NAFLD / MASLD: The Hepatic Arm of Insulin Resistance
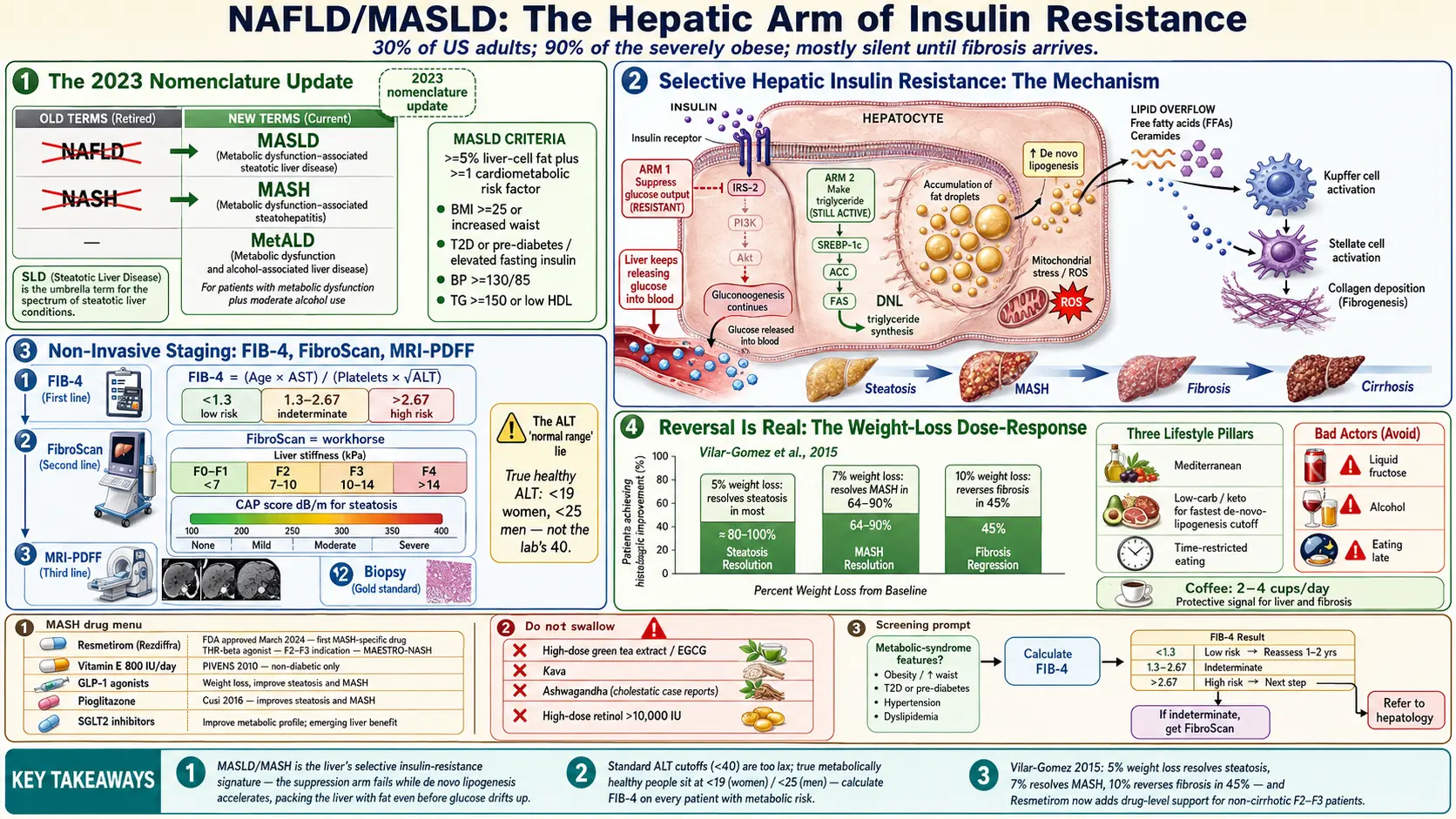
Table of Contents
- The 2023 Name Change: NAFLD Becomes MASLD
- MASLD Diagnostic Criteria
- How Common It Is — and Why It's Silent
- The Mechanism: Hepatic Insulin Resistance
- The ALT "Normal Range" Lie
- FIB-4, NAFLD Fibrosis Score, and ELF
- FibroScan: The Non-Invasive Workhorse
- MRI-PDFF and Liver Biopsy
- Staging: F0 to F4
- Reversal Is Real: The Weight-Loss Dose-Response
- Diet: Mediterranean, Low-Carb, TRE
- Fructose, Alcohol, and Coffee
- Drugs: Vitamin E, Resmetirom, GLP-1s, Pioglitazone
- Supplements to Avoid
- Who Should Be Screened
- Key Research Papers
- Connections
- Featured Videos
The 2023 Name Change: NAFLD Becomes MASLD
In June 2023, a multinational consensus convened by three major liver societies (AASLD, EASL, ALEH) retired the term non-alcoholic fatty liver disease (NAFLD) after more than four decades of use. In its place came a new name: metabolic dysfunction-associated steatotic liver disease (MASLD). The accompanying inflammatory form, formerly NASH (non-alcoholic steatohepatitis), was renamed MASH (metabolic dysfunction-associated steatohepatitis).
The change was not cosmetic. Three problems drove it. First, the old term defined the disease by what it was not — "non-alcoholic" — which is clinically awkward and tells you nothing about the actual mechanism. Second, "fatty" carried stigma; patients heard it as a personal failing. Third, the old framework excluded patients who drank any alcohol, even though the underlying metabolic biology is identical whether or not a person enjoys wine on weekends. A new category, MetALD, now covers people who have metabolic-driven steatosis and drink moderate-to-heavy amounts — because in real life, the two problems are routinely stacked.
If you were diagnosed before 2023 and your chart says "NAFLD" or "fatty liver," you have MASLD by the new criteria unless something unusual was going on (e.g., a genetic or drug-induced steatosis). The biology is the same. The testing is the same. The treatments are the same. Only the label has changed.
