MCAS Mediators and Symptom Map
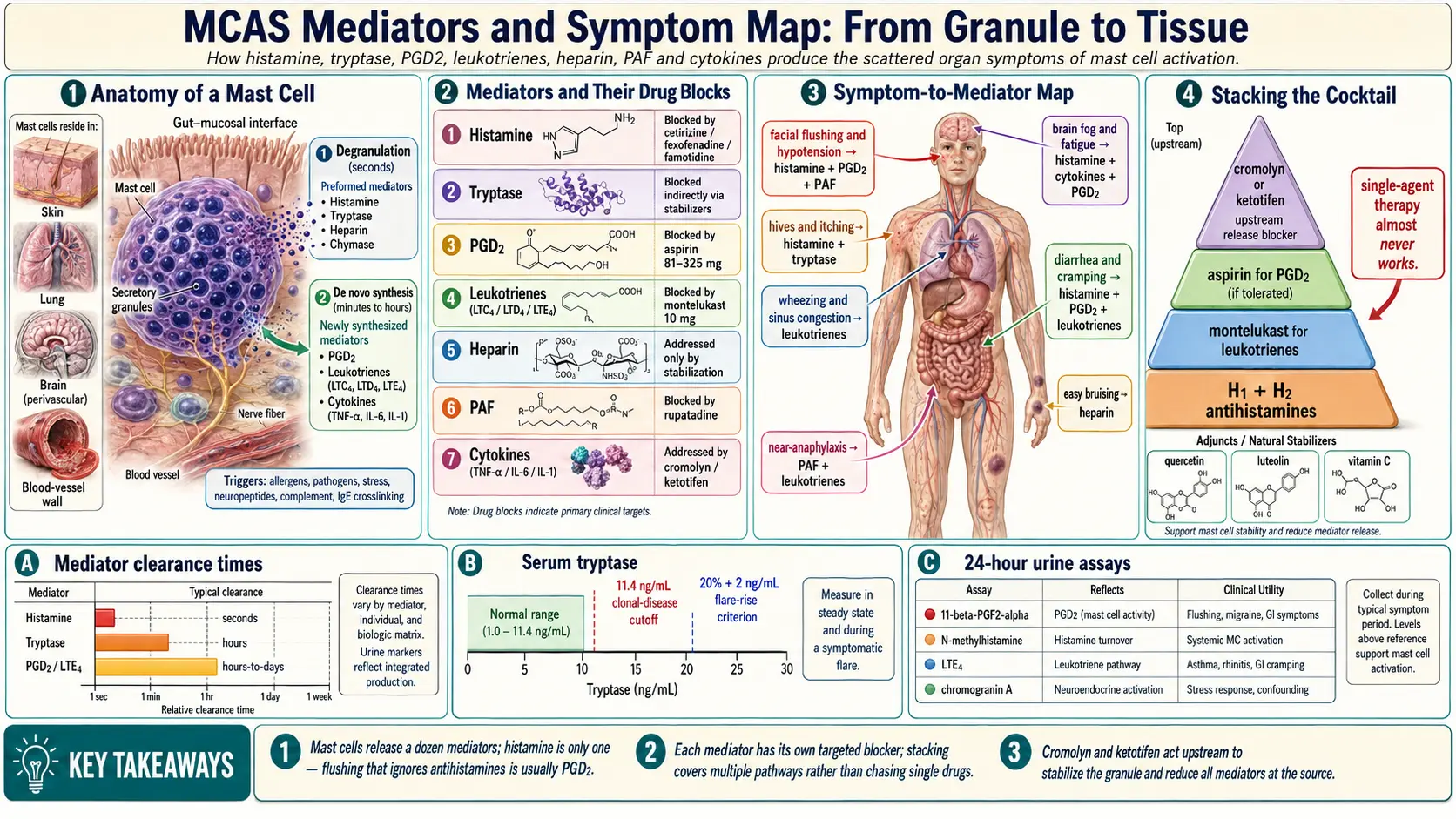
Table of Contents
- What a Mast Cell Is
- The Two Release Modes
- Histamine
- Tryptase
- Prostaglandin D2 (PGD2)
- Leukotrienes (LTC4, LTD4, LTE4)
- Heparin
- Chromogranin A
- Chymase
- TNF-alpha, IL-6, IL-1
- Platelet Activating Factor (PAF)
- A Quick Symptom-to-Mediator Map
- Why Targeting the Right Mediator Matters
- Stacking Medications to Cover Multiple Mediators
- Mediators Amplified by Hormones, Stress, Infection
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
1. What a Mast Cell Is
Mast cells are immune cells stationed at every tissue-environment interface in your body — the skin, the lining of the gut, the airways of the lungs, the nasal passages, the brain's perivascular spaces, and the walls of your blood vessels. They sit exactly where the outside world meets the inside of you, and their job is to notice threats and sound the alarm.
