Insulin Resistance and PCOS
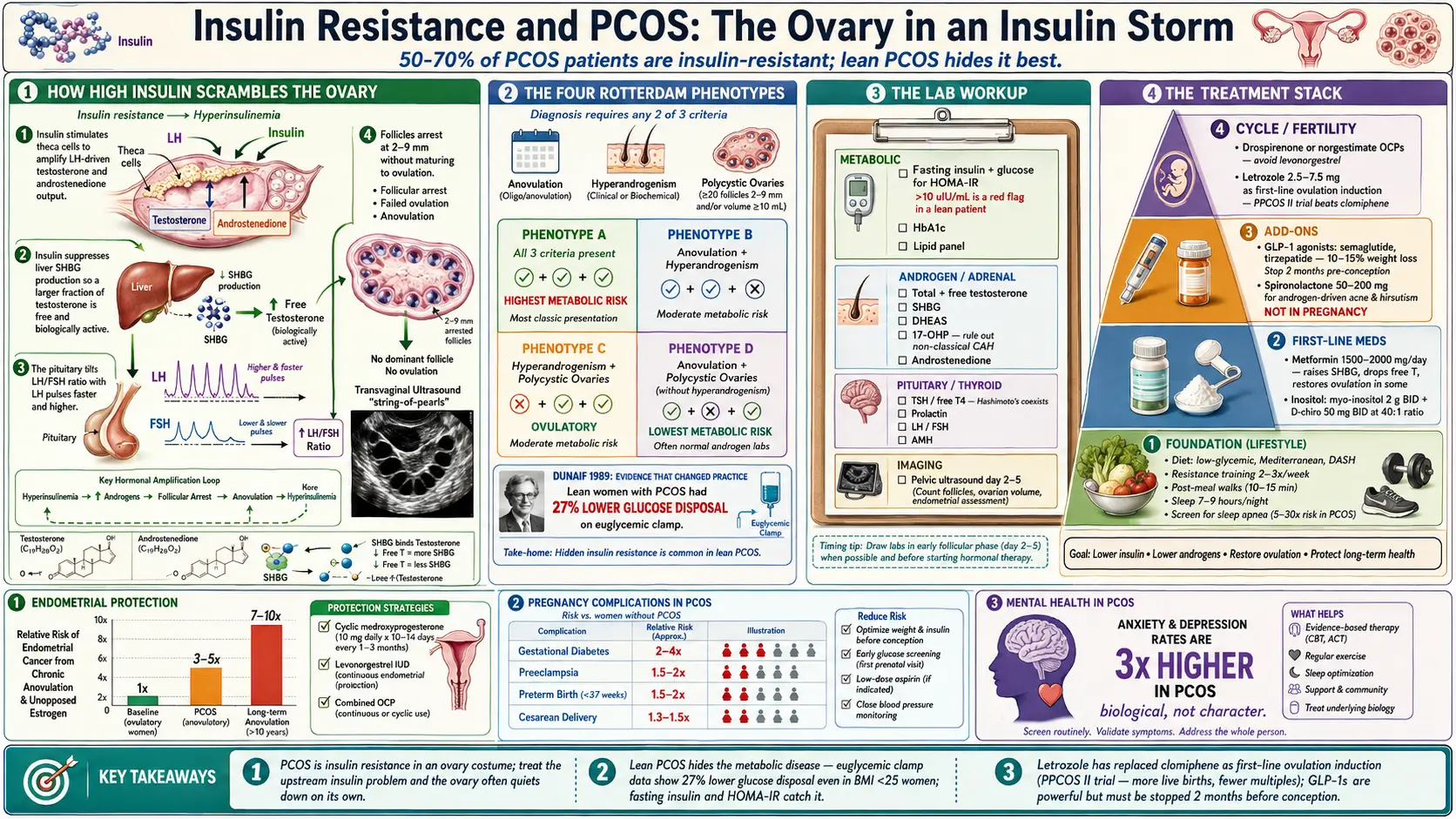
Table of Contents
- Why This Connection Matters
- How Common Is IR in PCOS?
