TPO and TgAb Antibodies Explained
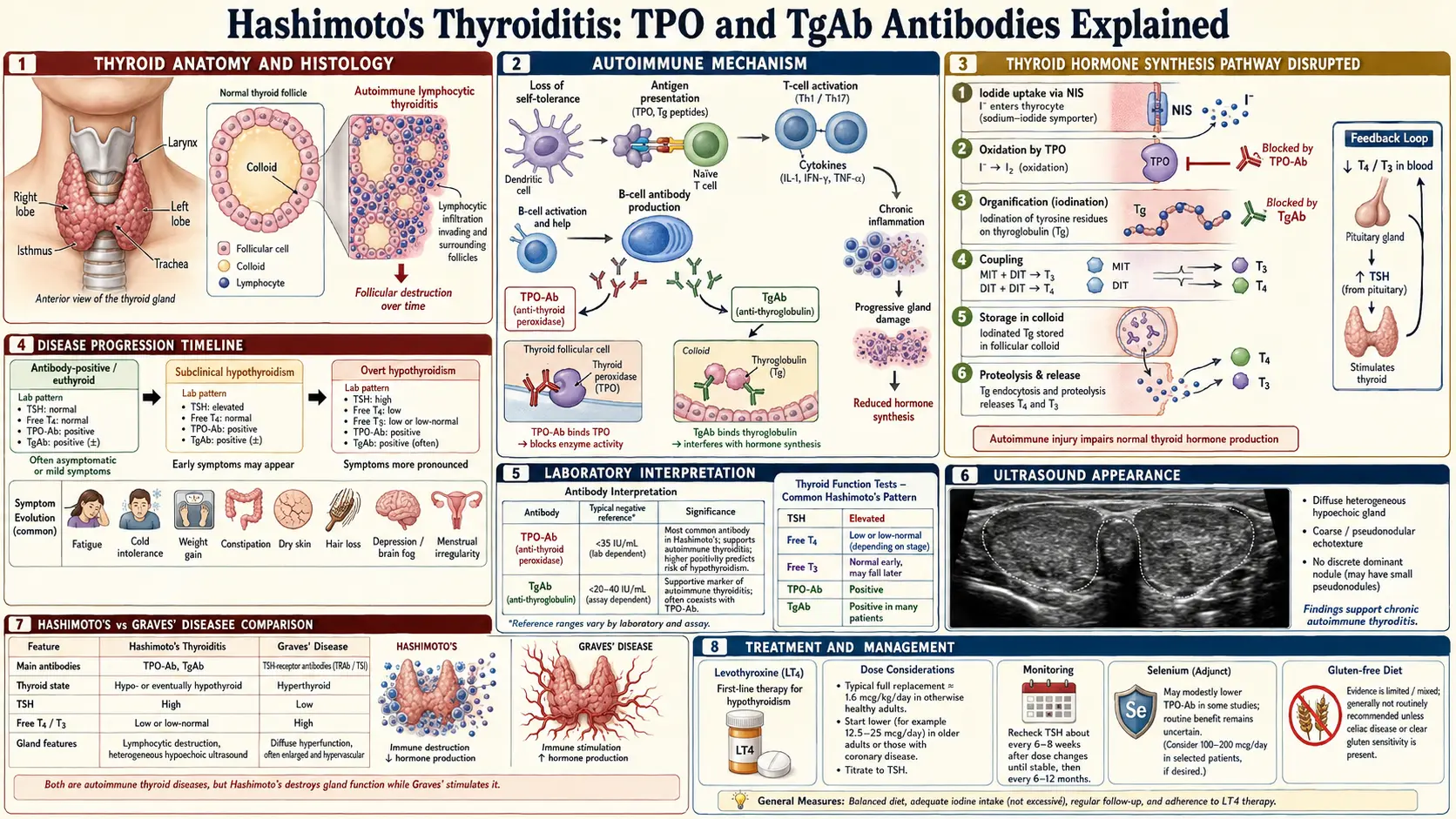
Table of Contents
- What TPO Is and What Anti-TPO Antibodies Mean
- What Thyroglobulin Is and What TgAb Means
- How They're Measured and Reference Ranges
- Prevalence in the General Population
- Positive Antibodies with a Normal TSH
- Severity, Titers, and Trajectory Over Time
- Other Conditions with Positive TPO
- Can Antibody Levels Be Lowered?
