AIP and Gluten Elimination for Hashimoto's
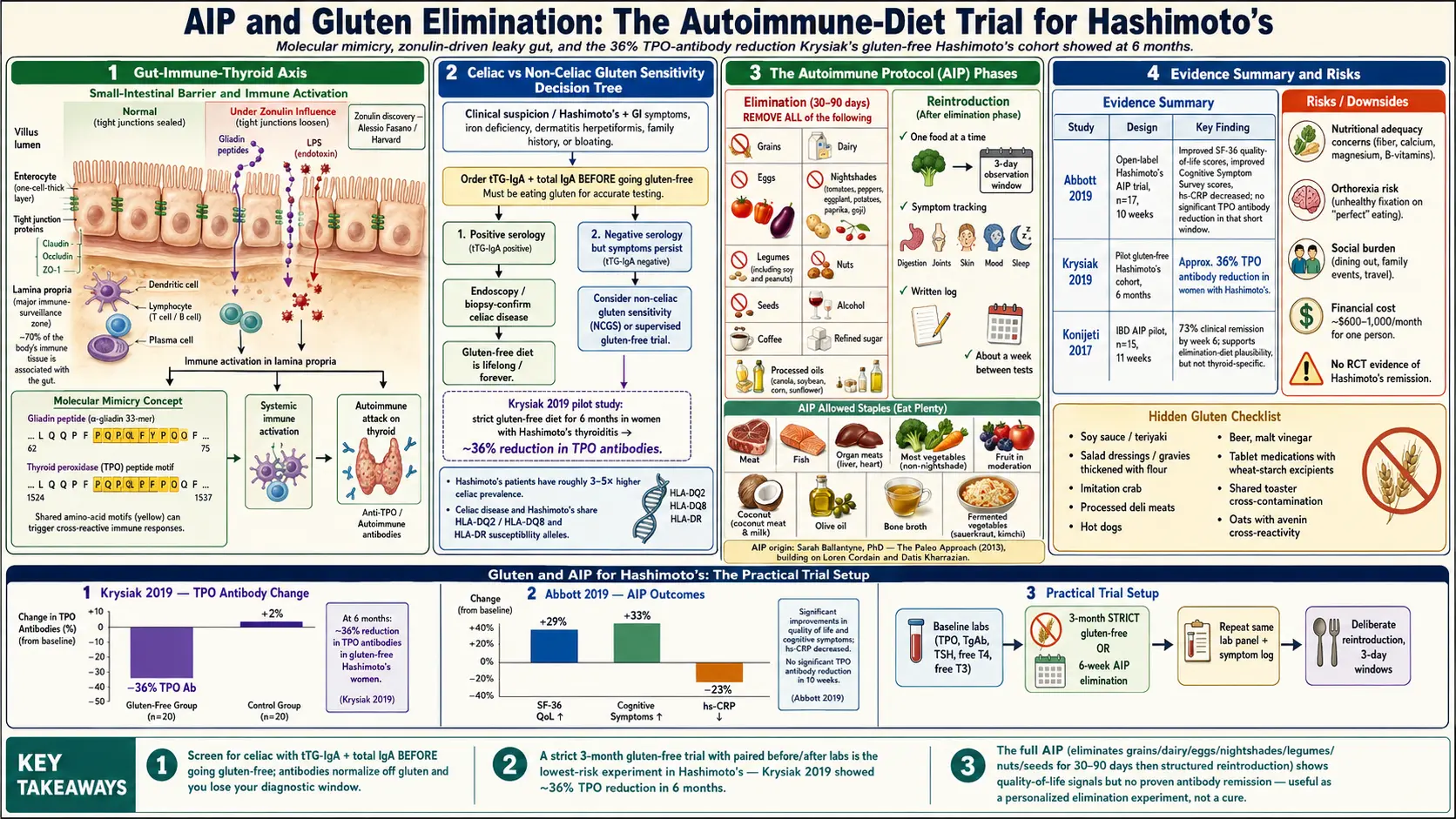
Table of Contents
- Why Diet Might Matter in Autoimmunity
- Gluten and Thyroid Molecular Mimicry
- Celiac Disease and Hashimoto's
- Non-Celiac Gluten Sensitivity and Hashimoto's
- How to Trial Gluten-Free
- Hidden Gluten Sources
- The Autoimmune Protocol (AIP)
- Who Created AIP
- Evidence for AIP in Hashimoto's
- Evidence for AIP in IBD
- Practical AIP Structure
- What AIP Foods Look Like
- What AIP Excludes
- Risks and Limits
- How to Trial Safely
- Mediterranean as a Lower-Bar Alternative
- What to Do if AIP Helps
- What to Do if AIP Doesn't Help
- Where AIP Hype Outruns Evidence
- Key Research Papers
- Connections
- Featured Videos
Why Diet Might Matter in Autoimmunity
The idea that food can drive an autoimmune disease of the thyroid sounds like a stretch — until you look at what the gut actually does. Your small-intestine lining is a single cell layer thick. On one side is the outside world (partly digested food, bacteria, viruses, fungal spores). On the other side is the largest concentration of immune tissue in your body: roughly 70% of your immune system patrols the gut. The barrier between those two worlds is held together by tight-junction proteins, and when those junctions loosen, food proteins and bacterial fragments can leak into circulation where the immune system is not used to seeing them.
