Vitiligo
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research and Advances
- References & Research
- Featured Videos
1. Overview
Vitiligo is a chronic autoimmune disorder characterized by the progressive loss of melanocytes, the pigment-producing cells in the skin, resulting in well-demarcated, depigmented white patches (macules and patches) on the skin, mucous membranes, and hair. The condition occurs when the immune system mistakenly attacks and destroys melanocytes, leading to areas of skin that lose their natural color. Vitiligo affects people of all skin types but is more visually apparent in individuals with darker skin tones, often causing significant psychological distress and diminished quality of life.
Vitiligo is classified into two major types: non-segmental vitiligo (also called generalized vitiligo), which accounts for approximately 85-90% of cases and presents with symmetric, bilateral depigmented patches that progressively enlarge over time, and segmental vitiligo, which accounts for 10-15% of cases and presents with unilateral patches confined to one dermatome that typically stabilize within one to two years. Non-segmental vitiligo is further subdivided into several clinical patterns:
- Acrofacial vitiligo — affecting distal extremities and the face
- Generalized (vulgaris) vitiligo — scattered patches across multiple body sites
- Universal vitiligo — complete or near-complete depigmentation involving more than 80% of body surface area
- Mucosal vitiligo — affecting oral and genital mucous membranes
- Focal vitiligo — isolated patches in a limited area without segmental distribution
The disease has been recognized since antiquity, with descriptions found in ancient Egyptian, Greek, and Indian medical texts. Despite being a non-contagious condition with no direct impact on physical health beyond melanocyte loss, vitiligo carries a profound psychosocial burden, particularly in cultures where visible skin differences may lead to stigmatization. Advances in understanding the autoimmune pathogenesis of vitiligo have led to groundbreaking targeted therapies, including JAK inhibitors such as ruxolitinib cream, the first FDA-approved topical treatment specifically indicated for non-segmental vitiligo in 2022.
2. Epidemiology
Vitiligo affects approximately 0.5% to 2% of the global population, with an estimated 70 to 100 million people affected worldwide. The condition occurs with equal frequency across all racial and ethnic groups, though it is more cosmetically noticeable in individuals with darker skin (Fitzpatrick skin types IV-VI). Both sexes are affected equally, although women tend to seek medical attention more frequently due to cosmetic concerns.
The disease can develop at any age, but approximately 50% of cases present before age 20, and 70-80% of cases develop before age 30. The peak onset is between 10 and 30 years of age. Segmental vitiligo tends to have an earlier onset, typically presenting in childhood or early adolescence. A positive family history of vitiligo is present in approximately 20-30% of patients, suggesting a significant genetic component. The concordance rate in monozygotic twins is approximately 23%, indicating that while genetics plays an important role, environmental and stochastic factors are also critical.
Geographic distribution shows some variation in reported prevalence, with higher rates documented in India (up to 8.8% in certain regions), parts of Mexico, and Japan. These differences may reflect both genuine prevalence variations and differences in health-seeking behavior related to cultural attitudes toward skin pigmentation. Vitiligo is associated with other autoimmune conditions, with 15-25% of patients having at least one concurrent autoimmune disorder, most commonly autoimmune thyroid disease (Hashimoto's thyroiditis or Graves' disease), which occurs in 10-15% of vitiligo patients.
3. Pathophysiology
The pathophysiology of vitiligo involves the selective destruction of melanocytes through a complex interplay of autoimmune, genetic, oxidative stress, and neural mechanisms. Current understanding supports the convergence theory, which integrates multiple pathogenic pathways leading to melanocyte loss.
