Pheochromocytoma
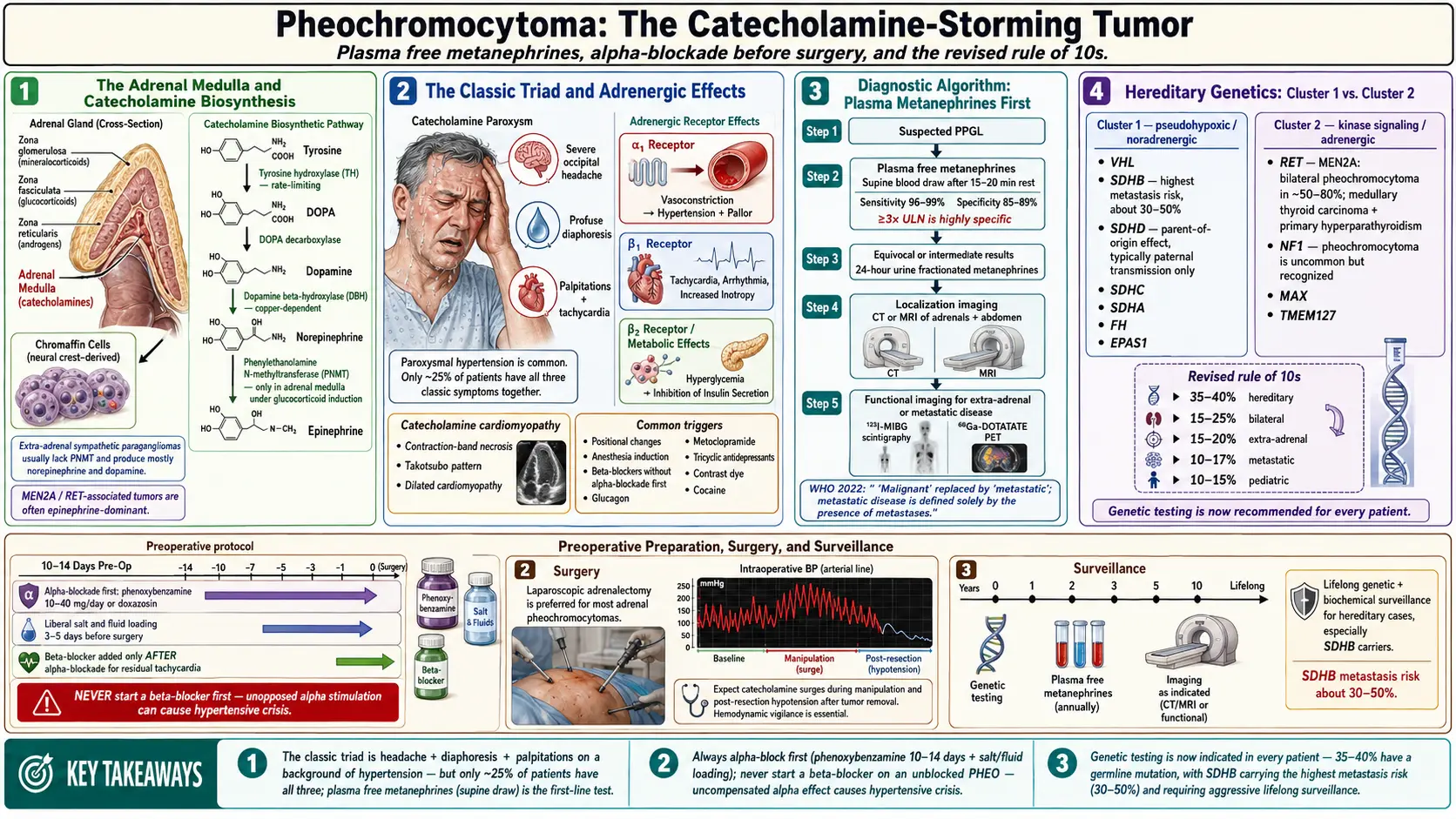
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research
- References
- Featured Videos
1. Overview
Pheochromocytoma (PHEO) is a catecholamine-secreting neuroendocrine tumor arising from the chromaffin cells of the adrenal medulla. Closely related tumors arising from extra-adrenal chromaffin cells (paraganglia) of the sympathetic chain or parasympathetic ganglia are termed paragangliomas (PGL). Together, they are referred to as pheochromocytoma-paraganglioma (PPGL) syndromes. Historically recognized by the "rule of 10s" — 10% bilateral, 10% extra-adrenal, 10% malignant, 10% familial, 10% pediatric — contemporary genomic data have substantially revised these estimates, particularly the hereditary fraction, now established at 35–40% of all cases.
PHEO is clinically important for several reasons: it is a surgically curable cause of hypertension that, if unrecognized and untreated, can precipitate life-threatening hypertensive crises, cardiovascular events, and death. Accurate preoperative preparation with alpha-adrenergic blockade is mandatory to prevent intraoperative hypertensive crisis. Additionally, the high heritability of PHEO necessitates systematic genetic evaluation of every patient, with implications for surveillance of other hereditary endocrine tumors and cascade screening of family members.
2. Epidemiology
- Incidence: Approximately 2–8 cases per million population per year. Population-based autopsy studies suggest many cases go unrecognized during life; PHEO is found incidentally in 0.05–0.1% of all autopsies.
- Prevalence in hypertension: PHEO accounts for 0.1–0.6% of hypertensive patients in the general population — a small proportion numerically but clinically critical given the lethal potential of missed diagnosis. In patients with adrenal incidentaloma (an adrenal mass found incidentally on imaging), PHEO accounts for approximately 4–7% of cases.
- Age at diagnosis: Peak incidence is in the third to fifth decades of life. However, PHEO occurs at all ages, including in children (where hereditary forms predominate). The median age at diagnosis in large series is approximately 45–50 years.
- Sex distribution: Roughly equal sex distribution, though some hereditary syndromes (e.g., MEN2A) have equal sex incidence while others may show slight sex differences.
- Revised "Rule of 10s": With modern genetic testing, the distribution is better described as:
- 15–25% bilateral (versus the historical 10%)
- 15–20% extra-adrenal (paraganglioma)
- 10–17% metastatic ("malignant" by current WHO 2022 definition — defined solely by the presence of metastases, not histology)
- 35–40% hereditary (versus the historical 10%)
- 10–15% pediatric
- Extra-adrenal (paraganglioma): Sympathetic paragangliomas arise most commonly in the abdomen (organ of Zuckerkandl near the aortic bifurcation, retroperitoneum, perirenal, bladder), thorax (posterior mediastinum), and pelvis. Parasympathetic paragangliomas arise predominantly in the head and neck (carotid body, jugular, vagal) and are most commonly non-secreting.