- What Doesn't Reliably Lower Antibodies
- Should You Retest Antibodies?
- Why Zero Isn't Always Possible or Necessary
- TgAb's Special Role in Thyroid Cancer Monitoring
- Antibody-Negative Hashimoto's
- What to Do with a Positive Result
- Key Research Papers
- Connections
- Featured Videos
What TPO Is and What Anti-TPO Antibodies Mean
Thyroid peroxidase (TPO) is an enzyme anchored to the inner surface of the thyroid follicle cell. Its job is to take the iodide your thyroid has absorbed from the bloodstream, oxidize it, and stitch it onto a large protein scaffold called thyroglobulin. That iodination step is how your thyroid actually manufactures the hormones T4 and T3. Without TPO, no hormone gets made.
Because TPO sits at the hinge of hormone production, the immune system pays close attention to it. In Hashimoto's thyroiditis, B cells start producing antibodies that recognize TPO as if it were a foreign invader. These anti-TPO antibodies (often written "TPOAb" or "anti-TPOab") are a marker of autoimmune damage. They are not just a passive flag — they can fix complement and help recruit cytotoxic T cells to destroy thyroid follicles over years and decades.
A positive TPO antibody test tells you three things at once: your immune system has targeted the thyroid, the gland is being gradually attacked, and you are at elevated risk of eventual hypothyroidism. It does not, by itself, tell you whether your thyroid hormone levels are currently normal, low, or high. That requires a separate TSH and free T4 measurement.
What Thyroglobulin Is and What TgAb Means
Thyroglobulin (Tg) is the massive storage protein — about 660,000 daltons — that fills the colloid inside each thyroid follicle. Think of it as the warehouse where newly made thyroid hormone is kept until the body calls for it. TPO iodinates specific tyrosine residues on thyroglobulin, and the resulting iodinated residues are later cleaved and released as T4 and T3.
Anti-thyroglobulin antibodies (TgAb or anti-Tg) target this warehouse protein. They are the second classical Hashimoto's antibody and are present in roughly 60–80% of Hashimoto's patients, often alongside TPO antibodies. A minority of patients (around 5–10%) are TgAb-positive but TPO-negative.
TgAb is generally considered a less specific marker than TPO. It is more common in healthy people without thyroid disease, and its titer tracks less cleanly with disease severity. But it matters for two reasons: it adds diagnostic confidence when TPO is borderline, and — critically — it interferes with thyroglobulin blood testing used to monitor thyroid cancer after surgery. More on that later.
How They're Measured and Reference Ranges
Both antibodies are measured on a simple blood draw using immunoassay platforms (most commonly chemiluminescent immunoassays on Roche, Abbott, Siemens, or Beckman analyzers). Each platform reports in international units per milliliter (IU/mL), but the assays are not standardized to each other. A TPO of 120 IU/mL on one lab's platform may correspond to a different number on another lab's platform. This matters if you switch labs between tests — always compare to the reference range printed on your current report, not to an old number from a different lab.
Typical upper limits of normal:
- TPO antibodies: usually <9 IU/mL, though some labs cut off at <35 or <60 IU/mL depending on the assay.
- TgAb: usually <1 IU/mL, though cutoffs of <4 or <20 IU/mL are also common.
Values are often reported as ranges: "low positive" (mildly above cutoff), "positive" (clearly elevated), and "strongly positive" (hundreds to thousands of IU/mL). Titers in the thousands are common in established Hashimoto's and are not, by themselves, worse than titers of a few hundred — both predict the same autoimmune process. A cost of $40–$120 per antibody test is typical in the U.S., and most insurers cover them when ordered with a TSH as part of a thyroid workup.
Prevalence in the General Population
Thyroid autoantibodies are startlingly common in healthy-looking people. The NHANES III population study of more than 17,000 Americans without known thyroid disease found TPO antibodies in about 13% of adults and TgAb in about 11%. Roughly 10% of adults have low-positive TPO titers and never develop clinical thyroid disease in their lifetime.