Autoimmune Mechanisms
The autoimmune hypothesis is the most strongly supported mechanism. CD8+ cytotoxic T lymphocytes are the primary effector cells responsible for melanocyte destruction. These autoreactive T cells recognize melanocyte-specific antigens, including tyrosinase, tyrosinase-related protein 1 (TRP-1), tyrosinase-related protein 2 (TRP-2/DCT), gp100/Pmel17, and melanin-concentrating hormone receptor 1 (MCHR1). Perilesional skin biopsies from active vitiligo demonstrate infiltration of CD8+ T cells at the margin of depigmented patches, directly adjacent to disappearing melanocytes. These melanocyte-specific CD8+ T cells produce interferon-gamma (IFN-gamma), which activates the JAK-STAT signaling pathway in keratinocytes, leading to upregulation of CXCL9 and CXCL10 chemokines that recruit additional autoreactive T cells to the skin, creating a self-amplifying inflammatory loop.
The IFN-gamma-CXCL10-CXCR3 Axis
A critical discovery in vitiligo pathogenesis is the central role of the IFN-gamma-CXCL10-CXCR3 signaling axis. Autoreactive CD8+ T cells secrete IFN-gamma, which activates JAK1 and JAK2 in keratinocytes, leading to STAT1 phosphorylation and transcription of CXCL9 and CXCL10. These chemokines bind to CXCR3 on circulating melanocyte-specific T cells, directing them to the skin and perpetuating melanocyte destruction. This pathway is the primary therapeutic target for JAK inhibitors in vitiligo treatment. Additionally, the Wnt signaling pathway is disrupted in vitiligo, impairing melanocyte stem cell differentiation and melanocyte regeneration in depigmented areas.
Oxidative Stress
Melanocytes in vitiligo-prone skin exhibit elevated levels of reactive oxygen species (ROS) and reduced antioxidant defenses. Specifically, there is increased hydrogen peroxide (H2O2) accumulation in the epidermis, reduced catalase activity, and diminished levels of thioredoxin reductase. Oxidative stress damages melanocyte cellular components, exposes intracellular antigens, activates the unfolded protein response in the endoplasmic reticulum, and generates damage-associated molecular patterns (DAMPs) that trigger innate immune activation. This oxidative stress may serve as the initial trigger that converts genetic susceptibility into active autoimmune melanocyte destruction.
Genetic Susceptibility
Genome-wide association studies (GWAS) have identified over 50 genetic susceptibility loci for vitiligo. Approximately 90% of these loci involve genes related to immune regulation, including HLA class I and II genes (particularly HLA-A*02:01), CTLA-4, PTPN22, IL2RA, IFIH1, and TYR (tyrosinase). The remaining 10% of loci involve genes related to melanocyte function and apoptosis. The genetic architecture of vitiligo overlaps significantly with other autoimmune diseases, explaining the frequent co-occurrence with thyroid disease, type 1 diabetes, and rheumatoid arthritis.
4. Etiology and Risk Factors
Genetic Factors
- Family history — first-degree relatives have a 5-8 fold increased risk
- Polygenic inheritance — over 50 susceptibility loci identified through GWAS
- HLA associations — HLA-A*02:01, HLA-DRB1*04, HLA-DRB1*07
- Immune regulatory genes — CTLA-4, PTPN22, IL2RA, NLRP1
- Melanocyte-related genes — TYR, OCA2, MC1R variants
Autoimmune Associations
- Autoimmune thyroid disease — Hashimoto's thyroiditis and Graves' disease (10-15% of patients)
- Type 1 diabetes mellitus — 1-7% of vitiligo patients
- Pernicious anemia — associated with anti-parietal cell antibodies
- Addison's disease — adrenal insufficiency
- Systemic lupus erythematosus
- Rheumatoid arthritis
- Alopecia areata — co-occurs in approximately 4% of vitiligo patients
Environmental Triggers
- Physical trauma — Koebner phenomenon (isomorphic response) triggers depigmentation at sites of skin injury, friction, or pressure
- Sunburn — severe UV exposure may trigger or worsen vitiligo in genetically susceptible individuals
- Chemical exposure — phenolic and catecholic compounds (e.g., para-phenylenediamine in hair dye, monobenzyl ether of hydroquinone, para-tertiary-butylphenol in rubber) can cause chemical leukoderma or trigger true vitiligo
- Psychological stress — emotional stress and major life events frequently precede disease onset or flares
- Viral infections — hepatitis C, cytomegalovirus, and Epstein-Barr virus have been implicated as triggers
Immunological Triggers
- Immune checkpoint inhibitors — anti-PD-1 and anti-CTLA-4 immunotherapy for cancer can induce vitiligo-like depigmentation (paradoxically associated with better tumor response in melanoma)
- Interferon therapy — IFN-alpha treatment can trigger vitiligo
5. Clinical Presentation
Characteristic Lesions
The hallmark of vitiligo is well-demarcated, chalk-white macules and patches with distinct margins. Lesions are typically non-scaly, non-pruritic, and range in size from a few millimeters to many centimeters. The depigmented patches contrast sharply with surrounding normal skin, and the border between affected and unaffected skin is characteristically sharp. In some patients, a trichrome pattern is observed, with zones of white, tan, and normally pigmented skin, or a quadrichrome pattern that includes a marginal zone of hyperpigmentation. An inflammatory variant may present with an erythematous, raised border around depigmented patches.