MASLD Diagnostic Criteria
MASLD requires two things at once:
- Hepatic steatosis — fat in at least 5% of liver cells, shown on imaging (ultrasound, FibroScan CAP score, MRI-PDFF) or biopsy.
- At least one cardiometabolic risk factor:
- Overweight or obesity (BMI ≥ 25 in most populations, ≥ 23 in Asian populations), or increased waist circumference.
- Type 2 diabetes or prediabetes / elevated fasting insulin.
- Hypertension (≥ 130/85 or on treatment).
- Elevated triglycerides (≥ 150 mg/dL) or low HDL (< 40 in men, < 50 in women), or on lipid-lowering therapy.
Almost everyone with steatosis meets at least one cardiometabolic criterion, which is the whole point. Liver fat is not a random accident — it is a downstream marker of systemic insulin resistance. If you have fat in your liver and none of the metabolic triggers, the steatosis is probably being driven by something else (alcohol, a medication, a rare genetic disorder) and needs a different workup.
How Common It Is — and Why It's Silent
MASLD is the single most common liver disease on the planet. Roughly 30% of US adults have it. Among people with type 2 diabetes, prevalence climbs to 55–70%. Among the severely obese, it exceeds 90%. About one in four people with MASLD progresses to MASH, where inflammation and fibrosis join the fat deposition, and a smaller subset progresses to cirrhosis or hepatocellular carcinoma.
Yet most people with MASLD have no idea they have it. The liver has no pain fibers in its parenchyma, so silent fibrosis can advance for years without a single symptom. By the time decompensated cirrhosis appears — ascites, variceal bleeding, hepatic encephalopathy, jaundice — the window for reversal has closed. The tragedy of MASLD is that it is both extremely common and extremely reversible in its early stages, but almost nobody catches it there.