This phenomenon has a scientific name — increased intestinal permeability, colloquially "leaky gut" — and a leading researcher behind it, Dr. Alessio Fasano of Harvard and Mass General. Fasano's lab discovered the human protein zonulin, which reversibly opens tight junctions. Elevated zonulin has now been documented in celiac disease, type 1 diabetes, multiple sclerosis, inflammatory bowel disease, and, in smaller studies, Hashimoto's thyroiditis. The hypothesis is not that leaky gut causes autoimmunity by itself but that it is one of three required conditions: a genetic predisposition, an environmental trigger (often a food protein or infection), and a permeable gut that lets the trigger reach immune tissue in a form the body reacts to.
The second piece of the puzzle is molecular mimicry. When the immune system is primed against a foreign protein, it can misread a similar-looking human protein as the same enemy. If a leaky gut keeps feeding the immune system food-derived peptides that happen to resemble thyroid tissue, antibodies against those peptides can spill over onto the thyroid gland itself. This is the theoretical bridge between what you eat and what your own antibodies attack.
Gluten and Thyroid Molecular Mimicry
Gluten is the protein complex in wheat, barley, and rye. Its most immune-reactive fragment is gliadin, a component that the human digestive tract does not fully break down — everyone leaves some intact gliadin peptides in the gut. Research published over the past two decades has shown that several gliadin sequences share short amino-acid motifs with thyroid peroxidase (TPO) and thyroglobulin, the two proteins that TPO and TgAb antibodies attack in Hashimoto's.
In laboratory experiments, antibodies raised against gliadin cross-react with thyroid tissue in vitro. That does not prove the same thing happens inside a living patient, but it gives the hypothesis biological teeth. Combined with an already-open gut barrier and a genetic susceptibility (HLA-DQ2 or HLA-DQ8 in celiac, and overlapping HLA risk alleles in Hashimoto's), the mimicry argument is at least mechanistically plausible.
What this does not mean: that every Hashimoto's patient has an immune reaction to gluten. It means the mechanism exists and, in a subset of patients, probably matters.
Celiac Disease and Hashimoto's
The cleanest evidence for a wheat-thyroid connection is the epidemiology of celiac disease. Celiac is a full-blown autoimmune reaction to gluten that damages the small-intestine villi. The co-prevalence with Hashimoto's is striking: people with Hashimoto's have roughly a 3- to 5-fold higher risk of also having celiac, and vice versa. The association is bidirectional, and untreated celiac can worsen thyroid antibody titers until gluten is removed.
This is why most endocrinologists and functional-medicine practitioners now recommend that every newly diagnosed Hashimoto's patient be screened for celiac disease. The standard first-line test is tissue transglutaminase IgA (tTG-IgA), with a total IgA level drawn at the same time to rule out IgA deficiency (which would make tTG-IgA falsely negative). If tTG-IgA is positive, confirmation is by upper endoscopy with duodenal biopsy.
Critical rule: do not go gluten-free before testing. Celiac serology and biopsy both require active gluten exposure to be accurate. Starting a gluten-free diet first can erase the evidence within weeks, leaving you in diagnostic limbo. Test first, then eliminate.
If celiac is diagnosed, gluten-free becomes non-negotiable and lifelong — not a trial, a treatment. Many patients in that group see their thyroid antibodies fall significantly on a strict gluten-free diet, and some recover thyroid function enough to reduce or discontinue levothyroxine (under physician guidance).
