Sleep, Stress & Cortisol in Insulin Resistance
If your fasting insulin is climbing, your waistline is widening, and nothing about your diet seems to explain it — look at your sleep and your stress. The hormones that rise when you are tired or overwhelmed are the same ones that drive glucose into your bloodstream and fat into your belly. You can eat a perfect diet and still be insulin resistant if you are sleeping six hours a night, grinding your teeth about work at 2 a.m., or quietly choking off your airway every thirty seconds because of undiagnosed sleep apnea.
This article walks through the science linking short sleep, circadian disruption, and chronic stress to insulin resistance — and, more importantly, what you can actually do about it tonight.
Table of Contents
- Short Sleep Drops Insulin Sensitivity by 40%
- Circadian Disruption and Shift Work
- Obstructive Sleep Apnea as a Root Cause
- Cortisol, Gluconeogenesis, and the Liver
- Chronic Stress and Visceral Belly Fat
- The HPA Axis in Plain English
- Testing Cortisol: Morning Levels, Saliva, Dexamethasone
- Cushing's Syndrome vs. "Pseudo-Cushing's"
- Lifestyle Interventions That Actually Move the Needle
- Supplements: Magnesium, Apigenin, Ashwagandha
- When to Push for a Sleep Study
- Key Research Papers
- PubMed Topic Searches
- Connections
- Featured Videos
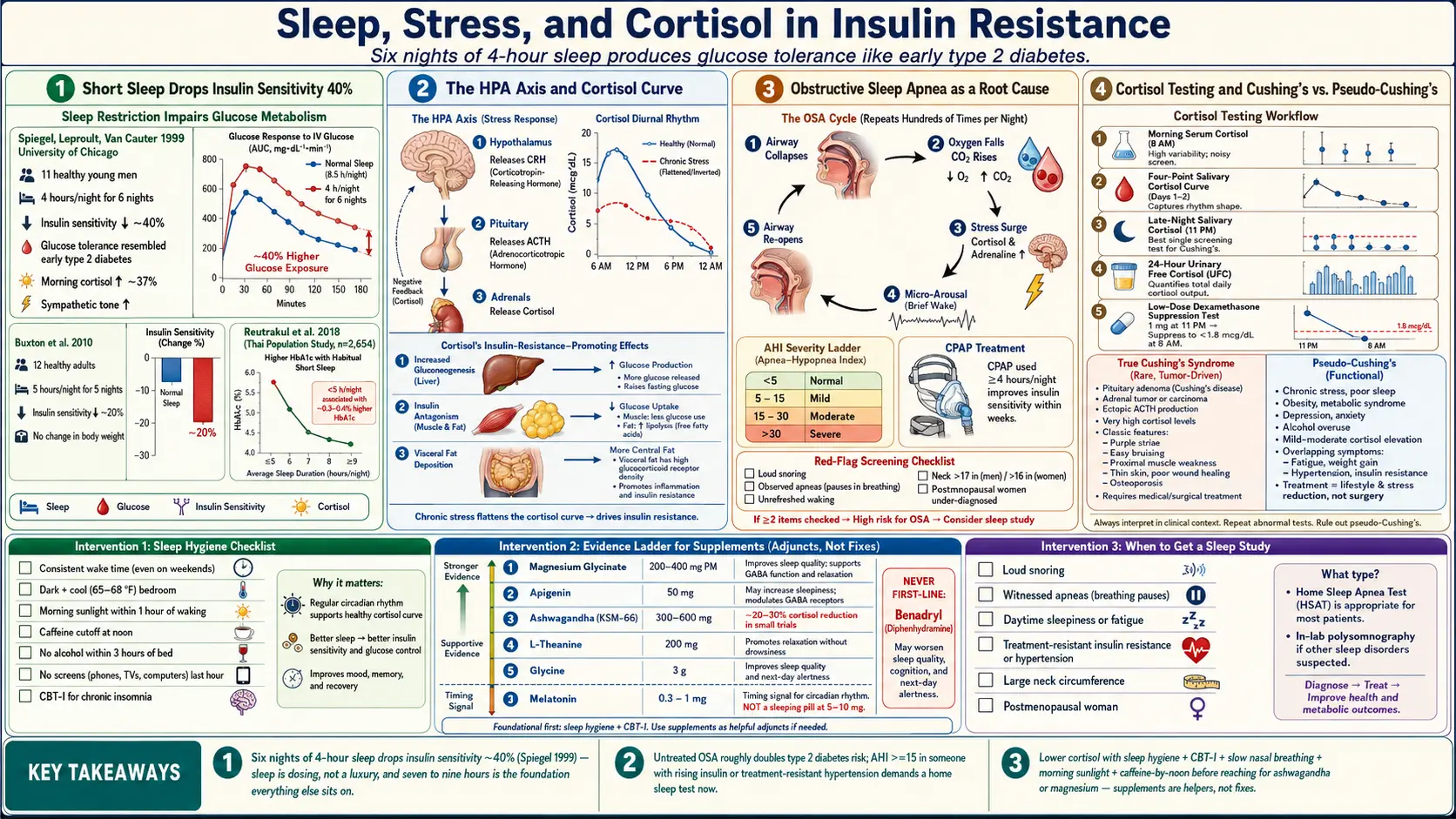
Short Sleep Drops Insulin Sensitivity by 40%
In 1999, a small but now-famous study by Karine Spiegel, Rachel Leproult, and Eve Van Cauter at the University of Chicago locked 11 healthy young men in a sleep lab and cut their time in bed to 4 hours a night for 6 nights. At the end of the week, their glucose tolerance looked like that of someone in early type 2 diabetes. Insulin sensitivity dropped roughly 40%. Morning cortisol rose. Sympathetic nervous system activity (fight-or-flight tone) climbed.
Then they were allowed to catch up on sleep — and everything normalized. The body was not broken. It was responding rationally to a stressor. But the implication is heavy: if you live chronically on 5–6 hours of sleep, you are walking around in a permanent state of induced insulin resistance, regardless of what you ate for lunch.