Distribution Patterns
- Non-segmental (generalized) vitiligo — bilateral, symmetric patches on the face (perioral, periorbital), hands, wrists, axillae, groin, and genitalia; progressive course with periods of stability and activity
- Segmental vitiligo — unilateral patches confined to one or few dermatomes; rapid onset followed by stabilization within 6-24 months; often associated with leukotrichia (white hair) in affected areas
- Acrofacial vitiligo — limited to distal fingers, toes, and facial orifices (lips, eyes, nose)
- Universal vitiligo — extensive depigmentation covering more than 80% of body surface area
Associated Features
- Leukotrichia — white hair (poliosis) within depigmented patches, indicating melanocyte stem cell reservoir destruction; presence suggests poorer repigmentation prognosis
- Koebner phenomenon — development of new lesions at sites of trauma, friction, or pressure (occurs in 20-60% of patients)
- Ocular involvement — uveitis, iris depigmentation, retinal pigment epithelium changes (up to 40% of patients have subclinical findings)
- Inner ear involvement — sensorineural hearing loss reported in up to 16% of patients due to melanocyte loss in the stria vascularis of the cochlea
- Premature graying of hair — canities before age 30 occurs more frequently in vitiligo patients
Psychosocial Impact
Vitiligo significantly affects quality of life, self-esteem, and social functioning. Studies using the Dermatology Life Quality Index (DLQI) consistently demonstrate moderate-to-severe impact on daily life. Patients frequently report embarrassment, anxiety, depression, and avoidance of social situations. The psychosocial burden is often disproportionately greater than the medical severity, particularly in individuals with darker skin tones and in cultures where pigmentation differences carry social stigma.
6. Diagnosis
Clinical Examination
Diagnosis of vitiligo is primarily clinical, based on the characteristic appearance of well-demarcated, chalk-white depigmented macules and patches on physical examination. A thorough skin examination should document the distribution, extent (using body surface area estimation or the Vitiligo Area Scoring Index, VASI), and activity status of lesions.
Wood's Lamp Examination
A Wood's lamp (365 nm UV-A light) examination is the most valuable diagnostic tool. Under Wood's lamp illumination, vitiligo lesions appear bright blue-white with sharp borders, which enhances contrast with surrounding skin, particularly in fair-skinned individuals where lesions may be subtle under ambient light. Wood's lamp also helps distinguish vitiligo from other hypopigmented conditions, identify subclinical depigmentation, and assess disease extent more accurately.
Dermoscopy
Dermoscopy of vitiligo reveals characteristic features including a white glow pattern, residual perifollicular pigmentation (reversed pigment network), starburst pattern in repigmenting lesions, and micro-Koebner phenomenon. In active vitiligo, dermoscopy may show a trichrome pattern and tapioca-sago appearance representing residual melanin islands. These features help differentiate vitiligo from other causes of leukoderma.