Non-Celiac Gluten Sensitivity and Hashimoto's
This is the murkier category and the one most Hashimoto's patients actually live in. Non-celiac gluten sensitivity (NCGS) describes people who test negative for celiac and for wheat allergy but nonetheless feel better off gluten. Mainstream gastroenterology has debated whether NCGS is real or mostly a response to FODMAPs in wheat, and the honest answer is that both are happening in different people.
For Hashimoto's specifically, the most-cited trial is Krysiak and colleagues (2019), a prospective study of women with Hashimoto's and euthyroid or subclinical hypothyroidism. Participants who adhered to a strict gluten-free diet for six months showed a roughly 36% reduction in TPO antibody levels and improved TSH and free T4 trends compared to controls who kept eating gluten. The study was not a double-blind RCT, the sample was modest, and it has not been independently replicated at the same scale. It is, however, the best published signal we have that gluten restriction alone can move thyroid antibody titers in Hashimoto's even without celiac.
Translation for a patient: a 3-month gluten-free trial is a reasonable experiment. The evidence is not overwhelming, but it is real, the intervention is cheap, and the downside is mostly social.
How to Trial Gluten-Free
A half-hearted gluten-free trial is almost worse than none, because a null result means nothing if you were not actually gluten-free. Rules for a meaningful trial:
- Duration — 3 months minimum. Thyroid antibodies turn over slowly. One month is not enough.
- Strict, not "mostly." A single beer, a shared cutting board, a pizza at a friend's house reintroduces enough gluten to restart the immune reaction in sensitive individuals.
- Baseline labs before starting. TPO, TgAb, TSH, free T4, free T3. Repeat the same panel at the 3-month mark so you can see the difference rather than guess it.
- Track symptoms. Keep a weekly log of fatigue, brain fog, joint aches, digestive symptoms, mood, and skin issues on a 1–10 scale.
- Reintroduce deliberately. After 3 months, reintroduce regular wheat bread or pasta for 3 days and watch the same symptom log. If symptoms flare and/or antibodies rise on follow-up labs, you have your answer.
Hidden Gluten Sources
Gluten hides in more places than any first-timer expects:
- Soy sauce, teriyaki, and many Asian marinades (wheat is the first ingredient in most soy sauce; tamari is the gluten-free version).
- Salad dressings, gravies, and soups thickened with flour.
- Imitation crab, processed deli meats, and hot dogs (wheat used as filler).
- Beer, malt vinegar, malted flavorings including many breakfast cereals.
- Medications and supplements — wheat starch is a common excipient in tablets. Ask the pharmacist or check the manufacturer's website; switch brands if needed.
- Cross-contamination at home. A shared toaster, a wooden spoon, or a knife used on regular bread is enough to transfer a crumb into your next meal. A dedicated toaster and cutting board are worth it.
- Oats. Pure oats are gluten-free, but most commodity oats are processed on the same equipment as wheat. Buy certified gluten-free oats (Bob's Red Mill, GF Harvest) and only after the first few months of the trial, since some celiac patients also react to the oat protein avenin.
The Autoimmune Protocol (AIP)
The Autoimmune Protocol, usually written AIP, is an extension of the paleo diet built specifically for autoimmune disease. It removes not only gluten but every food group suspected of driving gut inflammation or immune cross-reactivity. The elimination phase removes gluten and all other grains, all dairy, all eggs, all nightshade vegetables (tomatoes, peppers, eggplant, potatoes, goji berries), all legumes including soy and peanuts, all nuts and seeds, alcohol, coffee, refined sugar, and all processed/industrial foods. What remains is meat, fish, most vegetables, fruit in moderation, coconut, olive oil, bone broth, and fermented foods.
After an elimination period of 30 to 90 days, foods are reintroduced one at a time on a structured schedule. Reactions are logged and any food that triggers symptoms is dropped. The goal is never to stay in strict elimination permanently; it is to identify the small subset of foods that actually cause trouble for the individual and then eat a much broader diet that simply avoids those.
Who Created AIP
The protocol was systematized by Sarah Ballantyne, PhD, a medical biophysicist who developed autoimmune conditions of her own and wrote The Paleo Approach (2013), the first comprehensive AIP book. She was building on earlier clinical work by Loren Cordain, PhD on paleolithic nutrition and by Datis Kharrazian, DC, a functional-medicine practitioner whose book Why Do I Still Have Thyroid Symptoms? popularized dietary approaches to Hashimoto's in the early 2010s. Ballantyne's contribution was turning a loose set of recommendations into a structured, phased protocol with reintroduction rules. The roots of AIP are in paleo and functional medicine, not in any mainstream gastroenterology or endocrinology guideline.