Each mast cell holds thousands of tiny packets called granules, pre-loaded with chemical messengers. When a mast cell is triggered, it releases those chemicals — which is how you fight off parasites, seal a wound, or mount an allergic reaction. In a healthy person, the release is proportionate to the threat.
In MCAS (Mast Cell Activation Syndrome), that release is no longer proportionate. The cells either leak mediators continuously at a low level — giving you the nagging daily symptoms many patients describe as "always running at 30% sick" — or they fire off massive bursts during flares, producing hives, flushing, brain fog, diarrhea, and near-anaphylactic drops in blood pressure all at once. Understanding which chemicals they release is the key to understanding why your symptoms look so scattered and to choosing the right medications to block them.
2. The Two Release Modes
Mast cells have two distinct ways of releasing their chemicals, and the difference explains why MCAS flares have both an immediate phase and a delayed tail.
Degranulation (seconds)
Preformed mediators already packaged inside granules are dumped out within seconds of triggering. This is the source of the immediate flare: the sudden flush, the pounding heart, the itching, the hive that erupts in under a minute. The chemicals released this way include histamine, tryptase, heparin, and chymase.
De novo synthesis (minutes to hours)
Newly manufactured mediators are assembled on demand from membrane lipids and from gene expression. These take minutes to hours to peak and are responsible for the delayed and sustained symptoms — the brain fog that sets in 90 minutes after a meal, the fatigue that hits the next day, the wheeze that builds slowly overnight. Prostaglandins, leukotrienes, and cytokines are made this way.
Most MCAS patients feel both phases — which is why a flare from one exposure can last 24 to 72 hours.
3. Histamine — the mediator most people think of
Histamine is the chemical that allergy medication ads have made famous. It is preformed, stored in granules, and released within seconds. It acts on four receptor subtypes (H1, H2, H3, H4) scattered across almost every organ.
What it does: widens blood vessels (vasodilation), makes capillaries leaky (vascular permeability), triggers itching on nerve endings, contracts smooth muscle in the lungs and gut, stimulates gastric acid secretion (H2), and modulates neurotransmitters in the brain (H3).
Symptoms driven by histamine: flushing, urticaria (hives), itching, vascular headache, runny nose, nasal congestion, GI cramping, diarrhea, bloating, gastric reflux, hypotension, palpitations, anxiety, insomnia, vertigo, and urinary urgency.
Blocked by: H1 antihistamines (cetirizine, fexofenadine, loratadine, hydroxyzine) combined with H2 antihistamines (famotidine, cimetidine). See H1 and H2 Blocker Stacks for dosing.
4. Tryptase — the most-measured marker of mast cell activity
Tryptase is a protein-cutting enzyme stored almost exclusively in mast cells, which is what makes it such a valuable diagnostic marker — if your tryptase is rising, a mast cell is almost certainly responsible.
What it does: cleaves tissue proteins, activates PAR-2 receptors on nerve endings and epithelium (triggering pain and inflammation locally), and amplifies the activity of other inflammatory molecules in its neighborhood. Chronic high tryptase is believed to contribute to small-fiber neuropathy by sensitizing peripheral nerves.
Symptoms driven by tryptase: itching, burning pain, flushing (indirect via kinin activation), bronchoconstriction, vascular permeability, neuropathic pain, and nerve-fiber sensitization.
Measured: serum total tryptase. A baseline above 11.4 ng/mL suggests clonal mast cell disease (mastocytosis or hereditary alpha-tryptasemia). A transient rise of at least 20% plus 2 ng/mL during a flare is one of the accepted criteria for MCAS. 24-hour urine is not useful for tryptase. See Testing Guide.
5. Prostaglandin D2 (PGD2) — the most underestimated driver of MCAS flares
If histamine is famous, PGD2 is the one that actually drives many of the hardest-to-treat symptoms. It is not preformed — it is synthesized on demand from arachidonic acid — so it peaks later in a flare and lasts longer.