Histopathology
Skin biopsy is rarely needed for diagnosis but, when performed, shows complete absence of melanocytes in depigmented epidermis, confirmed by negative Melan-A (MART-1) and HMB-45 immunohistochemical staining. In active lesional borders, a lymphocytic infiltrate at the dermoepidermal junction (interface dermatitis) adjacent to residual melanocytes may be observed. Fontana-Masson staining confirms absence of melanin in affected areas.
Laboratory Evaluation
Routine laboratory testing in vitiligo patients should include screening for associated autoimmune conditions:
- Thyroid function tests — TSH, free T4, anti-thyroid peroxidase (anti-TPO) antibodies, anti-thyroglobulin antibodies (recommended at baseline and annually)
- Complete blood count — to evaluate for pernicious anemia
- Vitamin B12 and folate levels
- Fasting glucose / HbA1c — screening for diabetes
- Antinuclear antibodies (ANA) — if systemic autoimmune disease suspected
- Cortisol level — if Addison's disease suspected
Differential Diagnosis
- Pityriasis alba — ill-defined, hypopigmented (not depigmented) patches, common in children
- Tinea versicolor — hypopigmented patches with fine scale; positive KOH preparation
- Post-inflammatory hypopigmentation — history of preceding inflammatory condition
- Chemical leukoderma — exposure history to phenolic compounds; confetti-like depigmentation
- Piebaldism — congenital; stable white forelock; KIT gene mutation
- Tuberous sclerosis (ash-leaf macules) — lancet-shaped hypopigmented patches present at birth
- Idiopathic guttate hypomelanosis — small, porcelain-white macules on sun-exposed areas in older adults
- Leprosy (Hansen's disease) — hypopigmented patches with decreased sensation
7. Treatment
Topical JAK Inhibitors
Ruxolitinib 1.5% cream (Opzelura) was approved by the FDA in July 2022 as the first topical treatment specifically indicated for non-segmental vitiligo in patients aged 12 years and older. Ruxolitinib inhibits JAK1 and JAK2, blocking the IFN-gamma-CXCL10 signaling pathway that drives melanocyte destruction. In the pivotal TRuE-V1 and TRuE-V2 phase 3 clinical trials, approximately 30% of patients achieved at least 75% improvement in facial Vitiligo Area Scoring Index (F-VASI75) at 24 weeks, compared to approximately 10% with vehicle. Continued treatment up to 52 weeks showed progressive improvement, with approximately 50% of patients achieving F-VASI75. Ruxolitinib cream is applied twice daily to affected areas (maximum 10% body surface area). Common side effects include application site acne and pruritus.
Topical Corticosteroids
Topical corticosteroids remain a first-line treatment for limited vitiligo (<10% body surface area). Potent (class III) or super-potent (class I-II) topical steroids are most effective. Treatment regimens typically use a pulsed schedule (e.g., applied daily for 2 weeks alternating with 2 weeks off, or weekday application with weekend breaks) to minimize adverse effects. Response rates of 40-60% for facial lesions have been reported. Prolonged use risks skin atrophy, striae, and telangiectasia, necessitating regular monitoring.
Topical Calcineurin Inhibitors
Tacrolimus 0.1% ointment and pimecrolimus 1% cream are effective alternatives, particularly for facial and intertriginous areas where corticosteroid-induced atrophy is a concern. These agents suppress T-cell activation and reduce local inflammation without the atrophogenic effects of steroids. Combination with narrowband UVB phototherapy enhances efficacy.
Phototherapy
Narrowband UVB (NB-UVB, 311 nm) phototherapy is the gold-standard treatment for generalized vitiligo affecting more than 10-20% body surface area. Treatment is typically administered 2-3 times per week for a minimum of 6-12 months. Response rates range from 40-70%, with the best results on the face, neck, trunk, and proximal extremities. Acral areas (hands, feet) respond poorly. NB-UVB promotes melanocyte stem cell proliferation, migration from hair follicles to the epidermis, and immunomodulation. Targeted phototherapy using excimer laser (308 nm) or excimer lamp delivers focused UVB to individual lesions, allowing higher doses with less surrounding skin exposure, and is effective for localized, treatment-resistant patches.