Several patterns are consistent across large cohorts:
- Women are roughly three times more likely than men to be TPO-positive. Prevalence climbs from about 5% in young adult women to 20%+ in women over 60.
- Prevalence rises with age in both sexes, reflecting the general drift of the aging immune system toward autoreactivity.
- Ethnicity matters. TPO positivity is highest in non-Hispanic white cohorts and lower in Black Americans, with intermediate rates in Hispanic and Asian American groups.
- Iodine-replete areas (including the U.S.) have higher antibody prevalence than mildly iodine-deficient regions — one of several observations linking iodine load to Hashimoto triggering.
The practical meaning: finding a positive TPO does not diagnose disease. It places you in a large, mostly healthy group that needs monitoring, not alarm.
Positive Antibodies with a Normal TSH
This is the most common real-world scenario: TSH in range, free T4 normal, TPO positive. Labs sometimes call this "euthyroid autoimmune thyroiditis" or euthyroid Hashimoto's. The gland is under attack but still compensating.
The data on what happens next are consistent across decades of follow-up:
- Roughly 3–5% per year of TPO-positive, euthyroid patients progress to overt hypothyroidism.
- Another fraction first passes through subclinical hypothyroidism (TSH elevated, free T4 still normal) before symptoms become unmistakable.
- The higher the TSH at baseline (even within the "normal" range, say above 2.5 mIU/L), and the higher the TPO titer, the faster progression tends to be.
- A small minority of patients regress — antibodies fall, TSH stabilizes, and they never develop clinical disease.
If you are in this group, the rational plan is annual TSH and free T4 monitoring, symptom awareness (fatigue, cold intolerance, weight gain, brain fog, hair thinning), and a decision about whether to trial a few lifestyle interventions discussed below. You do not need treatment for antibody positivity alone.
Severity, Titers, and Trajectory Over Time
Patients often ask whether a TPO of 900 is "worse" than a TPO of 200. The short answer: sort of, but not as much as you might expect.
At the population level, higher titers correlate with faster progression to hypothyroidism, more ultrasound heterogeneity (the classic hypoechoic, patchy Hashimoto's pattern), and a higher chance of needing levothyroxine within a given follow-up window. At the individual level, the signal is noisy. Many people with titers of 2,000+ IU/mL stay euthyroid for years. Many with titers of 60 IU/mL progress within 18 months.
Titers also fluctuate, sometimes dramatically, over years. A drop from 1,200 to 400 is not proof that an intervention worked — it may be natural variability, assay drift, or a change in the autoimmune activity of the moment. A single-timepoint change should never be treated as definitive. Trends over multiple measurements on the same assay tell you more.
Other Conditions with Positive TPO
Hashimoto's is the headline association, but TPO antibodies appear in a broader cluster of conditions:
- Graves' disease — roughly 50% of Graves' patients have positive TPO, even though the driver of hyperthyroidism is a different antibody (TSH receptor antibody, TRAb). TPO positivity in Graves' predicts a higher risk of eventually swinging into hypothyroidism after treatment.
- Postpartum thyroiditis — up to 50% of women who are TPO-positive in pregnancy develop a transient thyroiditis in the first year after delivery, often biphasic (hyperthyroid then hypothyroid).
- Type 1 diabetes — around 20–25% of type 1 diabetics carry TPO, reflecting polyglandular autoimmunity.
- Celiac disease — significantly elevated TPO positivity; celiac patients also have higher Hashimoto's risk.
- Other autoimmune conditions — rheumatoid arthritis, Sjögren's, lupus, pernicious anemia, Addison's disease, vitiligo, and autoimmune hepatitis all cluster with positive TPO above general-population rates.
- Unexplained infertility and recurrent miscarriage — TPO positivity is associated with higher miscarriage risk even in euthyroid women, which is why many reproductive endocrinologists screen for it.
Can Antibody Levels Be Lowered?