Follow-up work by Orfeu Buxton's group (2010) confirmed the effect under more real-world conditions: five nights of 5-hour sleep cut insulin sensitivity by about 20%, without any change in weight. Sirimon Reutrakul and colleagues have since linked habitual short sleep (less than 6 hours) with measurably higher HbA1c, higher fasting insulin, and greater long-term diabetes risk in population studies.
The practical takeaway: sleep is not a luxury you earn after fixing your diet. It is the foundation the diet sits on. Seven to nine hours is not a suggestion — for your pancreas, it is dosing.
Circadian Disruption and Shift Work
Your pancreas, liver, and muscle cells all have internal clocks. They expect glucose to arrive during the day and to be quiet at night. When you eat a large meal at 11 p.m. or work a rotating night shift, you feed glucose into a system that has already clocked out — and your insulin response becomes sluggish and disordered.
Studies of nurses, long-haul drivers, and airline crews consistently show that long-term shift work raises type 2 diabetes risk by roughly 40–60%, independent of body weight. Even short-term lab simulations of jet lag produce measurable drops in insulin sensitivity within days.
You cannot always avoid irregular hours, but you can cluster your eating. Time-restricted eating — finishing your last meal 3 hours before bed and compressing most calories into daylight — lets the circadian machinery recover even if your sleep schedule is imperfect.
Obstructive Sleep Apnea as a Root Cause
If you are overweight, snore, wake unrefreshed, or notice your partner nudging you because you stopped breathing — untreated obstructive sleep apnea (OSA) may be the biggest single driver of your insulin resistance.
Here is what is happening: every time your airway collapses, oxygen falls, carbon dioxide rises, and your body dumps a burst of cortisol and adrenaline to wake you up just enough to re-open the airway. You do not remember these micro-arousals, but they can happen hundreds of times a night. Each one is a small surge of stress hormones and a small spike of glucose. Multiplied across years, it hammers insulin sensitivity, raises blood pressure, and fattens the liver.
The key number is the Apnea-Hypopnea Index (AHI), measured during a sleep study:
- AHI < 5 — normal.
- AHI 5–15 — mild OSA.
- AHI 15–30 — moderate OSA.