Systemic Treatments
- Oral mini-pulse corticosteroids — dexamethasone 2.5 mg on two consecutive days per week (oral mini-pulse therapy) for 3-6 months to halt disease progression in rapidly progressive vitiligo
- Oral JAK inhibitors — tofacitinib (5 mg twice daily) and ritlecitinib have shown promise in clinical trials for widespread vitiligo, though not yet FDA-approved for this indication
- Methotrexate — low-dose methotrexate (10-15 mg weekly) has been studied as a steroid-sparing agent for progressive disease
Surgical Options
Surgical interventions are reserved for stable vitiligo (no new lesions or progression for at least 12 months) that has failed medical therapy:
- Autologous melanocyte transplantation — non-cultured epidermal cell suspension (NCES) or cultured melanocyte transplantation; success rates of 60-90% in properly selected patients
- Suction blister grafting — transfer of ultra-thin epidermal grafts from pigmented donor sites to depigmented recipient sites
- Split-thickness skin grafting — transfer of thin skin grafts to dermabraded vitiligo patches
- Punch grafting — small punch biopsies from pigmented skin transplanted to recipient sites
Depigmentation Therapy
For patients with universal vitiligo (>80% body surface area) or extensive, treatment-resistant disease, depigmentation of remaining pigmented skin may be considered. Monobenzyl ether of hydroquinone (MBEH) 20% cream (Benoquin) is applied twice daily to remaining pigmented areas to achieve uniform depigmentation. This is an irreversible treatment and requires thorough counseling regarding permanent sun sensitivity.
Camouflage and Cosmetic Options
- Cosmetic camouflage — specialized cover creams and foundations matched to surrounding skin tone
- Self-tanning products — dihydroxyacetone-based products to temporarily darken depigmented areas
- Micropigmentation (tattooing) — semi-permanent tattooing of depigmented areas, particularly lips and fingertips
8. Complications
- Psychological morbidity — depression, anxiety, social phobia, low self-esteem, and impaired quality of life, particularly in individuals with darker skin tones and visible areas of involvement
- Sunburn — depigmented skin lacks melanin photoprotection and is highly susceptible to sunburn and UV damage
- Ocular complications — uveitis, chorioretinal depigmentation, and iris transillumination defects
- Hearing impairment — sensorineural hearing loss due to melanocyte destruction in the inner ear
- Koebner phenomenon — new lesions triggered by physical trauma, surgery, or skin injury
- Associated autoimmune diseases — thyroid disease, type 1 diabetes, pernicious anemia, Addison's disease, alopecia areata
- Treatment-related complications — corticosteroid-induced skin atrophy, phototherapy-induced phototoxicity, surgical scarring
- Paradoxical photocarcinogenesis risk — while vitiligo patients may have a lower overall risk of melanoma (due to enhanced anti-melanocyte immunity), depigmented skin is vulnerable to UV-induced non-melanoma skin cancer
9. Prognosis
Vitiligo is a chronic, often progressive condition with an unpredictable course. Spontaneous repigmentation occurs in 10-20% of patients, typically in sun-exposed areas and often partial. The disease course is characterized by periods of stability alternating with episodes of progression, sometimes triggered by stress, illness, or skin trauma.
Several factors influence treatment response and prognosis. Facial and neck lesions respond best to treatment, with repigmentation rates of 60-70% with NB-UVB phototherapy. Trunk and proximal extremity lesions have intermediate response rates of 40-50%. Acral lesions (hands, feet, bony prominences) are the most treatment-resistant, with repigmentation rates below 20%. Segmental vitiligo generally has a better prognosis than non-segmental types, as it tends to stabilize early and responds well to surgical interventions. The presence of leukotrichia within depigmented patches indicates destruction of the melanocyte stem cell reservoir and predicts poorer repigmentation potential. Early treatment, younger patient age, and recent-onset lesions are associated with better outcomes.