3. Pathophysiology
Embryology and Cell of Origin
Pheochromocytoma cells derive from neural crest progenitors that migrate to the adrenal gland during embryogenesis and differentiate into chromaffin cells (named for their brown coloration upon chromate salt staining due to catecholamine oxidation). These cells are the functional equivalent of postganglionic sympathetic neurons; they synthesize, store, and secrete catecholamines in response to autonomic and endocrine stimuli.
Catecholamine Biosynthesis
The catecholamine biosynthetic pathway in pheochromocytoma cells proceeds as follows:
- Tyrosine → DOPA: Tyrosine hydroxylase (TH) — the rate-limiting enzyme — converts tyrosine to dihydroxyphenylalanine (DOPA) using tetrahydrobiopterin (BH₄) as cofactor. TH activity is stimulated by neural firing, reduced by feedback inhibition from catecholamines themselves.
- DOPA → Dopamine: Aromatic L-amino acid decarboxylase (DOPA decarboxylase) rapidly converts DOPA to dopamine.
- Dopamine → Norepinephrine (NE): Dopamine beta-hydroxylase (DBH) in the vesicle lumen converts dopamine to norepinephrine. Copper-dependent enzyme; reaction produces NE stored in chromaffin granules.
- Norepinephrine → Epinephrine (EPI): Phenylethanolamine N-methyltransferase (PNMT) — exclusively in the adrenal medulla and a few brainstem neurons — N-methylates NE to epinephrine. PNMT requires glucocorticoid induction (cortisol from the adrenal cortex reaches the medulla at high concentrations via the portal blood system). Extra-adrenal paragangliomas lack PNMT and therefore produce predominantly NE and dopamine, not epinephrine.
Pheochromocytomas predominantly secrete norepinephrine; those associated with MEN2A or RET mutations often secrete predominantly or exclusively epinephrine (due to retained PNMT expression and glucocorticoid exposure). SDH-mutant tumors have a distinct biochemical phenotype characterized by elevated methoxytyramine (a dopamine metabolite), reflecting dopaminergic secretion. VHL-mutant tumors are also predominantly noradrenergic.
Catecholamine Storage and Secretion
Catecholamines are stored in chromaffin granules (large dense-core vesicles) at very high concentrations (~0.5 M) complexed with chromogranin A/B proteins, ATP, and enkephalins. Secretion occurs by exocytosis triggered by acetylcholine (acting on nicotinic receptors from splanchnic nerve stimulation), histamine, glucagon, ACTH, corticotropin-releasing factor, and direct physical compression of the tumor. This explains why activities that compress the tumor (palpation, positional changes, micturition for bladder paraganglioma, physical exertion) can trigger paroxysmal catecholamine surges. Unlike postganglionic sympathetic neurons where re-uptake terminates catecholamine action, PHEO cells have limited neuronal re-uptake (NET) capacity, leading to prolonged systemic catecholamine exposure.
Catecholamine Metabolism and Metanephrines
Catecholamines are metabolized by two principal enzymes:
- Catechol-O-methyltransferase (COMT): O-methylates catecholamines to metanephrines (normetanephrine from NE, metanephrine from EPI, methoxytyramine from dopamine). This intra-tumoral metabolism — occurring continuously and independent of secretory events — is the metabolic basis for measuring plasma free metanephrines and urine fractionated metanephrines as the most sensitive diagnostic biomarkers. Metanephrines are produced constitutively by the tumor even when catecholamine secretion is intermittent.
- Monoamine oxidase (MAO): Deaminates catecholamines to reactive aldehydes, then to vanillylmandelic acid (VMA) via aldehyde dehydrogenase. VMA was the first biochemical marker for PHEO but has lower sensitivity than metanephrines.
Cardiovascular Effects
The clinical manifestations of PHEO are largely mediated through adrenergic receptor activation:
- Alpha-1 adrenergic receptors (vascular smooth muscle): Vasoconstriction → hypertension, reduced tissue perfusion.
- Alpha-2 adrenergic receptors (presynaptic, platelet, fat cell): Feedback inhibition of NE release; platelet aggregation; lipolysis inhibition.
- Beta-1 adrenergic receptors (cardiac): Positive inotropy, chronotropy, dromotropy — tachycardia, increased cardiac output, arrhythmias.
- Beta-2 adrenergic receptors (vascular, bronchial, hepatic, pancreatic): Vasodilation (paradoxical in EPI-secreting PHEO), bronchodilation, glycogenolysis (hyperglycemia), insulin inhibition (glucose intolerance).
Catecholamine cardiomyopathy: Prolonged catecholamine excess causes catecholamine-induced cardiomyopathy — characterized by myocyte necrosis, contraction band necrosis (hypercontraction injury from calcium overload), and fibrosis. This can present as dilated or takotsubo-like cardiomyopathy, heart failure, and malignant arrhythmias. Epinephrine-predominant tumors are particularly prone to causing this complication.
Volume depletion: Chronic catecholamine-induced vasoconstriction reduces effective circulating volume (venous capacitance is chronically reduced); when the PHEO is resected and catecholamine levels plummet, massive reflex vasodilation occurs, causing profound hypotension unless adequate preoperative volume expansion has occurred — the rationale for alpha-blockade and liberal salt/fluid loading before surgery.
4. Etiology and Risk Factors
Modern genomic characterization has revealed that pheochromocytoma is among the most heritable of all human solid tumors, with germline mutations identified in 35–40% of apparently sporadic cases. Somatic mutations are identified in an additional 30–35%. The major driver mutations cluster around two transcriptomic signaling clusters:
Cluster 1: Pseudohypoxic Pathway Genes
Mutations in this cluster upregulate hypoxia-inducible factor (HIF) target genes, mimicking chronic hypoxia and promoting angiogenesis and tumor growth. These tumors are predominantly noradrenergic with high dopamine/normetanephrine output.