You should assume MASLD is possible if you have any of the following: a waistline that has quietly expanded, a fasting glucose creeping above 100, a fasting triglyceride above 150, borderline blood pressure, PCOS, obstructive sleep apnea, or a family history of type 2 diabetes. This is not hypochondria — it is base rates.
The Mechanism: Hepatic Insulin Resistance
The liver is the central clearinghouse for incoming calories. When you eat carbohydrate, glucose floods the portal vein, and insulin tells the liver to do three things in sequence: take up glucose and store it as glycogen, turn off glucose production, and convert excess carbohydrate into triglyceride for storage (a process called de novo lipogenesis). When insulin signaling works, the system is elegant.
In insulin resistance, the liver's response becomes paradoxically selective. The suppression arm of insulin signaling — "stop making glucose" — weakens, so the liver keeps pouring glucose into the bloodstream even after meals. But the lipogenic arm — "make triglyceride" — remains fully on, and actually accelerates under chronically high insulin. This is called selective hepatic insulin resistance, and it is the reason insulin-resistant livers both raise blood sugar and simultaneously pack themselves with fat.
At the same time, fatty-acid oxidation — the liver's ability to burn stored fat — is impaired. Triglycerides accumulate in hepatocytes. Once the storage droplets exceed a threshold, they leak free fatty acids and ceramides that poison mitochondria, activate inflammatory Kupffer cells, and recruit stellate cells that lay down collagen. Steatosis turns into steatohepatitis; steatohepatitis turns into fibrosis; fibrosis, over years, turns into cirrhosis.
The driver at every step is insulin resistance. MASLD is not a liver disease that happens to coexist with diabetes. MASLD is the hepatic expression of the same systemic problem that causes type 2 diabetes, metabolic syndrome, and PCOS.
The ALT "Normal Range" Lie
When your doctor looks at your liver enzymes, the single most important number is ALT (alanine aminotransferase). Most US labs report the upper limit of normal as 40 U/L for both sexes. This cutoff is misleading in a way that matters clinically.
The "normal" was set decades ago using reference populations that, by today's standards, were not healthy — they included plenty of unrecognized fatty liver. When researchers restricted the reference group to truly metabolically healthy people (lean, non-diabetic, no viral hepatitis, no steatosis on imaging), the true upper limit of normal dropped to roughly 19 U/L for women and 25 U/L for men. An ALT of 35 may read as "within normal limits" on your report, but in a metabolically healthy person that number is distinctly high.
Practical translation: if your ALT is persistently above the mid-20s and you have any cardiometabolic risk factor, do not let a "normal" flag on your lab report end the conversation. Ask for a FIB-4 calculation and consider a FibroScan. Conversely — and this is equally important — a normal ALT does not rule out MASLD or even MASH with advanced fibrosis. Up to half of patients with significant fibrosis have ALTs in the normal range. You cannot screen for liver disease with ALT alone.
FIB-4, NAFLD Fibrosis Score, and ELF
Because ALT is unreliable as a standalone test, several composite scores were developed to estimate fibrosis from routinely available blood work.
FIB-4 is the workhorse. The formula is:
FIB-4 = (Age × AST) / (Platelet count × √ALT)
Free calculators are everywhere — search "FIB-4 calculator" and any major hepatology site will have one. You already have all four inputs on any recent CBC plus comprehensive metabolic panel. Interpretation:
- < 1.3 — low probability of advanced fibrosis. Reasonable to recheck annually if you have metabolic risk factors.
- 1.3 to 2.67 — indeterminate. Move to FibroScan or ELF.
- > 2.67 — high probability of advanced fibrosis. Hepatology referral.
FIB-4 loses specificity in people over 65, where age inflates the number. The threshold for "low risk" is sometimes raised to 2.0 in older patients.