With modern therapies including JAK inhibitors and combination approaches, significant repigmentation is achievable in a majority of patients, though complete and sustained repigmentation remains challenging. Maintenance therapy is often required to prevent relapse, as repigmented areas can redepigment when treatment is discontinued.
10. Prevention
Primary prevention of vitiligo is not currently possible given the complex interplay of genetic susceptibility and environmental triggers. However, several strategies may help prevent disease progression and reduce the risk of new lesions:
- Sun protection — regular use of broad-spectrum SPF 30+ sunscreen on depigmented areas to prevent sunburn and reduce Koebner phenomenon; sun-protective clothing and hats
- Avoidance of skin trauma — minimize friction, pressure, and skin injury to reduce Koebner phenomenon-triggered new patches
- Chemical avoidance — avoid phenolic and catecholic compounds in hair dyes, rubber products, and cleaning agents that may trigger chemical-induced depigmentation
- Stress management — psychological stress reduction through counseling, mindfulness, cognitive behavioral therapy, and support groups
- Early treatment — prompt initiation of therapy at first signs of depigmentation to halt progression and maximize repigmentation potential
- Autoimmune screening — regular monitoring for thyroid dysfunction and other associated autoimmune conditions, particularly in patients with family history
- Vitamin D supplementation — maintaining adequate vitamin D levels, as vitiligo patients may have increased risk of deficiency due to sun avoidance behaviors
- Antioxidant supplementation — oral antioxidants (vitamins C, E, alpha-lipoic acid, polypodium leucotomos) as adjunctive therapy may support melanocyte protection, though evidence is limited
11. Recent Research and Advances
Vitiligo research has undergone a transformation in recent years, driven by improved understanding of the IFN-gamma-CXCL10 signaling axis and the development of targeted therapies.
Ruxolitinib cream (Opzelura) received FDA approval in July 2022, marking a paradigm shift in vitiligo treatment as the first and currently only FDA-approved therapy specifically for non-segmental vitiligo. Long-term extension data from the TRuE-V studies demonstrate continued improvement beyond 52 weeks with sustained use, and acceptable safety profile. Additional topical JAK inhibitors are under investigation, including delgocitinib cream (pan-JAK inhibitor) and cerdulatinib.
Oral JAK inhibitors are being investigated for extensive disease. Ritlecitinib (a selective JAK3/TEC family kinase inhibitor, already approved for alopecia areata) is being studied in phase 3 trials for vitiligo. Povorcitinib (a selective JAK1 inhibitor) has shown promising results in phase 2b trials, with significant facial repigmentation. Combination of oral JAK inhibitors with phototherapy appears to produce synergistic results.
Research into melanocyte stem cell biology and Wnt signaling pathway activation offers potential for enhancing melanocyte regeneration from hair follicle reservoirs. Tissue-resident memory T cells (TRM) have been identified as key mediators of vitiligo relapse, persisting in previously affected skin even after successful repigmentation. Strategies to deplete TRM cells or prevent their reactivation could lead to durable remission. IL-15 blockade is emerging as a promising approach to eliminate skin-resident autoreactive memory T cells and prevent relapse.
Afamelanotide, a synthetic alpha-melanocyte-stimulating hormone (alpha-MSH) analog administered as a subcutaneous implant, has shown synergistic benefit when combined with NB-UVB phototherapy in clinical trials, accelerating and enhancing repigmentation, particularly in patients with darker skin types. Prostaglandin E2 analogs (bimatoprost) and platelet-rich plasma (PRP) injections are being explored as adjunctive treatments to stimulate melanocyte regeneration.
