowers the threshold for degranulation. Estradiol primes mast cells so a trigger that would normally cause mild flushing now causes a full histamine dump — hives, tachycardia, GI cramping, brain fog.
- It can directly trigger release. At physiologic concentrations, estradiol all by itself causes measurable histamine and tryptase release from human mast cells in culture (Zaitsu 2007).
Progesterone, by contrast, is generally stabilizing. It reduces mast-cell reactivity and tends to calm mediator release. This asymmetry — estrogen stirs, progesterone settles — is the physiologic engine behind almost every pattern discussed on this page.
Mapping the Cycle to Your Symptoms
A textbook 28-day cycle has two estrogen peaks and one progesterone peak. Your MCAS symptoms usually track them like this:
- Days 1–5 (menstrual phase). Both hormones are low. Some patients are relatively calm here; others flare from the sudden progesterone withdrawal that triggered the bleed.
- Days 6–13 (follicular/late follicular). Estrogen climbs. Symptoms creep up.
- Days 13–15 (ovulation). Estrogen spikes sharply, then falls. This is the mid-cycle flare window — hives, migraines, flushing, GI attacks, sometimes frank anaphylactoid reactions.
- Days 16–24 (mid-luteal). Progesterone dominates. Many MCAS patients feel their best here.
- Days 25–28 (late luteal/PMS window). Progesterone plummets while estrogen is still elevated — the estrogen-to-progesterone ratio is at its worst. This is the single biggest MCAS flare window of the month: rashes, insomnia, panic surges, migraines, diarrhea, joint pain, dysautonomia crashes.
If your worst week is the week before your period and you always thought of it as "bad PMS," it is worth reframing as a hormone-mast-cell event. The treatments are different.
Why MCAS Often Appears at Puberty, Postpartum, or Perimenopause
MCAS rarely announces itself out of nowhere. It almost always has a precipitating event, and in women and AFAB patients the precipitant is frequently a hormonal transition:
- Puberty. A nervous system that tolerated food, exercise, and stress through childhood suddenly develops hives, POTS, IBS, or migraines the year estrogen comes online.
- Pregnancy & postpartum. Some patients actually feel better while pregnant (high progesterone), then collapse in the six to twelve weeks after delivery as estrogen and progesterone both crash to near-zero. Sleep deprivation, blood loss, and immune reset pile on.
- Perimenopause. Not the low-estrogen stage people expect — it is the wildly fluctuating stage, typically ages 40–52. Estradiol can spike 2–3x normal one week and crash below postmenopausal levels the next. Mast cells, which prefer steady signals, go haywire.
- Surgical menopause. Sudden ovary removal is the most abrupt hormone cliff a body can experience and is a classic MCAS-unmasking event.
If your timeline shows a healthy decade followed by a symptom explosion that lines up with one of these transitions, hormone-driven mast cell activation is a leading suspect.
Catamenial Anaphylaxis — When Your Period Is Life-Threatening
Catamenial anaphylaxis is anaphylaxis tied directly to the menstrual cycle, classically occurring in the late luteal phase or the first days of bleeding. It was first described in the medical literature by Robinson and colleagues in 1980 and has been reported many times since. Proposed mechanisms include progesterone withdrawal, direct sensitivity to endogenous progesterone (autoimmune progesterone dermatitis), and estrogen-primed mast cell instability.
Signs that your monthly flare has crossed into catamenial anaphylaxis territory:
- Throat tightness, lip/tongue swelling, or trouble swallowing in a predictable cycle window.
- Sudden hypotension, near-syncope, or presyncope that tracks your cycle.
- Widespread urticaria plus wheeze or GI collapse cyclically.
- Need for epinephrine or ER visits that cluster around ovulation or pre-menses.
If any of this sounds like you, this is an allergist/immunologist conversation — and you should carry two epinephrine auto-injectors. Suppression of ovulation (continuous progestin, GnRH analogues) is sometimes the treatment that finally breaks the cycle.
Cycle Tracking: How to Prove It to Yourself (and Your Doctor)
Doctors are trained to be skeptical of "my symptoms are hormonal." Data wins arguments. Track for at least two full cycles, ideally three:
- Cycle day. Count day 1 as the first day of full flow.
- Symptom score 0–10 for each of: flushing, hives, GI, migraine, brain fog, POTS/tachycardia, mood.
- Ovulation marker. LH strips, basal body temperature, or a tracking app. Ovulation lets you label the luteal phase precisely.
- Medication & food held as constant as possible so the cycle pattern is isolated from lifestyle noise.
Plot the results. If peaks line up at ovulation and the late luteal window across multiple cycles, you have a defensible case for hormone-driven MCAS and a target for intervention.