What it does: powerful vasodilation, bronchoconstriction, sleep induction, platelet inhibition.
Symptoms driven by PGD2: facial flushing (especially the bright, warm kind that does not respond to antihistamines), hypotension, tachycardia, GI cramping, diarrhea, migraine, brain fog, and somnolence after meals (the "post-meal coma" many MCAS patients describe).
Blocked by: aspirin or other NSAIDs when tolerated. Patients described as "aspirin-responsive MCAS" are almost always PGD2-dominant — their symptoms come not from histamine but from prostaglandin synthesis, and 81–325 mg of aspirin can be transformative.
Measured: 24-hour urine 11β-PGF2α, a stable metabolite of PGD2, is the gold standard.
6. Leukotrienes (LTC4, LTD4, LTE4) — the asthma-respiratory mediators
The leukotrienes are another family of lipid mediators synthesized on demand. They are the main drivers of the respiratory and allergic-shock components of MCAS.
What they do: potent bronchoconstriction (roughly 1,000 times more potent than histamine on airway smooth muscle), mucus production, vascular permeability, and chemotaxis — they actively recruit more immune cells to the site, escalating a local reaction into a systemic one.
Symptoms driven by leukotrienes: wheezing, chest tightness, cough, chronic rhinosinusitis, nasal polyps, GI spasm, and severity of anaphylactic shock.
Blocked by: montelukast (Singulair) 10 mg daily or zafirlukast. Adding montelukast is often what breaks through stubborn chest tightness and sinus congestion that antihistamines alone cannot touch. See H1 and H2 Blocker Stacks.
Measured: 24-hour urine LTE4.
7. Heparin — the bleeding mediator
Heparin is best known as an injectable anticoagulant drug — but it is naturally made by mast cells and stored in their granules. During mast cell activation, small amounts are released locally, enough to affect clotting.
What it does: inhibits the coagulation cascade and thins blood locally.
Symptoms driven by heparin: easy bruising, prolonged bleeding after minor cuts or dental work, hemorrhagic or unusually heavy menses, and unexplained hematomas after bumps.
Heparin is not directly blocked by any MCAS medication. It is addressed by stabilizing the mast cells overall so less is released in the first place.
8. Chromogranin A
Chromogranin A is an adjunct marker — a protein that is packaged into granules alongside the main mediators and spilled out when they are released. It does not directly cause a recognizable symptom, but its level in blood rises with mast cell activity. It is also elevated in neuroendocrine tumors, so it must be interpreted alongside other markers. Useful for confirming that activation is happening when tryptase is stubbornly normal.
9. Chymase
Chymase is another protease, similar to tryptase, and drives tissue remodeling, scarring, and fibrosis over time. Patients with long-standing, untreated MCAS may develop chronic tissue changes — thickened gut walls, fibrotic skin, cardiac stiffness — traceable in part to chymase. This is why early mast-cell stabilization matters: you are not only treating today's symptoms but preventing decades of silent tissue damage.
10. TNF-alpha, IL-6, IL-1
These are the cytokines — small signaling proteins that coordinate a systemic inflammatory response. Mast cells synthesize and release them over hours following activation.
Symptoms driven by cytokines: flu-like malaise, fatigue, low-grade fever or chills, brain fog, depression, post-exertional malaise, and diffuse body aches. These are exactly the symptoms that overlap with long COVID and ME/CFS, and the cytokine pathway is thought to be a major point of overlap between those conditions and MCAS.
11. Platelet Activating Factor (PAF)
PAF is a lipid mediator that is extremely potent at low concentrations. It is a powerful vasodilator and bronchoconstrictor, and it is now recognized as the main driver of severe anaphylactic shock — the life-threatening drop in blood pressure that goes beyond what histamine alone can explain.
Patients whose reactions escalate to near-anaphylaxis despite good H1/H2 coverage often have a PAF-heavy component. Rupatadine is a second-generation antihistamine that also has anti-PAF activity and is used in some countries for this purpose.
12. A Quick Symptom-to-Mediator Map
Use this as a bookmark. When a new symptom shows up, trace it to its likely mediators, then ask which of your medications targets them.