- AHI > 30 — severe OSA.
Moderate-to-severe OSA roughly doubles the risk of developing type 2 diabetes. The good news: CPAP therapy, used for at least 4 hours a night, measurably improves insulin sensitivity within weeks to months. Weight loss of 10% can halve the AHI. Side- or stomach-sleeping, avoiding alcohol within 3 hours of bed, and treating nasal congestion also help.
If you have any suspicion at all — ask your physician for a home sleep test. They run $200–$500 out of pocket and are usually covered when there is clinical suspicion.
Cortisol, Gluconeogenesis, and the Liver
Cortisol is not the villain it is sometimes painted as. It is the hormone that gets you out of bed in the morning, pulls you through a hard workout, and keeps you alive in a real emergency. The problem is chronic, low-grade elevation — cortisol that never comes down.
One of cortisol's jobs is to make sure glucose is available when you need it. It does this by:
- Gluconeogenesis — telling the liver to manufacture new glucose from amino acids and glycerol, even when you have not eaten.
- Antagonizing insulin — actively blunting insulin's signal at the muscle and fat cell.
- Promoting lipolysis centrally while encouraging visceral fat storage — fat around the organs, not under the skin.
So chronically high cortisol gives you more glucose in the blood and more resistance at the tissues that should absorb it. That is essentially the definition of insulin resistance — and it happens without a single donut.
Chronic Stress and Visceral Belly Fat
This is why the classic stressed-out executive grows a hard, round belly rather than soft subcutaneous fat on the hips and thighs. Visceral fat cells have a high density of cortisol receptors. They preferentially respond to high cortisol by taking up circulating fatty acids and storing them right next to the liver, pancreas, and intestines — exactly the worst real estate for metabolic health.
Visceral fat is not passive storage. It secretes inflammatory cytokines (TNF-alpha, IL-6) that further impair insulin signaling in muscle and liver. The loop feeds itself: stress raises cortisol, cortisol grows visceral fat, visceral fat drives inflammation, inflammation worsens insulin resistance, insulin resistance raises glucose, and the brain interprets high glucose as another stressor.
Breaking the loop requires lowering cortisol and shrinking the visceral depot — and the interventions that do one almost always do the other.
The HPA Axis in Plain English
The Hypothalamic-Pituitary-Adrenal (HPA) axis is the stress hormone assembly line. It has three stations:
- Hypothalamus (deep in the brain) senses a stressor and releases CRH (corticotropin-releasing hormone).
- Pituitary gland (just below the hypothalamus) responds to CRH by releasing ACTH (adrenocorticotropic hormone) into the bloodstream.
- Adrenal glands (sitting on top of the kidneys) respond to ACTH by releasing cortisol.
Cortisol then feeds back to the hypothalamus and pituitary to shut off its own production — a negative feedback loop. In a healthy axis, cortisol surges in the morning, gently declines through the day, and bottoms out around midnight. In chronic stress, sleep loss, or shift work, the curve flattens — high at night, low in the morning, with the brakes on the feedback loop partially broken.
A flat or inverted cortisol curve is one of the most consistent findings in metabolic syndrome, burnout, and chronic insomnia.
Testing Cortisol: Morning Levels, Saliva, Dexamethasone
If your doctor suspects cortisol excess is driving your metabolic picture, a few standard tests sort it out:
- Morning serum cortisol (8 a.m. blood draw). A useful screen but noisy — stress of the needle itself can raise it.
- Salivary cortisol curve (4 samples across the day at home). Captures the shape of the rhythm, not just one point. Very useful for identifying flat curves.
- Late-night salivary cortisol (11 p.m. sample). Should be near zero; elevation is one of the earliest signs of true cortisol excess.
- 24-hour urinary free cortisol. Measures total daily output, bypassing moment-to-moment noise.
- Low-dose dexamethasone suppression test. You take 1 mg of dexamethasone (a synthetic cortisol-like steroid) at 11 p.m. and measure cortisol at 8 a.m. In a healthy axis, the pituitary senses the dexamethasone and shuts off ACTH, so morning cortisol is suppressed below 1.8 μg/dL. Failure to suppress is a red flag for Cushing's.