- VHL (von Hippel-Lindau) gene: Encodes the VHL protein — part of the ubiquitin E3 ligase complex that targets HIF-1α for proteasomal degradation under normoxic conditions. Loss of VHL results in constitutive HIF-1α accumulation. VHL syndrome: autosomal dominant; PHEO in 10–20% of patients; also hemangioblastomas (cerebellar, spinal, retinal), clear cell renal cell carcinoma, pancreatic cysts, endolymphatic sac tumors. PHEO in VHL is typically bilateral, multifocal, predominantly noradrenergic, and rarely malignant.
- SDHx genes (succinate dehydrogenase subunits A, B, C, D, and cofactor SDHAF2): SDH is a mitochondrial enzyme (Complex II) that oxidizes succinate to fumarate and reduces ubiquinone. Loss of SDH function causes succinate accumulation, which competitively inhibits alpha-ketoglutarate-dependent dioxygenases (prolyl hydroxylases), preventing HIF-1α degradation and also inhibiting TET methylcytosine dioxygenases and histone demethylases — producing a hypermethylator epigenetic phenotype. Key clinical associations:
- SDHB: Highest risk of malignant PHEO/PGL (30–50% lifetime risk of metastasis). Associated with extra-adrenal paraganglioma, renal cell carcinoma (SDHB-associated RCC — distinct histologic subtype), GIST. Aggressive surveillance essential.
- SDHD: Predominantly head and neck paragangliomas (parasympathetic, non-secreting); adrenal PHEO also occurs. Unique parent-of-origin effect: pathogenic only when inherited from the father (maternal imprinting of chromosome 11q).
- SDHC: Predominantly head and neck paragangliomas; lower malignancy risk.
- SDHA: PHEO, PGL, GIST; lower penetrance than SDHB.
- EPAS1 (HIF-2α): Gain-of-function somatic mutations cause polycythemia-paraganglioma-somatostatinoma syndrome; multiple PGL, polycythemia, and duodenal somatostatinoma.
- FH (fumarate hydratase): Loss-of-function mutations produce fumarate accumulation (analogous mechanism to succinate in SDH). Associated with hereditary leiomyomatosis and renal cell carcinoma (HLRCC) and PHEO/PGL.
Cluster 2: Kinase Signaling Pathway Genes
Mutations activating RAS/MAPK/PI3K-AKT-mTOR pathways; produce predominantly epinephrine-secreting tumors (adrenal location).
- RET proto-oncogene: Gain-of-function mutations cause constitutive activation of RET tyrosine kinase. The central genetic driver of MEN2A and MEN2B.
- MEN2A (multiple endocrine neoplasia type 2A): Medullary thyroid carcinoma (MTC, 95–100% penetrance), PHEO (40–60% lifetime risk), primary hyperparathyroidism (20–30%). RET codon 634 mutations (exon 11) are most strongly associated with MEN2A and PHEO. PHEO is bilateral in 50–80% of MEN2A patients but rarely malignant. Critically, PHEO must be excluded before thyroid surgery in MEN2A patients — operating on an unrecognized PHEO without alpha-blockade is potentially fatal.
- MEN2B: MTC (most aggressive subtype), PHEO, mucosal neuromas, marfanoid habitus. PHEO less commonly malignant.
- NF1 (neurofibromin): Loss-of-function mutations cause neurofibromatosis type 1 (NF1). PHEO occurs in 0.1–3.3% of NF1 patients; may be the first endocrine manifestation. NF1-associated PHEO is almost exclusively adrenal, predominantly epinephrine-secreting, and rarely metastatic.
- MAX (MYC-associated factor X): Tumor suppressor; heterodimer partner of MYC. Autosomal dominant PHEO with paternal inheritance effect; bilateral; elevated norepinephrine and methoxytyramine.
- TMEM127: Negative regulator of mTOR; autosomal dominant; bilateral adrenal PHEO.
Sporadic PHEO
In the 25–35% of patients without detectable germline mutations, somatic mutations are identified in many cases: HRAS (~7%), NF1 (~20%), VHL (~9%), EPAS1 (~7%), and others. Truly sporadic (no germline, no identified somatic driver) PHEO likely represents a heterogeneous group with unidentified driver mutations or epigenetic driver events.
Risk Factors for Metastatic Disease
- SDHB germline mutation (30–50% metastatic rate)
- Extra-adrenal location (particularly retroperitoneal and thoracic paraganglioma)
- Tumor size greater than 5–6 cm
- ATRX mutation (somatic; associated with metastatic behavior)
- High Ki-67 index on histology
- Elevated methoxytyramine (dopamine metabolite) — a biomarker for aggressive/metastatic behavior
5. Clinical Presentation
Classic Symptomatic Presentation
The classic triad of PHEO consists of episodic headache, diaphoresis, and palpitations in association with hypertension. This triad has a sensitivity of approximately 90% and a specificity greater than 94% for PHEO when all three components are present. However, fewer than 25% of patients present with all three classic symptoms.
Paroxysmal Features
PHEO may present with dramatic paroxysmal episodes ("attacks" or "spells") lasting minutes to hours, characterized by any combination of:
- Headache: Severe, throbbing, often occipital; typically resolving rapidly after the episode. Occurs in 60–90% of symptomatic patients.
- Diaphoresis: Profuse sweating, often generalized; present in 50–70%.
- Palpitations and tachycardia: Paroxysmal; occasionally bradycardia from reflex vagal activation or when alpha-mediated hypertension is dominant; present in 50–70%.
- Pallor: From alpha-1-mediated cutaneous vasoconstriction (in contrast to other causes of hypertensive urgency where facial flushing may predominate). Followed by reactive flushing in some cases as catecholamines clear.
- Anxiety, tremor, sense of impending doom: From central and peripheral catecholamine effects.
- Nausea and vomiting
- Visual disturbances: Blurred vision from hypertensive retinopathy; occasionally transient cortical blindness.
- Abdominal or chest pain
Paroxysms can be spontaneous or precipitated by:
- Physical activity, position change, bending forward, or abdominal pressure (direct tumor compression)
- Urination or defecation (particularly for bladder paraganglioma)
- Anesthesia induction, intubation, tumor manipulation during surgery
- Medications: beta-blockers (uncompensated alpha effect), glucagon (diagnostic concern when used for hypoglycemia), metoclopramide, tricyclic antidepressants, tyramine-containing foods (in patients on MAO inhibitors), contrast dye, cocaine, corticosteroids (ACTH stimulation)
- Chemotherapy agents (vincristine)
Hypertension Patterns
- Sustained hypertension: Present in approximately 50–60% of PHEO patients; may be indistinguishable from essential hypertension on clinical grounds alone, which is why PHEO is a "silent killer" in many cases.