- Flushing + hypotension → histamine + PGD2 + PAF
- Diarrhea + cramping → histamine + PGD2 + leukotrienes
- Hives + itching → histamine (primary) + tryptase
- Brain fog + fatigue → histamine + cytokines (TNF-α, IL-6) + PGD2
- Wheezing + sinus congestion → leukotrienes + histamine
- Easy bruising → heparin
- Anxiety + insomnia → histamine (H1/H3 in brain) + PGD2
- Food reactions → histamine + tryptase + leukotrienes
- Menstrual flares → estrogen amplifies release of all mediators
- Near-anaphylaxis → PAF + leukotrienes + histamine
- Chronic nerve pain → tryptase + chymase
13. Why Targeting the Right Mediator Matters
The most common MCAS disappointment is the patient who takes Zyrtec and Pepcid for three months, sees partial improvement, and assumes that is as good as it gets. Often the remaining symptoms — the flushing, the diarrhea, the wheeze — are not being driven by histamine at all.
A classic example: flushing that does not respond to H1 or H2 antihistamines is almost always PGD2-dominant and can respond dramatically to low-dose aspirin. Diarrhea that ignores antihistamines is often prostaglandin- or leukotriene-driven and may require montelukast or cromolyn, which blocks the release of all mediators rather than any single one. Wheeze without clear allergy trigger points squarely at the leukotriene pathway.
If you know which mediator is dominant, you know which drug class to add.
14. Stacking Medications to Cover Multiple Mediators
Because mast cells release many chemicals, single-agent therapy almost never works. The typical MCAS "cocktail" is built to cover each mediator class:
- H1 + H2 antihistamines — histamine on both receptor families
- Montelukast — leukotrienes
- Aspirin (when tolerated, cautiously) — PGD2 and other prostaglandins
- Cromolyn or ketotifen — release blocker; reduces all mediators at the source
- Quercetin, luteolin, vitamin C — natural stabilizers that reduce granule release and scavenge downstream oxidants
- LDN (low-dose naltrexone) — dampens glial and cytokine component
See Natural Mast Cell Stabilizers for the flavonoid-based layer that complements the prescription drugs.
15. Mediators Amplified by Hormones, Stress, Infection
Mast cells sit at the intersection of the immune, endocrine, and nervous systems, and they listen to all three. Three specific amplifiers show up in almost every patient's history:
- Estrogen — peaks during ovulation, PMS, pregnancy, and HRT amplify release of all mediators. Many women trace their MCAS onset to puberty, pregnancy, or perimenopause.
- Cortisol and adrenaline — acute stress spikes trigger degranulation. A bad phone call can set off a flare as reliably as a food trigger.
- Infections and vaccines — viral infections (EBV, influenza, SARS-CoV-2 no longer excepted in the literature) and some vaccines can acutely flare MCAS by stimulating the innate immune system that mast cells are embedded in.
Flares that track a menstrual cycle, stressful week, or recent illness are not imagined — they are the predictable output of this amplifier network.
16. Key Research Papers
- Valent P, et al. Proposed diagnostic algorithm for patients with suspected MCAS. J Allergy Clin Immunol. 2019.
- Afrin LB, et al. Diagnosis of mast cell activation syndrome: a global "consensus-2". Diagnosis (Berl). 2020.
- Weiler CR, et al. AAAAI mast cell disorders committee work group report. J Allergy Clin Immunol. 2019.
- Akin C. Mast cell activation syndromes. Immunol Allergy Clin North Am. 2018.
17. Research Papers
Open searches on PubMed for ongoing literature on each mediator class:
- Mast cell mediators and symptoms
- Histamine intolerance and MCAS
- Prostaglandin D2 and flushing
- Tryptase and mast cell activation
- Leukotriene and mast cell activation
- Heparin and mast cell
- MCAS, cytokines, and TNF
- Platelet activating factor and anaphylaxis
Connections
- MCAS Overview
- H1/H2 Blocker Stacks
- Natural Mast Cell Stabilizers
- Testing Guide
- Cromolyn and Ketotifen Protocols
- Triggers and Flare Management
- Low-Histamine Diet
- Hereditary Alpha-Tryptasemia
- Alpha-Gal Syndrome
- POTS
- SIBO
- Allergies
- Quercetin
- Brain Fog
- Fatigue
- Vitamin C
- Testing and Diagnosis
- POTS MCAS and the EDS Triad