Yes, modestly, for some people, with specific interventions. No intervention reliably drives antibodies to zero. Here is the evidence, in rough order of strength:
Selenium 200 mcg/day
The best-studied supplement for Hashimoto's. In the landmark Gärtner 2002 RCT, 200 mcg/day of selenium (as selenite) for 3 months reduced mean TPO titer by roughly 40% versus placebo in women with Hashimoto's, with a subset normalizing. Multiple follow-up trials and the Toulis meta-analysis confirmed a real but modest effect: titers typically fall 20–40% over 3–6 months in responders. Selenium does not change TSH or cure disease, and benefits plateau or reverse if you stop. Selenomethionine and selenite both work; selenomethionine is better tolerated. Do not exceed 400 mcg/day long-term — chronic high-dose selenium increases type 2 diabetes risk and can cause selenosis (hair loss, brittle nails, garlic breath).
Gluten-free diet
The Krysiak 2019 RCT in euthyroid Hashimoto's women without celiac disease found that 6 months of a strict gluten-free diet reduced mean TPO titer by about 36%, with associated improvements in 25-hydroxyvitamin D. Observational cohorts show similar patterns, and Hashimoto's patients with undiagnosed celiac frequently see larger drops. A 90-day strict trial is a reasonable self-experiment; "mostly gluten-free" doesn't reproduce the signal in the data.
Myo-inositol plus selenium
Small Italian studies (Nordio, Pace, others) using myo-inositol 600 mg + selenium 83 mcg twice daily have reported TPO reductions and TSH improvements in subclinical hypothyroid Hashimoto's. Sample sizes are small and industry-sponsored, but the combination is safe and inexpensive. Reasonable to trial for 6 months if you're already taking selenium without benefit.
Vitamin D correction
Observational data link low 25-hydroxyvitamin D to higher TPO titers. Correcting deficiency (aiming for 40–60 ng/mL) is associated with modest TPO reductions in several small trials. The effect size is smaller than selenium or gluten-free, but the intervention is cheap, safe, and has many independent health benefits.
Low-dose naltrexone (LDN)
LDN at 1.5–4.5 mg nightly has a growing anecdotal following in autoimmune disease, including Hashimoto's. Patient-reported symptom improvement is common; antibody data are limited to case series and small retrospective cohorts. Not first-line, but a reasonable third-line option with a supportive prescriber.
Statins
Observational studies and a few small trials report that statin therapy lowers TPO titers, possibly via anti-inflammatory/immunomodulatory effects. Nobody prescribes statins for Hashimoto's alone, but if you are already on one for cardiovascular reasons, expect this as an incidental benefit.
What Doesn't Reliably Lower Antibodies
- Iodine supplementation. In established Hashimoto's, extra iodine (kelp, high-dose supplements, iodine-rich "thyroid support" blends) often raises antibodies and can precipitate hypothyroidism. Stay at RDA levels (150 mcg/day for adults, 220 mcg in pregnancy) from diet or a standard prenatal. Avoid high-dose iodine protocols.
- Generic "anti-inflammatory" eating without a specific elimination. Swapping soda for green smoothies while still eating gluten produces little measurable antibody change.
- Most supplement stacks — turmeric, ashwagandha, reishi, NAC — have no controlled evidence of lowering TPO. Some may help symptoms; none reliably moves the antibody number.
- Levothyroxine in euthyroid patients. Starting thyroid hormone when TSH is normal does not lower antibodies and is not standard of care.
Should You Retest Antibodies?
Less than you probably think. Once the diagnosis is confirmed, antibody retesting has limited value in routine care because the number does not change management of hormone levels. Clinical decisions are driven by TSH, free T4, and symptoms.
The reasonable retesting situations:
- Monitoring response to an intervention (selenium, gluten-free, LDN). Retest at baseline and again at 6 months on the same assay/lab.
- Pregnancy planning and early pregnancy — TPO status guides risk of postpartum thyroiditis and miscarriage, and often changes thyroid hormone targets.
- Diagnostic uncertainty — borderline first result, ambiguous imaging, or a confusing clinical picture.
Otherwise, once you know you have Hashimoto's, annual antibody measurement is optional. Annual TSH and free T4 are not.
Why Zero Isn't Always Possible or Necessary
Some patients set a goal of driving TPO into the undetectable range. For most people, this is neither achievable nor necessary. The autoimmune process, once established, tends to smolder for life at some baseline level. The clinically meaningful goals are different:
- Symptom control — feel well on whatever combination of hormone replacement and lifestyle works for you.