- Lean PCOS — The Subset That Gets Missed
- The Mechanism: How High Insulin Scrambles Ovaries
- Rotterdam Criteria and the Four Phenotypes
- The Lab Workup: What to Order
- Metformin — The Workhorse
- Inositol — The Supplement With Real Data
- GLP-1 Receptor Agonists — The New Wave
- Spironolactone for Androgen Symptoms
- Combined Oral Contraceptives — Pick Carefully
- Letrozole for Ovulation Induction
- Diet and Exercise That Actually Work
- Sleep Apnea and Mental Health
- Endometrial Protection
- Fertility Pathway and Pregnancy Risks
- Key Research Papers
- Connections
- Featured Videos
Why This Connection Matters
If you have PCOS, there is a better-than-even chance that insulin resistance is the engine driving your symptoms — whether you are thin, average, or carrying extra weight. This is not a footnote in your chart. It is often the central metabolic problem, and the reason cycles are irregular, hair is growing where it shouldn’t, acne is breaking through, and conception is harder than expected.
Most PCOS guidance focuses on the ovary. That is a mistake. The ovary is usually a bystander reacting to a whole-body insulin problem upstream. Fix the insulin signal, and the ovary often quiets down on its own: androgens fall, periods return, fertility improves, weight becomes easier to manage. Treat only the ovary and you are always chasing the fire downstream of the gas line.
This page is for patients who want to understand why their endocrinologist orders certain labs, why metformin keeps coming up even though they “don’t have diabetes,” and what options exist beyond “lose weight and take the pill.”
How Common Is IR in PCOS?
Depending on how it is measured, between 50% and 70% of PCOS patients have insulin resistance. In a landmark 1989 study by Andrea Dunaif, PCOS patients were shown to have a substantial defect in insulin action independent of obesity — lean women with PCOS were insulin-resistant compared with lean controls, and women with obesity and PCOS were more insulin-resistant than their weight-matched peers without PCOS. That paper reframed PCOS as a metabolic disease, not just a reproductive one.
The overlap is so consistent that many endocrinologists treat a new PCOS diagnosis as a prompt to screen for IR by default, even in patients who look metabolically healthy. The reason is simple: you cannot reliably predict insulin resistance by looking at someone. A patient with a BMI of 22, normal fasting glucose, and a normal A1c can still have a fasting insulin of 18 µIU/mL and a HOMA-IR of 4. That patient has meaningful insulin resistance that will not show up on any standard primary-care panel.
Lean PCOS — The Subset That Gets Missed
Roughly 20-30% of PCOS patients have a BMI under 25. These women often spend years being told their symptoms can’t be PCOS because “you’re not overweight.” They also get dismissed from metabolic workup because their fasting glucose is fine and their A1c is 5.2%.
A 2013 study by Stepto and colleagues used the gold-standard euglycemic-hyperinsulinemic clamp — the most accurate way to measure insulin action — and showed that lean women with PCOS had a 27% lower glucose disposal rate than lean controls. In other words, lean PCOS really does come with real insulin resistance, it is just hidden. The standard fasting glucose test misses it because glucose stays normal for years while insulin climbs to keep it there.
If you are a lean PCOS patient, the IR workup is more important for you, not less. Your weight is hiding the problem rather than signalling it.
The Mechanism: How High Insulin Scrambles Ovaries
Understanding the mechanism makes the treatment logic obvious. Four things happen when insulin runs chronically high:
- Theca cells make more testosterone. Insulin acts directly on ovarian theca cells — the androgen-producing cells — and amplifies their response to luteinizing hormone (LH). More LH stimulation plus more insulin equals more testosterone and androstenedione pouring into circulation.
- SHBG drops. The liver makes sex hormone-binding globulin (SHBG), the carrier protein that keeps most circulating testosterone inactive. Insulin suppresses SHBG production. Less SHBG means a larger fraction of testosterone is “free” — biologically active — even if total testosterone looks only modestly elevated. This is why two women with the same total testosterone can have very different acne and hair growth.
- LH/FSH ratio tilts. The pituitary normally pulses LH and follicle-stimulating hormone (FSH) in a balanced rhythm. In PCOS, LH pulses faster and higher while FSH lags. The result is an LH-dominant environment that pushes theca cells to make still more androgen and leaves granulosa cells understimulated.