Evidence for AIP in Hashimoto's
The most-cited Hashimoto's-specific AIP trial is the Abbott 2019 open-label study. Seventeen women with Hashimoto's followed the AIP elimination phase for six weeks followed by four weeks of maintenance, guided by a certified health coach and a functional-medicine nurse practitioner. At ten weeks the group showed statistically significant improvements in two standardized measures: the SF-36 quality-of-life survey and the Cognitive Symptom Survey. The inflammatory marker hs-CRP fell modestly.
The caveats are real. No control group. No placebo arm. Seventeen participants is a small cohort. Intensive coaching — which most patients will never receive — is itself a powerful intervention that a real-world self-directed attempt lacks. And, notably, the study did not show a statistically significant drop in TPO antibodies in the time frame studied. Symptoms and quality of life improved; the antibody titer did not consistently follow.
So AIP has a signal for Hashimoto's, but a modest one, in one small study, without blinding. It is a reasonable experiment, not a proven therapy.
Evidence for AIP in IBD
AIP's best trial evidence is not in thyroid disease at all but in inflammatory bowel disease. The Konijeti 2017 pilot at Scripps enrolled 15 patients with active Crohn's disease or ulcerative colitis on AIP for six weeks of elimination plus five weeks of maintenance. At week six, 73% achieved clinical remission, with improvements on endoscopy and in inflammatory markers. Again, small sample and no control group, but a dramatic effect size in a notoriously treatment-resistant population.
This matters for Hashimoto's patients because autoimmune conditions cluster. If gut inflammation is doing real damage in IBD — where we can see it directly on endoscopy — the same gut inflammation may be feeding thyroid autoimmunity in parallel.
Practical AIP Structure
- Elimination phase: 30–90 days. Most people need at least 6 weeks before symptoms shift noticeably. Ninety days is the usual upper bound before reintroduction starts.
- Reintroduction phase: one food at a time. Pick the food you miss most that is likely to be safe (typical first reintros: egg yolks, seed-based spices, ghee, then nuts, then nightshades, then dairy last).
- The 3-day window. Eat a normal portion of the test food on day one, then stop. Watch for symptoms over the next 72 hours: digestive changes, joint aches, skin flares, mood shifts, fatigue, sleep disturbance. If nothing, the food is in. If a reaction, drop it and wait a week before testing the next food.
- Log everything. Memory will lie to you. A written log is the only way to know what actually reacted.
What AIP Foods Look Like
A typical AIP day might be:
- Breakfast: salmon, sauteed greens, half an avocado, bone broth.
- Lunch: grass-fed beef, roasted carrots and beets, olive oil, sauerkraut.
- Dinner: chicken thigh, steamed broccoli, sweet potato, coconut oil.
- Snacks: berries, cucumber, olives, coconut yogurt (unsweetened, AIP-compliant brands).
Allowed staples: all meat and fish, organ meats, most vegetables (except nightshades), fruit in moderation, coconut products, olive oil, avocado oil, bone broth, fermented vegetables (sauerkraut, kimchi without chili), fresh herbs, sea salt, apple cider vinegar.
What AIP Excludes
The complete "no" list, organized the way a grocery trip feels:
- Grains: bread, pasta, rice, oats, quinoa, corn, anything with wheat, barley, or rye.
- Dairy: milk, cheese, yogurt, butter, cream (ghee is reintroduced first, often tolerated).
- Eggs: both yolks and whites during elimination; yolks usually reintroduced first and often fine.
- Nightshades: tomatoes, bell peppers, hot peppers, eggplant, white potatoes, paprika, cayenne.
- Legumes: beans, lentils, peas, peanuts, soy (tofu, tempeh, soy sauce, edamame).
- Nuts and seeds: almonds, cashews, walnuts, chia, flax, sesame (and seed-based spices like cumin and coriander).
- Other: coffee, alcohol, wine, chocolate, refined sugar, artificial sweeteners, processed oils (canola, soy, corn, sunflower).
Reading that list in one sitting is when most people panic. It is genuinely restrictive.
Risks and Limits
AIP is not risk-free. Honest warnings:
- Nutritional adequacy. Cutting grains, legumes, dairy, eggs, nuts, and seeds simultaneously drops fiber, calcium, magnesium, and certain B vitamins. Without careful planning, deficiencies follow within months.