- Paroxysmal hypertension: Present in 25–30%; marked by episodic severe BP elevations (often to 200–250/130–150 mmHg) against a background of normal or mildly elevated baseline BP.
- Sustained + paroxysmal (mixed): The most common pattern (~50%); persistently elevated blood pressure with superimposed paroxysmal surges.
- Normotension: Approximately 10–15% of PHEO patients are normotensive, particularly those with small tumors, predominantly dopamine-secreting tumors, or parasympathetic head-neck paragangliomas.
Atypical Presentations
- Incidentaloma: Increasing number of PHEOs discovered incidentally on CT or MRI performed for unrelated indications. Biochemical evaluation is mandatory for all adrenal incidentalomas regardless of symptoms.
- Catecholamine cardiomyopathy: Presenting as dilated cardiomyopathy, Takotsubo (stress) cardiomyopathy, or acute heart failure. May be confused with myocarditis or primary cardiomyopathy. PHEO should be excluded in all cases of unexplained cardiomyopathy.
- Hypertensive crisis and encephalopathy
- Pulmonary edema
- MI or stroke from hypertensive or vasospastic complications
- Hyperglycemia: Catecholamine-induced insulin inhibition and hepatic glycogenolysis can cause significant hyperglycemia mimicking new-onset diabetes.
- Weight loss, fever, raised ESR: From catecholamine-mediated metabolic hyperstimulation and the endocrine milieu of the tumor. May mimic malignancy.
- Bilateral adrenal masses in hereditary syndromes (MEN2A, VHL).
Pediatric PHEO
Children with PHEO are more likely to have hereditary disease, extra-adrenal paraganglioma, bilateral disease, and sustained hypertension (rather than paroxysmal). Growth failure, polydipsia, and visual disturbances may be prominent.
6. Diagnosis
Biochemical confirmation precedes imaging in the diagnostic algorithm, except in the acutely ill patient where imaging may be obtained urgently. The Endocrine Society guidelines (2014, updated 2019) recommend plasma free metanephrines as the first-line biochemical test.
Biochemical Testing
Plasma free metanephrines (normetanephrine and metanephrine):
- Preferred first-line test; sensitivity 96–99% for adrenal PHEO, specificity 85–89%.
- Blood draw should be performed in the supine position after 15–20 minutes of rest; sitting/standing position produces a 3–4-fold increase in plasma normetanephrine from sympathetic noradrenergic nerve activity, causing false-positive results in patients without PHEO.
- Plasma metanephrines greater than 3× the upper limit of normal (ULN) are highly specific for PHEO; values between 1× and 3× ULN require clinical context and often confirmatory testing.
- Methoxytyramine (plasma or urine): The O-methylated metabolite of dopamine; elevated in dopamine-secreting tumors (SDH-mutant, extra-adrenal PGL) and malignant PHEO. Addition of methoxytyramine to the metanephrine panel increases sensitivity for SDH-mutant and metastatic tumors. Now recommended as routine by European guidelines.
24-hour urine fractionated metanephrines (normetanephrine + metanephrine + methoxytyramine):
- Sensitivity 87–97%, specificity 86–99%. Complementary to plasma testing; particularly useful when plasma testing is borderline or unavailable.
- Urine collection requires careful patient instruction (first voided specimen discarded; all subsequent urine for 24 hours collected in acidified container; refrigerated).
- Urine total metanephrines greater than 900 mcg/24h or fractionated normetanephrine/metanephrine greater than 2× ULN are highly suggestive.
Urine catecholamines (norepinephrine, epinephrine, dopamine):
- Sensitivity 79–91%. Less sensitive than metanephrines due to episodic catecholamine secretion; a timed urine collection may miss intermittent secretors. Still useful as adjunct; elevated epinephrine points to adrenal origin (PNMT-expressing tumor).
Urine VMA (vanillylmandelic acid):
- Sensitivity 64–74%; lowest sensitivity of the available biomarkers. No longer recommended as first-line; replaced by fractionated metanephrines.
Chromogranin A (CgA):
- A non-specific neuroendocrine marker elevated in many NETs; elevated in ~80% of PHEOs. Useful for monitoring tumor burden and treatment response in metastatic disease. False positives: proton pump inhibitor use, renal failure, liver disease.
Clonidine suppression test:
- In patients with borderline plasma normetanephrine elevation (especially in the context of sympathetic activation such as anxiety or sitting position), clonidine 0.3 mg orally suppresses sympathetically released NE (plasma NE falls by greater than 50% or to normal in normals) but does not suppress tumor-derived NE. Plasma NE measured at baseline and 3 hours after clonidine. Failure to suppress plasma normetanephrine below 112 pg/mL or by less than 40% supports PHEO. Limited clinical utility in the era of plasma free metanephrines.
Medications Causing False-Positive Metanephrines
This is a critically important practical consideration:
- Tricyclic antidepressants (TCAs): Block norepinephrine reuptake; increase plasma normetanephrine substantially. Must be discontinued 2 weeks before testing when clinically safe.
- Serotonin-norepinephrine reuptake inhibitors (SNRIs; venlafaxine, duloxetine): Similar mechanism to TCAs for norepinephrine.
- Levodopa: Increases catecholamine precursors; elevates urine dopamine and methoxytyramine.
- Alpha-methyldopa: Converted to alpha-methyl-NE; elevates normetanephrine.
- Buspirone, acetaminophen (in some assays), monoamine oxidase inhibitors.
- Physiologic states causing false positives: Renal failure (reduced metanephrine clearance — plasma metanephrines are unreliable in CKD; prefer urine collection over a shorter timed period or use urine metanephrine-to-creatinine ratios), obstructive sleep apnea, acute illness, intense physical activity, sympathomimetic drug use (cocaine, amphetamines).
Anatomic Imaging
Imaging is performed only after biochemical confirmation of PHEO (or simultaneously in suspected crisis). Anatomic localization precedes functional imaging.