Contraception When You Have MCAS
This is one of the most common questions MCAS patients ask, and the honest answer is: it depends, and you may need to try more than one.
- Combined oral contraceptives (estrogen + progestin). The daily estrogen dose, especially ethinyl estradiol, can provoke worse MCAS symptoms in sensitive patients. Some tolerate them fine, but for many the first three cycles on a combined pill are miserable.
- Progestin-only options. Low-dose progestin-only pills ("mini-pill"), the etonogestrel implant, levonorgestrel IUDs (Mirena, Kyleena, Liletta, Skyla), or DMPA (Depo-Provera) avoid the exogenous estrogen load. Many MCAS patients do noticeably better on these.
- Continuous / no-placebo regimens. Taking an active pill every day with no withdrawal week eliminates the monthly hormone crash and the flare it drives. Works with either combined or progestin-only formulations under physician guidance.
- Levonorgestrel IUD. Often well tolerated because systemic hormone levels stay low while local endometrial progestin suppresses bleeding and ovulation in a meaningful fraction of users.
- Non-hormonal. Copper IUD, barrier methods, or tubal options sidestep the hormonal question entirely — but do not address the endogenous cycle flares, which is fine if those are mild.
The contraceptive conversation with your clinician should explicitly name MCAS and mast-cell instability as the constraint. A prescriber who has never heard of MCAS will default to combined pills; one who understands it will usually start with a progestin-only or IUD approach.
Pregnancy, Postpartum & MCAS
Pregnancy is a natural experiment in high-progesterone physiology. For many MCAS patients, the first trimester is rough — nausea, histamine-intolerance foods newly intolerable, migraines — but the second and third trimesters often bring dramatic improvement. Progesterone rises roughly 10-fold by term, and the normal immune tilt of pregnancy (away from Th1, toward tolerogenic Th2/regulatory signaling) appears to quiet mast cells along with everything else.
The postpartum period is the mirror image. Within 48–72 hours of delivery, progesterone drops from its peak to nearly undetectable levels. Estrogen falls similarly. Patients who glided through the third trimester can crash in the first two weeks postpartum with hives, rage-level insomnia, skin-crawling anxiety, GI chaos, and a dysautonomia flare that overlaps with postpartum depression. This is not in your head; it is one of the most extreme hormone transitions in human physiology, and mast cells feel every pixel of it.
Planning ahead matters:
- Continue safe antihistamines. Most H1 blockers (cetirizine, loratadine) and H2 blockers (famotidine) are generally compatible with pregnancy and breastfeeding — confirm with your OB.
- Discuss cromolyn. Oral cromolyn is minimally absorbed and is often considered one of the safer mast-cell stabilizers in pregnancy. See the cromolyn and ketotifen protocols.
- Plan for postpartum. Do not taper medication in the last trimester just because you feel great. Keep the baseline stack in place and expect a bumpier postpartum than a typical patient.
- Epinephrine readiness. If you have a history of anaphylaxis, carry auto-injectors and review the anesthesia plan with your delivery team in advance.
Perimenopause, Menopause & MCAS-Friendly HRT
Perimenopause is where many women receive their first formal MCAS diagnosis, often after decades of being told their symptoms were "just anxiety." The wild estrogen swings of the 40s do two things at once: they directly destabilize mast cells, and they unmask latent sensitivities (to foods, fragrances, weather) that a younger, more stable endocrine system was masking.
The question of hormone replacement is nuanced. Done poorly, HRT makes MCAS worse. Done well, it can be stabilizing because it replaces chaotic hormonal surges with a steady baseline.
MCAS-friendlier HRT principles (discuss with a menopause-literate clinician):
- Transdermal estradiol (patch, gel, or spray) rather than oral estradiol. Bypasses first-pass liver metabolism, gives smoother blood levels, and avoids the oral-estrogen inflammatory signature.
- Lowest effective dose. Start low (e.g., a 0.025 mg/day patch) and titrate by symptoms, not a preset target.
- Oral micronized progesterone (not synthetic progestins) taken nightly, cyclically or continuously. Bioidentical progesterone is generally better tolerated by mast cells than medroxyprogesterone or norethindrone.
- Continuous, not cyclic. Daily dosing without a withdrawal bleed eliminates monthly crashes.
- Introduce slowly. Pre-medicate with your H1/H2 stack for the first two weeks. Flares in the first month do not necessarily mean you cannot tolerate HRT — they often settle as the system re-stabilizes.
For readers who want the broader menopause picture beyond MCAS, see Menopause & HRT and Perimenopause.