- Orthorexia risk. AIP rewards rule-following and vigilance. In people prone to disordered eating, the protocol can calcify into an obsessive, fear-based pattern that persists long after any health benefit. Signs: dreading restaurants, cutting social events, panic when "forbidden" food is accidentally consumed, shrinking safe-food list over time rather than growing it.
- Not evidence-based for cure. There is no RCT showing AIP puts Hashimoto's in remission. The best studies are small, open-label, and measure quality of life more than hard disease markers.
- Social cost. Weddings, work lunches, travel, holidays. Over 90 days this is manageable; as a lifestyle, it isolates people.
- Financial cost. Grass-fed meat, wild fish, organic produce, coconut products, and specialty AIP-compliant packaged foods add up. A realistic monthly grocery budget on strict AIP for one person is often $600–$1,000, noticeably higher than an omnivorous baseline.
How to Trial Safely
- Work with a registered dietitian familiar with AIP — ideally one certified through the Institute for Functional Medicine (IFM) or listed in Dr. Sarah Ballantyne's practitioner directory. A week of menu planning with an RD prevents most of the nutrition pitfalls.
- Baseline labs at day zero: TPO, TgAb, TSH, free T4, free T3, 25-hydroxy vitamin D, ferritin, serum iron, vitamin B12, and a comprehensive metabolic panel. Repeat the same panel at 90 days.
- Do not stay in strict elimination forever. The elimination phase is diagnostic, not a lifestyle. The target endpoint is the broadest diet you can eat without symptoms, not the narrowest.
- Stop if you are deteriorating. Weight loss you did not want, new hair loss, worsening fatigue, or menstrual changes are signals to stop and regroup with your clinician, not to cut more foods.
Mediterranean as a Lower-Bar Alternative
Before jumping into AIP, it is worth asking whether a Mediterranean-style diet gets you most of the benefit at a fraction of the restriction. Mediterranean patterns — abundant vegetables, fruit, legumes, fish, olive oil, moderate whole grains, minimal processed food and refined sugar — are among the best-studied anti-inflammatory diets in medicine, with RCT evidence for cardiovascular disease, rheumatoid arthritis, and cognitive outcomes.
For many Hashimoto's patients, dropping ultra-processed food, seed oils, and refined sugar while adding fatty fish three times a week delivers the majority of the inflammation-lowering effect that full AIP aims for — without nightshade, legume, nut, seed, or coffee restriction. If Mediterranean plus gluten-free is not enough after 3 months of honest trial, then AIP is a reasonable next step.
What to Do if AIP Helps
Success on AIP means the elimination phase cleared symptoms and reintroductions identified specific foods that flare you. The long-term plan is not permanent strict AIP. It is a personalized post-AIP diet, typically:
- Gluten out permanently (for most Hashimoto's patients who respond).
- One to three specific individual triggers out (for many people: dairy, or eggs, or nightshades, almost never all of them).
- Everything else back on the plate. Grains, legumes, nuts, seeds, coffee, chocolate, wine — whatever did not react gets reintroduced.
- Repeat labs at 6 and 12 months to confirm antibody trends and catch any drift.
What to Do if AIP Doesn't Help
If you complete a genuine 90-day AIP elimination with careful reintroduction and neither symptoms nor labs improve, accept the result honestly. Dietary approaches did not move the needle for you. That is a legitimate answer, not a personal failure. Shift energy to the interventions that have stronger evidence in Hashimoto's:
- Selenium 200 mcg/day (see the selenium article) — the one supplement with RCT-level evidence for lowering TPO antibodies.
- Low-dose naltrexone (LDN) — off-label, 1.5–4.5 mg at bedtime, growing evidence in multiple autoimmune conditions.
- Optimizing thyroid hormone replacement — levothyroxine dose refinement, adding T3 or switching to NDT if symptoms persist on adequate T4 alone (see levo vs NDT vs T3).
- Sleep, stress, and resistance training — unglamorous and non-negotiable.
Where AIP Hype Outruns Evidence
AIP is a useful diagnostic tool for finding personal food triggers. It is not a cure for Hashimoto's, and no credible evidence supports the claim that it puts the disease into lasting remission in most people. You will see blog posts, social-media testimonials, and book jackets implying otherwise — read them with a skeptical eye.