- CT of the abdomen and pelvis with contrast: First-line imaging for most patients. CT has sensitivity greater than 93–100% for adrenal PHEO (adrenal PHEO typically greater than 3 cm, well-defined, heterogeneous if large, high-density on unenhanced CT — typically greater than 10 HU, unlike benign lipid-rich adenomas which are less than 10 HU). Washout characteristics at 10–15 minutes: PHEOs show slow (less than 50%) absolute washout, unlike lipid-poor adenomas and consistent with malignant adrenal masses — though PHEOs are not always reliably distinguished from metastases by washout alone.
- MRI abdomen/pelvis: Equivalent sensitivity to CT for adrenal PHEO (greater than 90%); preferred in pediatric patients (no radiation), pregnancy, patients with contrast allergy, or when iodine-based contrast is contraindicated. Classic MRI appearance: markedly hyperintense on T2-weighted sequences ("light bulb" sign) — though this is not universally present, particularly in cystic or hemorrhagic lesions.
- CT chest: For thoracic paragangliomas (posterior mediastinal location); also part of staging in suspected malignant disease.
Caution with contrast and other agents: Standard low-osmolality contrast agents (iohexol, iopamidol) are safe in well alpha-blocked PHEO patients. Ionic high-osmolality agents (historically avoided) carry higher risk of catecholamine release. Alpha-blockade before imaging in biochemically confirmed PHEO is prudent.
Functional Imaging
Functional imaging is recommended to confirm adrenal PHEO prior to surgery, to identify multifocal or metastatic disease, to evaluate extra-adrenal paragangliomas, and for postoperative surveillance when biochemical recurrence is detected.
- [¹²³I]-MIBG (metaiodobenzylguanidine) scintigraphy: MIBG is a structural analog of norepinephrine taken up by the norepinephrine transporter (NET, SLC6A2) and stored in catecholamine vesicles. SPECT/CT acquisition improves sensitivity and anatomic localization. Sensitivity for adrenal PHEO: 85–88%; lower for malignant/metastatic and SDH-mutant tumors (which often express reduced NET). MIBG scintigraphy qualifies the tumor for therapeutic [¹³¹I]-MIBG treatment if positive — making its theranostic relevance important in metastatic disease.
- [¹⁸F]-FDG PET/CT: High sensitivity (greater than 90%) for metastatic and malignant PHEO, particularly SDHB-mutant tumors (which are FDG-avid due to the pseudohypoxic metabolic shift). Less sensitive for benign adrenal PHEO.
- [¹⁸F]-FDA (fluorodopamine) PET/CT: Very high sensitivity (95–100%) for adrenal PHEO; uses the NET transporter pathway. Limited availability.
- [¹⁸F]-FDOPA (fluorodihydroxyphenylalanine) PET/CT: High sensitivity for adrenal PHEO and head-neck paraganglioma; relies on active DOPA transport. Best for MEN2- and NF1-associated PHEO.
- [⁶⁸Ga]-DOTATATE PET/CT (somatostatin receptor PET): PPGL tumors express somatostatin receptors (particularly SSTR2 and SSTR3). [⁶⁸Ga]-DOTATATE PET/CT has become the preferred functional imaging in most guidelines for metastatic PHEO/PGL, with sensitivity exceeding 90% for both SDH-mutant and SDH-wildtype tumors — superior to MIBG SPECT for many patient subgroups. Also used for peptide receptor radionuclide therapy (PRRT) patient selection (lutetium-177 DOTATATE). The Endocrine Society recommends [⁶⁸Ga]-DOTATATE or [¹⁸F]-FDG PET/CT as the preferred modalities for metastatic disease.
Genetic Testing
Germline genetic testing is recommended for all patients with PHEO/PGL by the Endocrine Society (2014) and European guidelines (ESMO, EES). Rationale: the 35–40% hereditary fraction means universal testing identifies far more genetic carriers than selective testing based on clinical features (bilateral disease, young age, positive family history) alone. Genetic diagnosis:
- Directs surveillance for associated tumors in the same syndrome
- Determines the appropriate functional imaging modality (e.g., FDOPA for MEN2/NF1; FDG or DOTATATE for SDH)
- Guides intensity of surveillance for malignancy risk (SDHB)
- Enables cascade testing and surveillance of first-degree family members
- Has therapeutic implications (e.g., belzutifan for VHL syndrome)
Standard germline panel includes at minimum: RET, VHL, SDHB, SDHD, SDHC, SDHA, NF1, MAX, TMEM127. Expanded panels add FH, SDHAF2, MDH2, and others. Somatic tumor sequencing is complementary, identifying driver mutations in the non-hereditary fraction and detecting clonal ATRX mutations that predict metastatic behavior.
7. Treatment
Preoperative Medical Preparation
Adequate preoperative medical preparation is the single most critical factor in reducing perioperative mortality from PHEO surgery. The goal is to block the hemodynamic effects of catecholamine surges during anesthesia induction and tumor manipulation, restore circulating volume, and prevent reflex tachycardia upon alpha-blockade.
Alpha-adrenergic blockade — ALWAYS before beta-blockade:
- Alpha-blockade is initiated a minimum of 7–14 days before surgery (ideally 2–4 weeks).
- Why alpha before beta: Beta-blockade in the presence of alpha-mediated vasoconstriction would block the compensatory beta-2-mediated vasodilation, leaving alpha vasoconstriction unopposed — precipitating a hypertensive crisis and severe reflex bradycardia. This is the critical pharmacologic principle of PHEO management.
- Phenoxybenzamine (non-selective, irreversible alpha-1/alpha-2 blocker): The traditional first-line agent in North America. Starting dose: 10 mg twice daily, titrated by 10–20 mg every 2–3 days to a target dose of 20–100 mg/day in divided doses. The irreversible binding provides sustained blockade regardless of intraoperative catecholamine surges. Disadvantages: prolonged post-operative hypotension (requires IV fluids and vasopressors), reflex tachycardia, nasal congestion, orthostatic hypotension. Target: systolic BP 100–130 mmHg seated; systolic BP allowed to fall to 80–90 mmHg standing (orthostatic hypotension is a sign of adequate blockade).
- Selective alpha-1 blockers (doxazosin, prazosin, terazosin): Preferred in many European centers and increasingly in North America. Competitive (reversible) alpha-1 blockade; fewer side effects than phenoxybenzamine; shorter duration post-operatively (shorter-acting vasoactive properties are actually an advantage for post-op management). Doxazosin: start 2 mg daily, titrate to 8–32 mg/day. Well tolerated. Some surgeons argue phenoxybenzamine provides more complete intraoperative protection; randomized comparative data are limited.