- Preserving remaining thyroid tissue — slowing progression to full gland failure, which extends the years you can live on lower doses or on no medication at all.
- Reducing flare frequency — periods of higher antibody activity often correlate with fatigue, hair loss, and anxiety even when TSH is in range.
- Lowering related-disease risk — addressing the autoimmune milieu may reduce the risk of picking up a second autoimmune condition.
A stable TPO of 150 on no treatment, with normal TSH and no symptoms, is a better outcome than a TPO of 10 achieved by an unsustainable protocol. Chase the downstream, not the number.
TgAb's Special Role in Thyroid Cancer Monitoring
After surgery and radioiodine ablation for differentiated thyroid cancer, doctors follow serum thyroglobulin as a tumor marker. A rising Tg after total thyroidectomy means residual or recurrent cancer. But TgAb interferes with the thyroglobulin immunoassay: when TgAb is present, it binds Tg in the sample tube and the assay falsely reports Tg as lower than it really is. A patient with undetectable Tg but positive TgAb may actually have active disease.
This is why cancer follow-up labs always include TgAb alongside Tg, and why persistent or rising TgAb after thyroidectomy can itself be a surrogate marker of disease recurrence. If you are a thyroid cancer survivor who also has coexisting Hashimoto's, ask whether your lab uses an LC-MS/MS-based thyroglobulin assay, which is less affected by antibody interference.
Antibody-Negative Hashimoto's
Roughly 5–10% of patients with biopsy-proven or imaging-proven Hashimoto's are antibody-negative. Their clinical picture looks like Hashimoto's — progressive hypothyroidism, a firm or heterogeneous thyroid on exam, the classic hypoechoic pseudonodular ultrasound pattern — but TPO and TgAb are both negative or borderline. Possible reasons include antibodies directed at non-measured epitopes, purely cell-mediated disease without significant antibody production, or low-titer antibodies below the assay threshold.
If symptoms, TSH, and ultrasound all point to Hashimoto's but antibodies are negative, the diagnosis can still be made on the clinical and imaging picture. Treatment is identical. Do not let a negative antibody panel override a strong clinical story.
What to Do with a Positive Result
A pragmatic sequence after a newly discovered positive TPO or TgAb:
- Get a full thyroid panel. TSH, free T4, and free T3 at minimum. Reverse T3 is optional — see the reverse T3 article. Thyroid ultrasound is reasonable once to document baseline architecture.
- Decide the monitoring cadence. If TSH is normal, repeat TSH and free T4 annually — sooner if symptoms develop. If TSH is subclinical-high or you're pregnant/planning pregnancy, monitor more often.
- Trial selenium 200 mcg/day (selenomethionine preferred) for 3–6 months, with baseline and end-of-trial TPO measured on the same assay. Don't exceed 400 mcg/day long-term.
- Trial a strict 90-day gluten-free diet. Strict, not casual. Consider celiac screening (tTG-IgA with total IgA) before starting, since a gluten-free diet invalidates celiac testing.
- Correct the common deficiencies. Check 25-hydroxyvitamin D (aim 40–60 ng/mL), ferritin (aim 50–100 ng/mL), and B12 (upper half of range). See Selenium, Zinc, Iron, Iodine for dosing guidance.
- Avoid iodine megadoses. Stay at RDA from diet or a standard prenatal.
- Manage the stressors that feed autoimmune flares — sleep, stable blood sugar, consistent exercise, and stress regulation all matter at the margins.
- If you convert to overt hypothyroidism, revisit levothyroxine vs NDT vs T3 and the optimal-range discussion in TSH, T4, T3 Optimal Ranges.
The overarching message: a positive antibody result is information, not an emergency. Most people with positive TPO live decades before — or without ever — needing thyroid hormone replacement. The actions above slow the process where they can and make sure the inflection point doesn't sneak up on you.
Key Research Papers
Foundational and recent peer-reviewed publications on thyroid autoantibodies, their epidemiology, diagnostic significance, and the interventions studied for lowering them. Each citation links to the full text via DOI.
- Caturegli P, De Remigis A, Rose NR. Hashimoto Thyroiditis: Clinical and Diagnostic Criteria. Autoimmunity Reviews. 2014;13(4-5):391–397.