- Follicle maturation halts. With low FSH signaling and high intraovarian androgen, follicles grow partway and stall. They never mature to the dominant follicle that would ovulate. Instead, the ovary accumulates dozens of small 2-9 mm follicles visible on ultrasound — the “polycystic” appearance — and skips ovulation entirely. That is anovulation, which means no progesterone, which means no regular period.
Every arm of PCOS treatment you will read about below is targeting one of these four steps.
Rotterdam Criteria and the Four Phenotypes
PCOS is diagnosed using the Rotterdam criteria (2003), which require two of three features:
- Oligo- or anovulation (fewer than 8-9 periods per year, or cycles longer than 35 days)
- Clinical or biochemical hyperandrogenism (hirsutism, acne, androgenic alopecia, OR elevated total/free testosterone or DHEAS)
- Polycystic ovaries on ultrasound (≥12 follicles 2-9 mm per ovary, or ovarian volume >10 mL)
Because only two of three are needed, PCOS splits into four phenotypes:
- Phenotype A — all three features (the “classic” form, highest metabolic risk)
- Phenotype B — anovulation plus hyperandrogenism (no cysts on imaging)
- Phenotype C — hyperandrogenism plus polycystic ovaries but ovulatory cycles (“ovulatory PCOS”)
- Phenotype D — anovulation plus polycystic ovaries without hyperandrogenism (lowest metabolic risk)
Phenotypes A and B carry the highest insulin resistance burden. Phenotype D is the mildest metabolically. This matters because it explains why two friends with “the same” diagnosis can have very different symptoms and very different long-term risks.
The Lab Workup: What to Order
A proper PCOS evaluation is not just a pregnancy test and a testosterone level. A complete first-visit panel should include:
- Fasting insulin + fasting glucose — to calculate HOMA-IR (see the fasting insulin and HOMA-IR guide for how to interpret the number). A fasting insulin above 10 µIU/mL in a lean patient is a red flag.
- Total testosterone and free testosterone (calculated or direct)
- SHBG — often low in insulin-resistant PCOS and one of the most useful single markers
- DHEAS — adrenal androgen; if markedly elevated, think about adrenal sources or late-onset congenital adrenal hyperplasia
- 17-hydroxyprogesterone (17-OHP), fasting, early morning — screens for non-classical congenital adrenal hyperplasia, which mimics PCOS
- TSH, free T4 — rule out thyroid disease (Hashimoto’s and PCOS coexist more often than chance)
- Prolactin — rule out hyperprolactinemia, which also causes anovulation
- LH and FSH — an LH/FSH ratio above 2 is suggestive but not required
- Androstenedione — an upstream androgen often elevated when testosterone is only borderline
- AMH (anti-Müllerian hormone) — often elevated in PCOS and useful for both diagnosis and fertility planning
- HbA1c and lipid panel — baseline metabolic screen
- Pelvic ultrasound — ideally transvaginal, day 2-5 of the cycle if possible
Any single piece of this panel in isolation can mislead. Interpreted together, they usually give a clear picture.
Metformin — The Workhorse
Metformin has been used in PCOS for decades and remains the first-line pharmacologic treatment for the insulin-resistance component. The typical dose is 1,500-2,000 mg per day, usually extended-release, taken with the largest meal to reduce GI side effects. Start at 500 mg per day and titrate up over 3-4 weeks.
What metformin does in PCOS:
- Lowers hepatic glucose output, which lowers insulin demand and circulating insulin
- Modestly improves peripheral insulin sensitivity
- Raises SHBG over 3-6 months, which lowers free testosterone
- Restores ovulation in a meaningful minority of patients, often within 3-6 months
- Modestly reduces pregnancy complications (gestational diabetes in particular) when continued through pregnancy
Side effects are primarily GI (nausea, loose stools, metallic taste) and resolve for most patients within a few weeks on the extended-release form. Long-term metformin can lower B12 — check levels yearly and supplement if needed. See the metformin, berberine, and GLP-1s page for dosing detail and the berberine comparison.
Inositol — The Supplement With Real Data
Most supplements marketed for PCOS have weak evidence. Inositol is the exception. It is a sugar alcohol that participates in insulin signaling inside cells, and in PCOS the ratio of myo-inositol to D-chiro-inositol in the ovary appears disordered.