Beta-blockade (added only after adequate alpha-blockade):
- Initiated only after alpha-blockade is established (typically 5–7 days later).
- Indications: persistent tachycardia (heart rate greater than 100 bpm), tachyarrhythmia, or EPI-predominant tumor (high risk of catecholamine cardiomyopathy).
- Cardioselective beta-1 blockers preferred: metoprolol succinate 25–100 mg/day; atenolol.
- Non-selective beta-blockers (propranolol, nadolol) are acceptable but provide no advantage over selective agents in this context.
Calcium channel blockers:
- Nicardipine, nifedipine, or amlodipine may be used as adjuncts to alpha-blockade to improve blood pressure control, particularly when alpha-blockade alone is insufficient at tolerated doses. Also useful for preventing coronary vasospasm in epinephrine-secreting tumors.
Metyrosine (alpha-methyltyrosine):
- Competitive inhibitor of tyrosine hydroxylase, the rate-limiting enzyme in catecholamine synthesis; reduces catecholamine production by 40–80%. Used as adjunct to alpha-blockade in large tumors, metastatic PHEO, or when complete alpha-blockade is difficult to achieve. Dose: 250–500 mg four times daily. Adverse effects: sedation, extrapyramidal symptoms, crystalluria (adequate hydration essential).
Preoperative volume expansion:
- High-salt diet (greater than 5 g NaCl/day) and liberal fluid intake (2–3 L/day) are instituted 1–2 weeks before surgery to restore contracted blood volume. This blunts the post-resection hypotension. Intravenous normal saline loading in the 12 hours before surgery at many centers.
Surgical Resection
- Laparoscopic adrenalectomy: The standard of care for unilateral, non-metastatic adrenal PHEO. Minimally invasive approach (transperitoneal or retroperitoneal) achieves equivalent oncologic outcomes with less morbidity, shorter hospitalization, and faster recovery than open surgery. Tumor size greater than 6 cm, suspected local invasion, or malignancy concern may favor conversion to or primary open surgery.
- Open adrenalectomy: Preferred for very large (greater than 6 cm) adrenal PHEO, suspected local invasion, and metastatic disease requiring debulking.
- Cortical-sparing adrenalectomy: Considered in bilateral PHEO (MEN2A, VHL) to preserve adrenocortical function and avoid permanent adrenal insufficiency. Preserves sufficient cortical tissue for steroid independence in approximately 60–80% of bilateral cases. Higher risk of local PHEO recurrence (5–10%) compared to total bilateral adrenalectomy.
- Anesthesia considerations: Short-acting vasoactive agents (IV phentolamine, nicardipine, sodium nitroprusside) should be immediately available for intraoperative hypertensive crises. IV labetalol (combined alpha/beta blocker) or esmolol for tachycardia. Magnesium sulfate is used by some anesthesiologists as an adjunct (inhibits catecholamine release and vasodilates). IV phenylephrine or norepinephrine are needed for post-resection hypotension after tumor is removed. Blood glucose monitoring every 30 minutes intraoperatively — rebound hypoglycemia from insulin release after catecholamine withdrawal.
- Intraoperative PTH monitoring analogy: Some centers now perform real-time plasma catecholamine or metanephrine monitoring during PHEO surgery, analogous to intraoperative PTH monitoring in parathyroid surgery, to confirm complete resection in complex cases.
Postoperative Management
- Hypotension is the primary postoperative concern; managed with IV fluids and vasopressors as needed.
- Hypoglycemia monitoring for 24–48 hours post-resection (rebound insulin surge).
- Blood pressure assessment at 2–4 weeks; some patients require continuation of antihypertensives if residual essential hypertension is present or endothelial/vascular changes from chronic catecholamine excess persist.
- Biochemical cure assessment: plasma free metanephrines measured at 2–6 weeks postoperatively. Normalization confirms complete resection. Persistent elevation indicates residual tumor or metastatic disease.
- Cortisol stimulation testing if bilateral adrenalectomy was performed; prescribe medical alert identification and emergency hydrocortisone kit.
Management of Metastatic/Malignant Pheochromocytoma
Metastatic PHEO is defined by the presence of chromaffin cell-containing lesions at sites lacking normal chromaffin tissue (lymph nodes, liver, lung, bone) — where PHEO cannot have arisen de novo. Metastatic disease affects approximately 10–17% of adrenal PHEOs and is much higher in SDHB-mutant and extra-adrenal paragangliomas.
- Surgical debulking: When feasible, reduces tumor burden and catecholamine excess, improving symptom control and potentially survival.
- [¹³¹I]-MIBG therapy (high-dose therapeutic MIBG): Approved by FDA (Azedra, iobenguane I-131) for MIBG-avid unresectable or metastatic PHEO/PGL in patients age 12 and older. Objective response rate approximately 25%; significant disease control (stable disease) in an additional 30–40%. Requires hospital isolation for radiation safety. Can cause bone marrow suppression.
- Peptide receptor radionuclide therapy (PRRT) with [¹⁷⁷Lu]-DOTATATE (Lutathera): For somatostatin receptor-positive (SSTR-positive) PHEO/PGL; most tumors express SSTR2. Approved for gastroenteropancreatic NETs; used off-label for PPGL with increasing evidence of efficacy. Now recommended by the Endocrine Society as a therapeutic option for SSTR-positive metastatic PHEO.
- Cytotoxic chemotherapy: CVD regimen (cyclophosphamide, vincristine, dacarbazine) is the standard chemotherapy for progressive metastatic PHEO, with objective response rates of 40–50%, though complete responses are rare and durability is limited. Used when MIBG therapy is not applicable.
- Targeted therapies:
- Belzutifan (HIF-2α inhibitor): FDA-approved for VHL disease-associated hemangioblastomas, RCC, and pNET. Activity in VHL-associated PHEO under investigation.
- Sunitinib, cabozantinib: VEGFR/c-MET/RET tyrosine kinase inhibitors; activity reported in case series for progressive metastatic PPGL; ongoing clinical trials.
- Temozolomide: Single-agent activity in SDH-mutant tumors (which have a hypermethylator phenotype that predicts response to alkylating agents).
- Bone-targeted therapy: Denosumab or bisphosphonates for bone metastases.
- Symptom control: Alpha-blockade (phenoxybenzamine, doxazosin) and/or metyrosine for ongoing catecholamine excess. Calcium channel blockers for refractory hypertension.