Cushing's Syndrome vs. "Pseudo-Cushing's"
Two scenarios can produce a Cushing-like picture — central weight gain, insulin resistance, high blood pressure, easy bruising, mood changes — and the distinction matters enormously.
True Cushing's syndrome is caused by a tumor (pituitary adenoma, adrenal adenoma, or ectopic ACTH-producing tumor) pumping out excess cortisol. It is rare (about 40–70 new cases per million per year). It requires imaging, specialized testing, and usually surgery. Red flags include purple abdominal stretch marks more than 1 cm wide, proximal muscle weakness (trouble standing from a chair), unexplained bone fractures, and rapidly progressive features.
Pseudo-Cushing's looks similar on the surface but is driven by chronic stress, poor sleep, obesity, depression, or heavy alcohol use — functional overdrive of a normal HPA axis rather than a tumor. It is vastly more common. The clinical features are milder, and the dexamethasone suppression test usually does suppress cortisol (though sometimes incompletely).
If you have insulin resistance plus the Cushing-looking picture, your endocrinologist will run screening tests (late-night salivary cortisol, 24-hour urinary free cortisol, low-dose dexamethasone). If those are clearly abnormal, imaging follows. If they are only mildly off, the diagnosis is almost always pseudo-Cushing's — and the treatment is the lifestyle work below, not surgery.
Lifestyle Interventions That Actually Move the Needle
Everything that follows is low-risk, cheap, and evidence-backed. Stack several of them and cortisol drops within weeks.
Sleep hygiene basics.
- Consistent wake time — same time every day including weekends. This is the single most powerful circadian anchor. Sleep-in drift is worse than slightly shorter sleep.
- Dark bedroom — blackout curtains or a sleep mask. Even low levels of ambient light (streetlamp, LED standby) measurably raise overnight heart rate and morning insulin resistance.
- Cool bedroom — 65–68°F (18–20°C). Your core temperature needs to drop to initiate deep sleep.
- Morning sunlight — 10 minutes of outdoor light within an hour of waking sets the cortisol rhythm for the day.
- No screens in the last hour — blue light suppresses melatonin. If you must use screens, night-mode filters help; glasses that block blue light help more.
- Caffeine cutoff by noon — caffeine's half-life is about 5–6 hours. An afternoon coffee still has 25% of the caffeine circulating at midnight.
- No alcohol within 3 hours of bed — it knocks you out but destroys deep sleep and worsens apnea.
CBT-I (Cognitive Behavioral Therapy for Insomnia). The first-line evidence-based treatment for chronic insomnia — more effective than sleeping pills long-term, no side effects. Available through trained therapists or guided apps (Sleepio, Somryst, CBT-i Coach). A 6–8 week program is the standard. See the insomnia page for specifics.
Mindfulness and breathing. Ten minutes of slow nasal breathing (inhale 4 seconds, exhale 6 seconds) before bed activates the parasympathetic — "rest and digest" — nervous system and measurably lowers cortisol. Apps like Insight Timer, Calm, or Headspace work; so does a timer and silence.
Exercise — but not too late. Regular exercise is one of the strongest long-term cortisol regulators, but intense evening training (within 3 hours of bed) raises core temperature and adrenaline and can wreck sleep. Aim for morning or early-afternoon training when possible. See the exercise and muscle page for the full dose-response picture.
Avoid late evening food. Eating within 2–3 hours of bed raises overnight glucose and insulin, fragments sleep, and worsens reflux. A 12–14 hour overnight fast is a zero-cost metabolic intervention.
Supplements: Magnesium, Apigenin, Ashwagandha
Evidence for supplements in this space is modest — they are helpers, not fixes. Consider them only after the lifestyle basics above are actually in place.
- Magnesium glycinate, 200–400 mg in the evening. Magnesium is a cofactor for GABA activity and parasympathetic tone. Deficiency is common. The glycinate form is well-absorbed and easy on the gut. Citrate works but is laxative at higher doses. See the magnesium page.
- Apigenin, 50 mg before bed. A flavonoid found in chamomile and parsley that binds benzodiazepine receptors mildly and has modest anxiolytic and sleep-promoting effects in small trials.