The NAFLD Fibrosis Score adds BMI, diabetes status, and albumin to the inputs and performs slightly better in some populations. The ELF test (Enhanced Liver Fibrosis) is a direct serum panel of three fibrosis biomarkers (hyaluronic acid, PIIINP, TIMP-1). It is more expensive and not always covered by insurance, but it has excellent performance for ruling advanced fibrosis in or out. Ask if your insurance covers it.
FibroScan: The Non-Invasive Workhorse
The single most useful test you have not heard of is the FibroScan. Officially called transient elastography, it is a bedside ultrasound-like probe that measures two things in about ten minutes, painlessly, without contrast or fasting:
- Liver stiffness in kilopascals (kPa) — a direct estimate of fibrosis. Roughly: < 7 kPa is F0–F1 (minimal), 7–10 kPa suggests F2 (moderate), 10–14 kPa suggests F3 (advanced), and > 14 kPa suggests F4 (cirrhosis). Interpretation adjusts for MASLD versus viral hepatitis.
- CAP score (controlled attenuation parameter, in dB/m) — a measure of steatosis (fat). Roughly: < 238 is normal, 238–260 is mild (S1), 260–290 is moderate (S2), and > 290 is severe (S3).
FibroScan is available at most hospital-based hepatology clinics, many gastroenterology practices, and an increasing number of obesity/metabolic clinics. Cash-pay prices typically run $100 to $300 in the US; insurance covers it with a fibrosis workup indication. It is the most important piece of equipment for staging MASLD without a biopsy, and for tracking response to treatment over months and years. If you have MASLD risk factors plus an indeterminate FIB-4, a FibroScan is the next step.
MRI-PDFF and Liver Biopsy
MRI-PDFF (proton-density fat fraction) is the research gold standard for quantifying liver fat, reported as a percentage. It is precise enough to detect changes of a few percentage points and is now the primary endpoint in most MASH drug trials. Availability is expanding but still concentrated at academic centers; expect $500–$1500 cash, variable insurance coverage. For routine care, FibroScan is sufficient. For enrollment in a trial or a question that FibroScan cannot answer, MRI-PDFF is the next step up.
Liver biopsy remains the ultimate arbiter of MASH versus simple steatosis, because only tissue can show the ballooning hepatocytes, inflammation, and fibrosis pattern that distinguishes MASH. Pathologists grade biopsies with the NAS (NAFLD Activity Score), which sums steatosis, ballooning, and lobular inflammation, and stage fibrosis separately on a 0–4 scale. Biopsies are invasive, carry a small risk of bleeding, and sample only 1/50,000th of the liver — so sampling error is real. In 2026, biopsy is reserved for cases where non-invasive tests are equivocal, where diagnosis is contested, or where a trial protocol requires it.
Staging: F0 to F4
Fibrosis stage is the strongest predictor of long-term mortality in MASLD. Inflammation matters, but what ultimately kills the liver is scar tissue. The scale is universal:
- F0 — no fibrosis.
- F1 — perisinusoidal or periportal fibrosis. Easily reversible.
- F2 — both perisinusoidal and periportal. Still highly reversible with sustained weight loss and metabolic correction.
- F3 — bridging fibrosis. Reversible but harder; this is the stage where drug therapy (Resmetirom, GLP-1s) most clearly adds value.
- F4 — cirrhosis. Architectural distortion. Partial reversal is possible; full reversal is not. Shifts the conversation to surveillance for hepatocellular carcinoma and portal hypertension.
The reason to know your stage is that it determines both your urgency and your expectations. F1 MASLD with a 10% weight loss over a year is essentially a cured problem. F3 MASLD needs an aggressive intervention plan, probably including a drug.
Reversal Is Real: The Weight-Loss Dose-Response
The most encouraging data in all of MASLD come from a single landmark study. In 2015, Vilar-Gomez and colleagues followed 293 biopsy-proven NASH patients through a year of intensive lifestyle intervention and re-biopsied them. The dose-response was clean:
- ≥ 5% body weight loss — steatosis resolved in most patients.