8. Complications
From the Tumor Itself
- Hypertensive crisis: Uncontrolled catecholamine surges producing BP greater than 200/120 mmHg; can precipitate hypertensive encephalopathy, hemorrhagic or ischemic stroke, aortic dissection, acute coronary syndrome, acute pulmonary edema, and death.
- Catecholamine cardiomyopathy: Dilated cardiomyopathy or Takotsubo syndrome from chronic or acute catecholamine cardiotoxicity. Reversible in many patients after tumor removal, though permanent myocardial fibrosis may develop in severe or prolonged cases.
- Arrhythmias: Atrial fibrillation, ventricular tachycardia, ventricular fibrillation — from catecholamine-induced cardiac electrical instability. Can cause sudden death.
- Multi-organ failure: Severe, prolonged catecholamine crises can cause ischemic injury to the liver, kidneys, bowel, and limbs from intense vasoconstriction.
- Tumor-associated polycythemia: EPO-producing PHEO/PGL (rare); or polycythemia from HIF-2α-driven erythropoietin production (as in EPAS1 mutation-associated syndrome).
From Surgery (Unprepared or Inadequately Prepared)
- Intraoperative hypertensive crisis leading to cardiovascular catastrophe
- Post-resection profound hypotension and circulatory collapse
- Rebound hypoglycemia (insulin surge after catecholamine withdrawal)
- Adrenal insufficiency after bilateral adrenalectomy
From Metastatic Disease
- Skeletal metastases causing pathologic fractures, hypercalcemia, spinal cord compression
- Hepatic metastases causing hepatic failure in advanced disease
- Persistent treatment-refractory hypertension
- Quality of life impairment from uncontrolled catecholamine secretion
9. Prognosis
- Benign (localized) PHEO: After complete surgical resection with adequate preoperative preparation, the 5-year survival rate approaches 95–100%, essentially equivalent to age-matched controls. Blood pressure normalizes in 50–70% of patients; the remainder require antihypertensive medication (residual essential hypertension or persistent vascular remodeling).
- Recurrence after surgery: Recurs in approximately 10–15% of apparently benign sporadic PHEO within 10 years of surgery; rate is higher in hereditary syndromes (particularly SDH-mutant, VHL, MEN2). Biochemical surveillance (plasma free metanephrines) annually for life is recommended for all patients after PHEO resection.
- Metastatic PHEO: 5-year survival approximately 40–60% overall; highly variable depending on SDHB status (SDHB-mutant: shorter survival), rate of disease progression, and response to therapy. Slow-growing tumors may remain stable for years; rapidly progressive disease has poor prognosis. Complete surgical resection of oligometastatic disease can achieve durable remissions.
- SDH mutations: SDHB carriers have the highest risk of aggressive disease; the Endocrine Society recommends annual biochemical and clinical surveillance and cross-sectional imaging every 2 years in SDHB-mutant PHEO/PGL.
- MEN2-associated PHEO: Rarely malignant (less than 5%); prognosis primarily determined by MTC. Excellent prognosis after adrenalectomy when detected as part of MEN2 surveillance protocols.
- Cardiovascular outcomes: Hypertension, cardiomyopathy, and left ventricular hypertrophy may not fully reverse after tumor resection, particularly if catecholamine exposure was prolonged. Long-term cardiovascular surveillance is warranted.
10. Prevention
- Genetic surveillance programs for hereditary syndromes:
- MEN2A carriers: annual biochemical screening for PHEO (plasma free metanephrines) beginning at age 11 (or from time of positive RET genetic testing), with clinical adrenal imaging at diagnosis of MEN2A and before any planned neck surgery.
- VHL syndrome: annual plasma free metanephrines and abdominal imaging beginning at age 10–15, with comprehensive multiorgan surveillance per VHL Alliance guidelines.
- SDH-mutant carriers: annual biochemical screening from childhood or time of positive genetic testing; cross-sectional imaging every 2 years.
- NF1: selective biochemical screening for hypertensive NF1 patients before surgical or procedural intervention; index of suspicion should be high given PHEO prevalence of 0.1–3.3%.
- Avoid precipitating medications in known PHEO: Beta-blockers without preceding alpha-blockade, glucagon, metoclopramide, high-dose corticosteroids, contrast without adequate alpha-blockade.
- Biochemical evaluation of all adrenal incidentalomas: Routine exclusion of PHEO (plasma free metanephrines) before any adrenal surgery or biopsy, regardless of imaging appearance.
- Cascade genetic testing: First-degree relatives of patients with confirmed germline PPGL-associated mutations should receive genetic counseling and testing. Asymptomatic mutation carriers with confirmed pathogenic variants should enter active surveillance programs.
- Long-term postoperative surveillance: Annual plasma free metanephrines for life; imaging when biochemistry is abnormal or in high-risk patients (SDHB, extra-adrenal PGL). Abdominal/pelvic imaging every 2 years in SDHB-mutant patients given high recurrence and malignancy risk.
11. Recent Research
- Belzutifan (MK-6482, Welireg) for VHL-associated PHEO: The Phase 2 LITESPARK-004 trial of belzutifan (a first-in-class HIF-2α inhibitor) demonstrated a 91% disease control rate in VHL-associated PHEO/PGL with an objective response rate of 26%. FDA expanded the approved indication for belzutifan to include VHL-associated pNET and hemangioblastomas in 2021. Studies specifically evaluating its activity in PHEO are ongoing.
- [¹⁷⁷Lu]-DOTATATE PRRT for SSTR-positive PHEO: Multiple Phase 2 trials and retrospective series demonstrate objective response rates of 25–37% and disease control rates exceeding 80% for SSTR-positive metastatic PHEO/PGL. The COMPETE Phase 3 trial (PRRT vs. sunitinib for progressive SSTR-positive PHEO/PGL) has completed enrollment; results are pending but will define the evidence base for this approach.
- Comprehensive genomic landscapes: Multi-center international collaborations (European PPGL Consortium; TCGA) have defined the molecular landscape of PPGL comprehensively. Integration of transcriptomics, methylomics, and proteomics identifies cluster 1 and cluster 2 tumors with high fidelity and reveals druggable targets. ATRX loss is confirmed as a robust biomarker of metastatic behavior across all PPGL genetic subtypes.