12. References & Research
Historical Background
Vitiligo has been recognized for millennia, with the earliest descriptions appearing in the Ebers Papyrus (circa 1550 BCE) of ancient Egypt and the Atharva Veda of ancient India (circa 1400 BCE), where the condition was referred to as "kilas" or "shweta kushtha." The Greek physician Celsus first used the term "vitiligo" in the 1st century CE, derived from the Latin "vitium" (blemish). For centuries, vitiligo was conflated with leprosy and other conditions causing skin discoloration. Modern understanding began in the 19th century when Moritz Kaposi distinguished vitiligo from other leukodermas. The autoimmune hypothesis was first proposed by A.B. Lerner in the 1950s-1960s, who also pioneered the use of psoralen-UVA (PUVA) therapy. The landmark discovery of the role of CD8+ T cells in melanocyte destruction by Caroline Le Poole and John Harris in the early 2000s laid the groundwork for targeted immunotherapy, culminating in the development and approval of ruxolitinib cream.
Key Research Papers
- Ezzedine K, Eleftheriadou V, Whitton M, van Geel N. Vitiligo. Lancet. 2015;386(9988):74-84. doi:10.1016/S0140-6736(14)60763-7
- Rodrigues M, Ezzedine K, Hamzavi I, et al. New discoveries in the pathogenesis and classification of vitiligo. J Am Acad Dermatol. 2017;77(1):1-13. doi:10.1016/j.jaad.2016.10.048
- Harris JE, Harris TH, Weninger W, et al. A mouse model of vitiligo with focused epidermal depigmentation requires IFN-gamma for autoreactive CD8+ T-cell accumulation in the skin. J Invest Dermatol. 2012;132(7):1869-1876. doi:10.1038/jid.2011.463
- Rashighi M, Agarwal P, Richmond JM, et al. CXCL10 is critical for the progression and maintenance of depigmentation in a mouse model of vitiligo. Sci Transl Med. 2014;6(223):223ra23. doi:10.1126/scitranslmed.3007811
- Rosmarin D, Pandya AG, Engstrom-Kasperson M, et al. Ruxolitinib cream for treatment of vitiligo (TRuE-V1 and TRuE-V2): results from two randomised, double-blind, phase 3 trials. Lancet. 2022;400(10344):562-571. doi:10.1016/S0140-6736(22)00475-6
- Jin Y, Andersen G, Yorgov D, et al. Genome-wide association studies of autoimmune vitiligo identify 23 new risk loci and highlight key pathways and regulatory variants. Nat Genet. 2016;48(11):1418-1424. doi:10.1038/ng.3680
- Spritz RA, Andersen GHL. Genetics of vitiligo. Dermatol Clin. 2017;35(2):245-255. doi:10.1016/j.det.2016.11.013
- Richmond JM, Strassner JP, Zapata L, et al. Antibody blockade of IL-15 signaling has the potential to durably reverse vitiligo. Sci Transl Med. 2018;10(450):eaam7710. doi:10.1126/scitranslmed.aam7710
- Picardo M, Dell'Anna ML, Ezzedine K, et al. Vitiligo. Nat Rev Dis Primers. 2015;1:15011. doi:10.1038/nrdp.2015.11
- Frisoli ML, Essien K, Harris JE. Vitiligo: mechanisms of pathogenesis and treatment. Annu Rev Immunol. 2020;38:621-648. doi:10.1146/annurev-immunol-100919-023531
- Boniface K, Seneschal J, Picardo M, Taieb A. Vitiligo: focus on clinical aspects, immunopathogenesis, and therapy. Clin Rev Allergy Immunol. 2018;54(1):52-67. doi:10.1007/s12016-017-8622-7
- Lotti T, Gori A, Zanieri F, et al. Vitiligo: new and emerging treatments. Dermatol Ther. 2008;21(2):110-117. doi:10.1111/j.1529-8019.2008.00178.x
- Pandya AG, Harris JE, Engstrom KM, et al. Long-term efficacy and safety of ruxolitinib cream in vitiligo: results from the TRuE-V long-term extension study. J Am Acad Dermatol. 2024;90(1):62-70. doi:10.1016/j.jaad.2023.08.049
- Le Poole IC, van den Wijngaard RM, Westerhof W, Das PK. Presence of T cells and macrophages in inflammatory vitiligo skin parallels melanocyte disappearance. Am J Pathol. 1996;148(4):1219-1228. doi:10.1016/S0002-9440(10)65381-0
- Passeron T, Ortonne JP. Use of the 308-nm excimer laser for psoriasis and vitiligo. Clin Dermatol. 2006;24(1):33-42. doi:10.1016/j.clindermatol.2005.10.025
Research Papers
Curated PubMed topic searches of peer-reviewed literature on Vitiligo. Each link opens a live PubMed query so you always see the most current studies.