- Ashwagandha (KSM-66 extract), 300–600 mg/day. An adaptogenic herb that has been shown in small randomized trials to lower morning cortisol by roughly 20–30% and reduce subjective stress. Avoid in pregnancy, in autoimmune thyroid disease, and alongside sedative medications. See the ashwagandha page.
- L-theanine, 200 mg, and glycine, 3 g, are also reasonable, mild, and well-tolerated additions.
What is not first-line: melatonin at high doses (it is a circadian timing signal, not a sleeping pill — 0.3–1 mg is physiologic; 5–10 mg pharmacologic and often counterproductive), diphenhydramine (Benadryl — it does not produce real sleep architecture and worsens cognition), and alcohol (obvious).
When to Push for a Sleep Study
Ask your physician for a sleep study if any of the following apply:
- You snore loudly, or a partner has observed you stop breathing.
- You wake unrefreshed despite adequate time in bed.
- You have daytime sleepiness (Epworth Sleepiness Scale > 10) or have fallen asleep driving.
- You have insulin resistance, prediabetes, or type 2 diabetes that is not responding to lifestyle change.
- You have hypertension that is resistant to medication.
- Your neck circumference is > 17 inches (men) or > 16 inches (women).
- You have a large tongue, recessed jaw, or crowded airway on exam.
- You are postmenopausal (OSA in women is dramatically under-diagnosed).
Home sleep tests (Level III studies) are now the usual first step — small portable devices measuring airflow, oxygen saturation, heart rate, and chest movement. Accurate for moderate-to-severe OSA; may miss mild cases, in which a full in-lab polysomnography is the next step. If you cannot get through your primary care doctor, telemedicine sleep clinics (Lofta, Sleepon, and similar) can order a home test directly for roughly $200–$400.
Key Research Papers
- Spiegel K, Leproult R, Van Cauter E. Impact of sleep debt on metabolic and endocrine function. Lancet. 1999;354(9188):1435–1439.
- Buxton OM, et al. Sleep restriction for 1 week reduces insulin sensitivity in healthy men. Diabetes. 2010;59(9):2126–2133.
- Reutrakul S, Van Cauter E. Interactions between sleep, circadian function, and glucose metabolism: implications for risk and severity of diabetes. Ann N Y Acad Sci. 2014.
- Buxton OM, et al. Adverse metabolic consequences in humans of prolonged sleep restriction combined with circadian disruption. Sci Transl Med. 2012.
- Punjabi NM, et al. Sleep-disordered breathing, glucose intolerance, and insulin resistance. Am J Epidemiol. 2004.
- Chandrasekhar K, Kapoor J, Anishetty S. A prospective, randomized double-blind, placebo-controlled study of safety and efficacy of a high-concentration full-spectrum extract of ashwagandha root in reducing stress and anxiety. Indian J Psychol Med. 2012.
PubMed Topic Searches
For further reading, the following PubMed searches return current peer-reviewed work:
- Sleep restriction and insulin sensitivity
- Shift work and type 2 diabetes risk
- Obstructive sleep apnea, insulin resistance, and CPAP
- Cortisol, gluconeogenesis, and insulin resistance
- Visceral adiposity, cortisol, and the HPA axis
- Dexamethasone suppression test and pseudo-Cushing's
- CBT-I, insomnia, and metabolic outcomes
- Ashwagandha and cortisol (randomized trials)
- Magnesium, sleep, and insulin sensitivity
- Time-restricted eating, circadian rhythm, and glucose
Connections
- Ashwagandha
- Insomnia
- Magnesium
- Exercise and Muscle Insulin Sensitivity
- Insulin Resistance
- Reversal Protocol: Diet and Fasting
- CGM for Non-Diabetics
- Fasting Insulin and HOMA-IR Testing
- Metformin, Berberine and GLP-1s
- Metabolic Syndrome Cardiovascular Risk
- NAFLD & MASLD Connection
- Insulin Resistance and PCOS
- Obstructive Sleep Apnea
- Cushing's Syndrome
- Visceral Fat
- Burnout
- Sleep Hygiene