GnRH Agonists and Letrozole — The Nuclear Options
In refractory hormone-driven MCAS — especially with catamenial anaphylaxis, severe endometriosis, or perimenopausal flares that do not respond to anything else — clinicians occasionally reach for cycle suppression with GnRH agonists (leuprolide) or aromatase inhibitors (letrozole). Both drive estrogen to near-zero. Some patients with hormone-flare MCAS find this transformational; others tolerate the medically induced menopause poorly. These are specialist-level interventions, usually short-term, often paired with low-dose add-back HRT to protect bone.
Endometriosis & PMDD Overlap
Two conditions overlap heavily with hormone-sensitive MCAS and are worth screening for if the cycle-symptom pattern is strong.
Endometriosis. Endometriotic lesions contain elevated mast-cell densities, and the chronic pelvic pain of endometriosis is partly a mast-cell neuroinflammatory phenomenon. MCAS patients with severe cyclic pelvic pain, dyspareunia, or infertility should be evaluated for endometriosis. See Endometriosis.
PMDD (premenstrual dysphoric disorder). Severe late-luteal mood, anxiety, and somatic symptoms with a clean cycle-locked pattern. Mast cell mediators (histamine, prostaglandin D2, leukotrienes, TNF-alpha) are plausible contributors to the "PMDD" phenotype in MCAS patients, and treating the mast cell side sometimes resolves what was labeled PMDD for years.
Practical Mitigation: Stable Dosing Across the Cycle
Concrete moves you can make without a prescription change:
- Do not skip H1/H2 doses in "good weeks." The most common mistake is tapering antihistamines when you feel fine mid-luteal, then getting blindsided by a pre-menstrual flare you are no longer covered for. Hold a stable baseline (e.g., cetirizine 10 mg AM, famotidine 20–40 mg BID) every single day. See H1 & H2 blocker stacks.
- Pre-emptive bump in the late luteal window. Some clinicians add a second daily H1 (e.g., fexofenadine in the afternoon), add an H2 dose, or add ketotifen starting three to five days before expected symptom onset. See cromolyn and ketotifen protocols.
- Oral cromolyn 30–60 minutes before meals during your flare week to quiet GI mast cells in particular.
- Quercetin 500–1000 mg twice daily is a classic flavonoid mast-cell stabilizer. Many patients start it five to seven days before their expected flare.
- Luteolin (often combined with rutin) has supportive evidence for mast-cell and neuroinflammatory calming and is useful when brain fog and anxiety dominate the luteal pattern.
- Vitamin C 1–2 g/day in divided doses supports histamine catabolism.
- Tighten the low-histamine diet during the flare week even if you tolerate more foods at mid-luteal. See Low-Histamine Diet.
- Sleep discipline. Short sleep amplifies histamine-driven symptoms. The late-luteal window is the wrong week to shortchange sleep.
- Alcohol at ovulation and pre-menses is a trap. Alcohol blocks DAO (diamine oxidase), the enzyme that clears histamine, right when your estrogen is already giving mast cells the green light. Skip it.
For the broader stabilizer toolkit see Natural Mast Cell Stabilizers and Triggers & Flare Management.
Key Research Papers
- Zaitsu M, et al. Estradiol activates mast cells via a non-genomic estrogen receptor-alpha and calcium influx. Mol Immunol. 2007.
- Bonds RS, Midoro-Horiuti T. A role for sex hormones in the allergic response and asthma (context of IL work cited as 10.4049/jimmunol.1202330). J Immunol / Curr Opin Allergy Clin Immunol. 2013.
- Vasiadi M, et al. Progesterone inhibits mast cell secretion. Int J Immunopathol Pharmacol. 2006.
- Robinson PD, et al. Recurrent catamenial anaphylaxis. Early case literature. 1980.
- Reviews of mast cell estrogen receptor biology (ER-alpha, ER-beta).
- Autoimmune progesterone dermatitis case series and pathophysiology.
PubMed Topic Searches
For further reading, these PubMed searches return current peer-reviewed work on hormone-mast-cell biology, cycle-driven symptoms, and treatment:
- Estrogen and mast cell degranulation
- Progesterone and mast cell stabilization
- Catamenial anaphylaxis
- Perimenopause, histamine, and mast cells
- Pregnancy and mast cell activation syndrome
- Endometriosis and mast cells
- PMDD and histamine
- Transdermal estradiol and micronized progesterone
- GnRH agonists in catamenial anaphylaxis
- Quercetin and luteolin as mast cell stabilizers
Connections
- MCAS Overview
- Perimenopause
- Endometriosis
- Menopause and HRT
- Cromolyn and Ketotifen Protocols
- Natural Mast Cell Stabilizers
- Triggers and Flare Management
- Low-Histamine Diet
- H1 and H2 Blocker Stacks
- MCAS Testing Guide
- Hereditary Alpha-Tryptasemia
- Mediators and Symptom Map
- POTS
- Alpha-Gal Syndrome
- Anxiety
- Quercetin
- Brain Fog