What is fair to say: some Hashimoto's patients see meaningful antibody reductions, symptom improvement, and quality-of-life gains on AIP or on gluten-free alone. That is worth something, especially when conventional care leaves symptoms on the table despite "normal" labs. What is not fair to say: that food alone fixes the disease for everyone, or that remaining on thyroid hormone replacement means you did the diet wrong. Hashimoto's is a chronic autoimmune condition of the thyroid gland; diet is one lever of several, and for most people the levothyroxine stays on the shelf.
Key Research Papers
Foundational and recent peer-reviewed publications on gluten, the autoimmune protocol diet, intestinal permeability, and the Hashimoto's-celiac overlap. Each citation links to the full text via DOI.
- Krysiak R, Szkróbka W, Okopień B. The Effect of Gluten-Free Diet on Thyroid Autoimmunity in Drug-Naïve Women with Hashimoto's Thyroiditis: A Pilot Study. Experimental and Clinical Endocrinology & Diabetes. 2019;127(7):417–422.
- Abbott RD, Sadowski A, Alt AG. Efficacy of the Autoimmune Protocol Diet as Part of a Multi-Disciplinary, Supported Lifestyle Intervention for Hashimoto's Thyroiditis. Cureus. 2019;11(4):e4556.
- Konijeti GG, Kim N, Lewis JD, et al. Efficacy of the Autoimmune Protocol Diet for Inflammatory Bowel Disease. Inflammatory Bowel Diseases. 2017;23(11):2054–2060.
- Fasano A. Zonulin and Its Regulation of Intestinal Barrier Function: The Biological Door to Inflammation, Autoimmunity, and Cancer. Physiological Reviews. 2011;91(1):151–175.
- Sategna-Guidetti C, Volta U, Ciacci C, et al. Prevalence of Thyroid Disorders in Untreated Adult Celiac Disease Patients and Effect of Gluten Withdrawal: An Italian Multicenter Study. American Journal of Gastroenterology. 2001;96(3):751–757.
- Lerner A, Jeremias P, Matthias T. Gut-Thyroid Axis and Celiac Disease. Endocrine Connections. 2017;6(4):R52–R58.
- Caturegli P, De Remigis A, Rose NR. Hashimoto Thyroiditis: Clinical and Diagnostic Criteria. Autoimmunity Reviews. 2014;13(4-5):391–397.
- Hollowell JG, Staehling NW, Flanders WD, et al. Serum TSH, T4, and Thyroid Antibodies in the United States Population (NHANES III). Journal of Clinical Endocrinology & Metabolism. 2002;87(2):489–499.
- Knezevic J, Starchl C, Tmava Berisha A, Amrein K. Thyroid–Gut-Axis: How Does the Microbiota Influence Thyroid Function? Nutrients. 2020;12(6):1769.
- Ch'ng CL, Jones MK, Kingham JG. Celiac Disease and Autoimmune Thyroid Disease. Clinical Medicine & Research. 2007;5(3):184–192.
- Volta U, Tovoli F, Cicola R, et al. Serological Tests in Gluten Sensitivity (Nonceliac Gluten Intolerance). Journal of Clinical Gastroenterology. 2012;46(8):680–685.
- Gärtner R, Gasnier BCH, Dietrich JW, Krebs B, Angstwurm MWA. Selenium Supplementation in Patients with Autoimmune Thyroiditis Decreases Thyroid Peroxidase Antibodies Concentrations. Journal of Clinical Endocrinology & Metabolism. 2002;87(4):1687–1691.
Live PubMed Searches
Live PubMed queries that update as new papers are indexed.
- PubMed: gluten-free Hashimoto's
- PubMed: autoimmune protocol diet
- PubMed: zonulin leaky gut autoimmune
- PubMed: celiac disease + Hashimoto's
- PubMed: gliadin thyroid molecular mimicry
- PubMed: non-celiac gluten sensitivity
- PubMed: Mediterranean diet + autoimmune thyroid
- PubMed: elimination diet orthorexia
Connections
- Hashimoto's Thyroiditis Overview
- Levothyroxine vs NDT vs T3
- Selenium, Zinc, Iron, and Iodine
- TSH, T4, and T3 Optimal Ranges
- TPO and TgAb Antibodies
- Reverse T3 and Low T3 Syndrome
- Pregnancy and Hashimoto's
- Hashimoto, POTS, and MCAS Overlap
- Gut-Thyroid Axis and Intestinal Permeability
- Celiac Disease
- Ulcerative Colitis
- Crohn's Disease
- Selenium
- Iron
- Elimination Diet
- Gut Healing
- Fatigue
- Thyroid Disorders