- Hollowell JG, Staehling NW, Flanders WD, et al. Serum TSH, T4, and Thyroid Antibodies in the United States Population (NHANES III). Journal of Clinical Endocrinology & Metabolism. 2002;87(2):489–499.
- Gärtner R, Gasnier BCH, Dietrich JW, Krebs B, Angstwurm MWA. Selenium Supplementation in Patients with Autoimmune Thyroiditis Decreases Thyroid Peroxidase Antibodies Concentrations. Journal of Clinical Endocrinology & Metabolism. 2002;87(4):1687–1691.
- Toulis KA, Anastasilakis AD, Tzellos TG, Goulis DG, Kouvelas D. Selenium Supplementation in the Treatment of Hashimoto's Thyroiditis: A Systematic Review and a Meta-Analysis. Thyroid. 2010;20(10):1163–1173.
- Krysiak R, Szkróbka W, Okopień B. The Effect of Gluten-Free Diet on Thyroid Autoimmunity in Drug-Naïve Women with Hashimoto's Thyroiditis: A Pilot Study. Experimental and Clinical Endocrinology & Diabetes. 2019;127(7):417–422.
- Sategna-Guidetti C, Volta U, Ciacci C, et al. Prevalence of Thyroid Disorders in Untreated Adult Celiac Disease Patients and Effect of Gluten Withdrawal: An Italian Multicenter Study. American Journal of Gastroenterology. 2001;96(3):751–757.
- Nordio M, Pajalich R. Combined Treatment with Myo-Inositol and Selenium Ensures Euthyroidism in Subclinical Hypothyroidism Patients with Autoimmune Thyroiditis. Journal of Thyroid Research. 2013;2013:424163.
- Mazokopakis EE, Papadakis JA, Papadomanolaki MG, et al. Effects of 12 Months Treatment with L-Selenomethionine on Serum Anti-TPO Levels in Patients with Hashimoto's Thyroiditis. Thyroid. 2007;17(7):609–612.
- Wichman J, Winther KH, Bonnema SJ, Hegedus L. Selenium Supplementation Significantly Reduces Thyroid Autoantibody Levels in Patients with Chronic Autoimmune Thyroiditis: A Systematic Review and Meta-Analysis. Thyroid. 2016;26(12):1681–1692.
- Vanderpump MPJ, Tunbridge WMG, French JM, et al. The Incidence of Thyroid Disorders in the Community: A Twenty-Year Follow-Up of the Whickham Survey. Clinical Endocrinology. 1995;43(1):55–68.
- Mazokopakis EE, Papadomanolaki MG, Tsekouras KC, et al. Is Vitamin D Related to Pathogenesis and Treatment of Hashimoto's Thyroiditis? Hellenic Journal of Nuclear Medicine. 2015;18(3):222–227.
- Spencer CA, Takeuchi M, Kazarosyan M, et al. Serum Thyroglobulin Autoantibodies: Prevalence, Influence on Serum Thyroglobulin Measurement, and Prognostic Significance in Patients with Differentiated Thyroid Carcinoma. Journal of Clinical Endocrinology & Metabolism. 1998;83(4):1121–1127.
Live PubMed Searches
Live PubMed queries that update as new papers are indexed.
- PubMed: TPO antibody Hashimoto's
- PubMed: selenium TPO antibody reduction
- PubMed: gluten-free Hashimoto's
- PubMed: TgAb thyroid-cancer interference
- PubMed: euthyroid autoimmune progression
- PubMed: myo-inositol + selenium
- PubMed: vitamin D thyroid autoimmunity
- PubMed: seronegative Hashimoto's
Connections
- Levothyroxine vs NDT vs T3
- TSH, T4 & T3 Optimal Ranges
- Reverse T3 and Low T3 Syndrome
- Selenium, Zinc, Iron & Iodine
- Selenium
- Hashimoto's Thyroiditis
- Thyroid Disorders
- Thyroid Panel
- Graves' Disease
- AIP and Gluten Elimination
- Pregnancy and Hashimoto's
- Hashimoto, POTS & MCAS Overlap
- Vitamin D3
- Iodine
- Thyroid Cancer
- Celiac Disease
- Inositol
- Infertility