- ≥ 7% weight loss — NASH (MASH) resolved in 64–90%, depending on fibrosis stage.
- ≥ 10% weight loss — fibrosis regressed in 45% of patients, including those with bridging fibrosis.
Almost no other chronic disease has a reversal curve this steep. The implication is practical: if you have MASLD, losing 10% of your starting body weight and keeping it off for a year is, in most cases, curative or nearly so. The challenge is the sustained part — the people in that trial who regained the weight lost their biopsy improvements.
This is why the broader insulin-resistance reversal protocol — diet change plus muscle-building exercise — matters so much. Losing the weight is half the problem. Not regaining it is the other half, and it is the harder half.
Diet: Mediterranean, Low-Carb, TRE
No single eating pattern dominates the evidence, but three approaches have held up well in MASLD trials:
Mediterranean diet. The best-studied pattern for MASLD. High in olive oil, nuts, vegetables, legumes, fish; low in red and processed meat, refined carbohydrate, and sugar. Multiple trials show liver-fat reduction independent of weight loss.
Low-carb or ketogenic diet. Removing the raw substrate for de novo lipogenesis (refined carbohydrate and added sugar) drops liver fat faster than any other dietary lever, often within two weeks. Whether you need to go all the way to ketogenic depends on goals and tolerance; cutting added sugar and refined grain is the high-value first step.
Time-restricted eating (TRE). Compressing all eating into an 8- to 10-hour window gives the liver a long daily fasting interval to oxidize stored fat rather than laying down more. TRE works modestly well even without changing what you eat; combined with a Mediterranean or low-carb pattern, the effect compounds.
The worst thing you can do nutritionally with a fatty liver is graze all day on snacks, drink juice or sweetened beverages, and eat late into the evening. That pattern keeps insulin elevated for 14+ hours a day and drives de novo lipogenesis nonstop.
Fructose, Alcohol, and Coffee
Fructose is uniquely bad for the liver. Unlike glucose, which any cell can metabolize, fructose is processed almost exclusively in the liver, where it feeds directly into de novo lipogenesis without triggering the normal satiety or insulin response. Sucrose (table sugar) is half fructose; high-fructose corn syrup is 55% fructose. Fruit juice — even "freshly squeezed" — delivers fructose without the fiber and chewing that slow absorption from whole fruit. In MASLD, cutting liquid fructose (juice, soda, sweetened coffee drinks, sweet teas) is one of the single highest-yield changes you can make. Whole fruit is fine in moderation; juice is not.
Alcohol is not optional to cut back on. Every drink adds hepatic fat-synthesis substrate on top of what carbohydrate is already doing. The old cutoff of "up to two drinks a day for men" was based on cardiovascular data, not liver data. In MASLD specifically, even "moderate" drinking accelerates fibrosis. The practical advice for anyone with confirmed MASLD or advanced MASH is to eliminate alcohol or reduce to rare social occasions. If you cannot, you are now a candidate for the MetALD category, and your disease will progress faster than it would otherwise.
Coffee is protective. Two to four cups a day of coffee (caffeinated or decaf, black or with a splash of milk, but not loaded with syrup) is consistently associated with lower MASLD progression, lower fibrosis, and lower hepatocellular carcinoma risk. The mechanism probably involves chlorogenic acids and caffeine's effects on adenosine signaling in the liver. This is one of the few dietary exposures where the "more is better" signal extends to four cups a day for liver outcomes.
Drugs: Vitamin E, Resmetirom, GLP-1s, Pioglitazone
Vitamin E 800 IU daily. In the 2010 PIVENS trial, vitamin E at 800 IU/day improved MASH histology in non-diabetic adults. It remains a reasonable adjunct for non-diabetic MASH, though long-term high-dose vitamin E has been flagged for a small increase in prostate cancer risk in some cohorts. It is not first-line and should be discussed with a hepatologist.