- PubMed: Vitiligo pathogenesis
- PubMed: Narrowband UVB
- PubMed: Ruxolitinib for vitiligo
- PubMed: Topical therapies
- PubMed: Melanocyte transplantation
- PubMed: Vitiligo and thyroid autoimmunity
- PubMed: Epidemiology
- PubMed: Psychological impact
- PubMed: Oxidative stress
- PubMed: Excimer laser therapy
- PubMed: Segmental vs non-segmental
- PubMed: Afamelanotide trials
Connections
- Dermatology
- Psoriasis
- Alopecia
- Eczema
- Celiac Disease
- Vitamin D
- Vitamin B12
- Zinc
- Stress Management
- Gut Health
Featured Videos

Vitiligo: Truth, Hope and Change (full-length)

Vitiligo: Causes, Symptoms, and Treatment

Vitiligo treatments- what works!

How I help people understand vitiligo | Lee Thomas
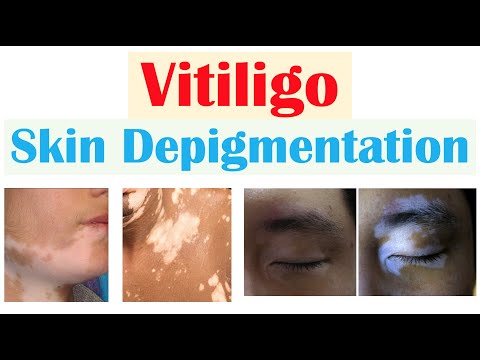
Vitiligo (Skin Depigmentation) | Pathophysiology, Signs and Symptoms, Diagnosis, Treatment

Living with Vitiligo | Megan Relin | TEDxDeerfield

A Deep Dive Into Vitiligo and Its Associated Comorbidities

Classifying Vitiligo: Non-Segmental Versus Segmental Disease

Understanding Vitiligo: Causes, Symptoms, and Treatment

Vitiligo: WHY YOU HAVE IT & WHAT TO AVOID

Doctor explains VITILIGO TREATMENT OPTIONS | Phototherapy, excimer laser, steroids & more

JAK Inhibition for the Treatment of Vitiligo

Autoimmune Disorders Like Mine (Vitiligo) Can Be Activated by Trauma

7 TIPS FOR VITILIGO
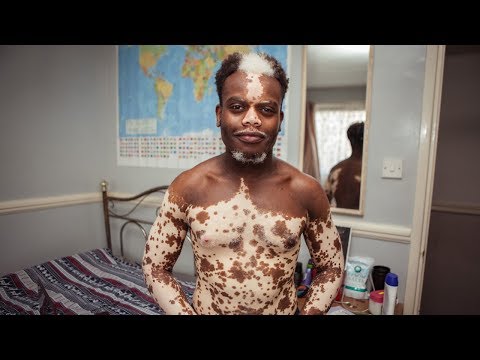
My Vitiligo is Disappearing - But I Don’t Want It To | BORN DIFFERENT

Vitiligo: Symptoms, Causes, Types & Management I Dr. Amit Vij