- Circulating tumor DNA (ctDNA) and liquid biopsy: Detection of ctDNA harboring SDHB, VHL, or ATRX mutations in plasma is being evaluated as a non-invasive biomarker for detecting residual disease, early recurrence, and monitoring treatment response in metastatic PHEO — complementing plasma free metanephrines.
- Prophylactic adrenalectomy timing in MEN2: The PANES study prospective data support earlier prophylactic bilateral adrenalectomy (at time of thyroidectomy or in the third decade) in MEN2A patients with detected adrenal lesions or early biochemical abnormalities, reducing the risk of operating on an established large tumor with higher intraoperative risk.
- Retroperitoneal laparoscopic adrenalectomy (RPLA) refinements: Technical refinements in the posterior retroperitoneoscopic approach allow complete gland removal with minimal bowel mobilization; particularly advantageous for bilateral PHEO (single positioning), redo surgery, and obese patients.
- Temozolomide for SDH-mutant PPGL: The hypermethylator phenotype of SDH-deficient PPGL (from succinate-mediated inhibition of TET enzymes) correlates with sensitivity to alkylating agents. Case series and retrospective analyses demonstrate objective responses to temozolomide in SDH-mutant tumors that fail MIBG therapy and CVD chemotherapy, providing a rational targeted approach pending prospective validation.
- Immunotherapy in PPGL: Preliminary data from Phase 2 trials of anti-PD1/PD-L1 agents (pembrolizumab, atezolizumab) as monotherapy demonstrate modest response rates (10–15%) in unselected metastatic PPGL. Combinations with antiangiogenic agents (cabozantinib, sunitinib) and with PRRT are under investigation to improve efficacy.
12. References
- Lenders JWM, Duh QY, Eisenhofer G, et al. Pheochromocytoma and paraganglioma: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2014;99(6):1915-1942.
- Pacak K, Eisenhofer G, Ahlman H, et al. Pheochromocytoma: recommendations for clinical practice from the First International Symposium. Nat Clin Pract Endocrinol Metab. 2007;3(2):92-102.
- Dahia PLM. Pheochromocytoma and paraganglioma pathogenesis: learning from genetic heterogeneity. Nat Rev Cancer. 2014;14(2):108-119.
- Crona J, Taïeb D, Pacak K. New perspectives on pheochromocytoma and paraganglioma: toward a molecular classification. Endocr Rev. 2017;38(6):489-515.
- Eisenhofer G, Lenders JWM, Linehan WM, Walther MM, Goldstein DS, Keiser HR. Plasma normetanephrine and metanephrine for detecting pheochromocytoma in von Hippel-Lindau disease and multiple endocrine neoplasia type 2. N Engl J Med. 1999;340(24):1872-1879.
- Lenders JWM, Pacak K, Walther MM, et al. Biochemical diagnosis of pheochromocytoma: which test is best? JAMA. 2002;287(11):1427-1434.
- Brauckhoff M, Stock K, Stock S, et al. Limitations of intraoperative adrenal remnant volume measurement in patients undergoing subtotal adrenalectomy. World J Surg. 2008;32(5):863-872.
- Ilias I, Pacak K. Current approaches and recommended algorithm for the diagnostic localization of pheochromocytoma. J Clin Endocrinol Metab. 2004;89(2):479-491.
- Carrasquillo JA, Pandit-Taskar N, Chen CC. I-131 metaiodobenzylguanidine therapy of pheochromocytoma and paraganglioma. Semin Nucl Med. 2016;46(3):203-214.
- Pryma DA, Chin BB, Noto RB, et al. Efficacy and safety of high-specific-activity ¹³¹I-MIBG therapy in patients with advanced pheochromocytoma or paraganglioma. J Nucl Med. 2019;60(5):623-630.
- Fassnacht M, Arlt W, Bancos I, et al. Management of adrenal incidentalomas: European Society of Endocrinology clinical practice guideline in collaboration with the European Network for the Study of Adrenal Tumors. Eur J Endocrinol. 2016;175(2):G1-G34.
- Jimenez C, Rohren E, Habra MA, et al. Current and future treatments for malignant pheochromocytoma and sympathetic paraganglioma. Curr Oncol Rep. 2013;15(4):356-371.
- Amar L, Baudin E, Burnichon N, et al. Succinate dehydrogenase B gene mutations predict survival in patients with malignant pheochromocytomas or paragangliomas. J Clin Endocrinol Metab. 2007;92(10):3822-3828.
- Jonasch E, Donskov F, Iliopoulos O, et al. Belzutifan for renal cell carcinoma in von Hippel-Lindau disease. N Engl J Med. 2021;385(22):2036-2046.
- Taïeb D, Jha A, Treglia G, Pacak K. Molecular imaging and radionuclide therapy of pheochromocytoma and paraganglioma in the era of genomic characterization of disease subgroups. Endocr Relat Cancer. 2019;26(11):R627-R652.
- Corssmit EPM, Snel M, Kapiteijn E. Malignant pheochromocytoma and paraganglioma: management options. Curr Opin Oncol. 2020;32(1):20-26.
- Lenders JWM, Eisenhofer G. Update on modern management of pheochromocytoma and paraganglioma. Endocrinol Metab Clin North Am. 2019;48(3):647-667.
- Neumann HPH, Young WF Jr, Eng C. Pheochromocytoma and paraganglioma. N Engl J Med. 2019;381(6):552-565.
Research Papers
The following PubMed topic searches aggregate the current peer-reviewed literature. Each link opens a live PubMed query — results update as new studies are indexed.
- PubMed — pheochromocytoma
- PubMed — paraganglioma
- PubMed — plasma metanephrines
- PubMed — urinary fractionated metanephrines
- PubMed — alpha adrenergic blockade pheochromocytoma
- PubMed — phenoxybenzamine
- PubMed — MIBG scan pheochromocytoma
- PubMed — laparoscopic adrenalectomy
- PubMed — SDHB SDHD mutation
- PubMed — von Hippel-Lindau pheochromocytoma
- PubMed — MEN2 pheochromocytoma
- PubMed — malignant pheochromocytoma
Connections
- Hyperparathyroidism
- Hypertension
- Atrial Fibrillation
- Addison's Disease
- Cushing's Syndrome
- Stress Management
- Tyrosine
- Cardiomyopathy
- Heart Failure
- Stroke
- Anxiety
- Headache
- Diabetes
- Diabetes Insipidus
- Calcium
- Chest Pain
- Thyroid Cancer
- Hives Flushing