Resmetirom (Rezdiffra). In March 2024, the FDA granted the first-ever approval for a MASH-specific drug: resmetirom, a thyroid hormone receptor-beta (THR-β) agonist that acts selectively in the liver to accelerate fat oxidation. The phase 3 MAESTRO-NASH trial (Harrison 2024) showed MASH resolution and fibrosis improvement in a meaningful minority of patients with F2–F3 fibrosis. It is approved for non-cirrhotic MASH with moderate to advanced fibrosis, taken orally once daily. This is a genuinely new option and has changed the conversation for F2–F3 patients who cannot reach their goals through lifestyle alone.
GLP-1 receptor agonists. Semaglutide (Ozempic, Wegovy) showed strong phase 2 MASH resolution signals; tirzepatide (Mounjaro, Zepbound) is in active trials with similar expectations. The weight-loss effect alone would be helpful; the emerging data suggest a direct liver effect on top of that. These drugs are discussed in more detail on the Metformin, Berberine, and GLP-1s page.
Pioglitazone. The 2016 Cusi trial showed that pioglitazone improves MASH histology even in patients without diabetes. It carries side effects (weight gain, edema, fracture risk in women, heart-failure caution) that have limited its use, but in the right patient — particularly someone with both type 2 diabetes and MASH — it remains a legitimate option.
SGLT2 inhibitors. Empagliflozin, dapagliflozin, and canagliflozin lower liver fat modestly and are already first-line for type 2 diabetes with cardiovascular or kidney risk. For a patient with both diabetes and MASLD, an SGLT2 inhibitor is a near-automatic pick.
Supplements to Avoid
The liver is the organ that metabolizes almost everything you swallow, and some popular supplements are directly hepatotoxic. If you have MASLD or MASH, avoid:
- High-dose green tea extract (EGCG). Standardized pill forms have caused acute liver failure at modest doses. Drinking green tea is fine; concentrated extracts are not.
- Kava. Banned or restricted in several countries for hepatotoxicity. Do not use.
- Ashwagandha. Case reports of cholestatic liver injury are accumulating. Use cautiously if at all.
- High-dose vitamin A / retinol. Above roughly 10,000 IU/day chronically, retinol is directly hepatotoxic. Beta-carotene is not the same problem.
- Anabolic steroids and "testosterone boosters" with undeclared ingredients — a common cause of drug-induced liver injury in younger patients.
- Unregulated herbal weight-loss products bought online, especially multi-ingredient powders. The literature on these is a rotating cast of liver-failure case reports.
If you are already taking a supplement and your ALT is rising, stop the supplement first and recheck labs in four to six weeks. The liver recovers quickly from drug-induced injury if the offending agent is removed early.
Who Should Be Screened
The 2023 AASLD guidance recommends that primary care check a FIB-4 annually in anyone with:
- Type 2 diabetes, or
- Two or more cardiometabolic risk factors (central obesity, hypertension, dyslipidemia, prediabetes).
FIB-4 is free, non-invasive, and calculable from any recent CBC plus CMP. Every metabolic-risk patient in a primary care panel should have a documented FIB-4 at least once a year, with FibroScan as the escalation step for indeterminate or elevated results. The reason this matters: by the time a patient presents with ascites or variceal bleeding from cirrhosis, the horse has left the barn. Catching fibrosis at F1 or F2 — while it is still reversible — requires active screening, not waiting for symptoms that will never come until it is too late.
If your primary care clinician is not running a FIB-4 on you despite metabolic risks, you can calculate it yourself from the labs you already have. A FIB-4 above 1.3 earns you the right to ask for a FibroScan.
Key Research Papers
Foundational publications on the metabolic-hepatic axis — from the original NAFLD/metabolic syndrome link, through global epidemiology, to the 2023 MASLD nomenclature shift and the first FDA-approved pharmacotherapy. Each citation links to the full text via DOI.
- Rinella ME, Lazarus JV, Ratziu V, et al. A Multisociety Delphi Consensus Statement on New Fatty Liver Disease Nomenclature. Hepatology. 2023;78(6):1966–1986.
- Younossi ZM, Koenig AB, Abdelatif D, Fazel Y, Henry L, Wymer M. Global Epidemiology of Nonalcoholic Fatty Liver Disease — Meta-Analytic Assessment of Prevalence, Incidence, and Outcomes. Hepatology. 2016;64(1):73–84.
- Marchesini G, Bugianesi E, Forlani G, et al. Nonalcoholic Fatty Liver, Steatohepatitis, and the Metabolic Syndrome. Hepatology. 2003;37(4):917–923.
- Vilar-Gomez E, Martinez-Perez Y, Calzadilla-Bertot L, et al. Weight Loss Through Lifestyle Modification Significantly Reduces Features of Nonalcoholic Steatohepatitis. Gastroenterology. 2015;149(2):367–378.
- Sanyal AJ, Chalasani N, Kowdley KV, et al. Pioglitazone, Vitamin E, or Placebo for Nonalcoholic Steatohepatitis (PIVENS). New England Journal of Medicine. 2010;362(18):1675–1685.
- Harrison SA, Bedossa P, Guy CD, et al. A Phase 3, Randomized, Controlled Trial of Resmetirom in NASH with Liver Fibrosis (MAESTRO-NASH). New England Journal of Medicine. 2024;390(6):497–509.
- Newsome PN, Sasso M, Deeks JJ, et al. FibroScan-AST (FAST) Score for the Non-Invasive Identification of Patients with Non-Alcoholic Steatohepatitis with Significant Activity and Fibrosis: A Prospective Derivation and Global Validation Study. Lancet Gastroenterology & Hepatology. 2020;5(4):362–373.
- Reaven GM. Banting Lecture 1988. Role of Insulin Resistance in Human Disease. Diabetes. 1988;37(12):1595–1607.
- Petersen MC, Shulman GI. Mechanisms of Insulin Action and Insulin Resistance. Physiological Reviews. 2018;98(4):2133–2223.
- Saltiel AR, Olefsky JM. Inflammatory Mechanisms Linking Obesity and Metabolic Disease. Journal of Clinical Investigation. 2017;127(1):1–4.
- Stanhope KL, Schwarz JM, Keim NL, et al. Consuming Fructose-Sweetened, Not Glucose-Sweetened, Beverages Increases Visceral Adiposity and Lipids and Decreases Insulin Sensitivity in Overweight/Obese Humans. Journal of Clinical Investigation. 2009;119(5):1322–1334.
- Newsome PN, Buchholtz K, Cusi K, et al. A Placebo-Controlled Trial of Subcutaneous Semaglutide in Nonalcoholic Steatohepatitis. New England Journal of Medicine. 2021;384(12):1113–1124.
Live PubMed Searches
Live PubMed queries that update as new papers are indexed.
- PubMed: MASLD nomenclature and consensus
- PubMed: NAFLD and de novo lipogenesis
- PubMed: FIB-4 fibrosis screening
- PubMed: FibroScan in MASLD
- PubMed: MRI-PDFF and liver fat
- PubMed: resmetirom and MAESTRO-NASH
- PubMed: semaglutide in MASH/NASH
- PubMed: weight loss and fibrosis regression
- PubMed: fructose and hepatic steatosis
- PubMed: coffee and liver fibrosis
Connections
- Diabetes
- Metabolic Syndrome
- Reversal Protocol: Diet and Fasting
- Fasting Insulin and HOMA-IR Testing
- Exercise and Muscle Insulin Sensitivity
- Metformin, Berberine and GLP-1s
- Polycystic Ovary Syndrome
- Insulin Resistance
- Non-Alcoholic Fatty Liver Disease
- Cirrhosis
- CGM for Non-Diabetics
- Insulin Resistance and PCOS
- Metabolic Syndrome Cardiovascular Risk
- Obesity
- Vitamin E
- GLP-1 Receptor Agonists
- Fatty Liver Disease
- Fasting