The evidence-based dose is myo-inositol 2 g twice daily plus D-chiro-inositol 50 mg twice daily — a 40:1 ratio that mirrors the physiologic blood ratio. A 2016 meta-analysis by Unfer and colleagues pooled multiple randomized trials and found consistent improvements in ovulation rate, menstrual regularity, insulin sensitivity, and androgen levels, with a side-effect profile indistinguishable from placebo. Effects take roughly 3 months to appear.
Inositol is available over the counter, does not require a prescription, and costs $20-40 per month. It is a reasonable first trial for lean PCOS patients who are not yet ready for prescription metformin, and it is commonly stacked with metformin in more severe cases. It is also safe in pregnancy — in fact some obstetricians use it for gestational diabetes prevention in high-risk patients.
GLP-1 Receptor Agonists — The New Wave
GLP-1 receptor agonists (semaglutide, tirzepatide, liraglutide) are approved for type 2 diabetes and obesity, not PCOS. But their use in PCOS is growing rapidly off-label because the mechanism is almost purpose-built for the problem: they slow gastric emptying, reduce appetite, lower insulin, and produce meaningful weight loss.
In PCOS specifically, early trials and real-world practice report:
- 10-15% body weight loss at 12 months on semaglutide, more on tirzepatide
- Drops in fasting insulin and HOMA-IR
- Increases in SHBG and drops in free testosterone
- Restoration of ovulation in a meaningful percentage of previously anovulatory patients
Two important cautions. First, these drugs are not safe in pregnancy and must be stopped at least 2 months before conception is attempted — and rebound ovulation can catch patients off guard, so reliable contraception during treatment is essential unless actively trying to conceive on the drug’s taper plan. Second, they are expensive and insurance coverage for PCOS alone (without documented type 2 diabetes or qualifying BMI) is inconsistent.
Spironolactone for Androgen Symptoms
Metformin, inositol, and GLP-1s treat insulin. They do not directly block androgen at the skin or hair follicle, which means acne and hirsutism often persist even after cycles regularize. That is where spironolactone comes in.
Spironolactone at 50-200 mg per day (usually started at 50 mg and titrated) blocks androgen receptors at the skin and hair follicle. It takes 3-6 months for acne to clear and 6-12 months for hair growth to slow. Side effects include increased urination in the first weeks, breast tenderness, and occasional menstrual irregularity.
The critical caveat: spironolactone is not safe to continue while trying to conceive, because it can feminize a male fetus. Patients who want children must stop spironolactone and use another method (laser hair removal, electrolysis, topical eflornithine) or accept some androgen symptoms during the TTC window.
Combined Oral Contraceptives — Pick Carefully
Combined oral contraceptives (COCs) are a reasonable option for cycle regularity, endometrial protection, and androgen symptom control in PCOS patients who are not trying to conceive. But the pill matters.
Some progestins in older COCs — particularly levonorgestrel — have mild androgenic activity and can worsen insulin resistance and lipid profiles. For PCOS, clinicians increasingly prefer formulations with:
- Drospirenone (e.g., Yaz, Yasmin) — mild anti-androgenic activity, favored in PCOS, though carries a slightly higher venous thromboembolism risk than older progestins
- Norgestimate (e.g., Ortho Tri-Cyclen) — low androgenicity, good metabolic profile
- Cyproterone acetate (not available in the US but common in Europe) — strong anti-androgen
If you are on a levonorgestrel-containing pill and your acne, hair, or metabolic labs are worsening, ask about switching. The pill is not one product; the progestin is a meaningful variable.
Letrozole for Ovulation Induction
When PCOS patients are ready to conceive and ovulation has not returned with lifestyle and metformin/inositol, the first-line ovulation induction drug is now letrozole, not clomiphene citrate.
The shift was driven by the PPCOS II trial (Legro et al., 2014), a large US multicenter randomized study that compared letrozole to clomiphene in PCOS patients pursuing pregnancy. Letrozole produced more ovulations, more live births, and fewer multiple pregnancies than clomiphene. It is now the preferred first-line agent in every major guideline.
Letrozole is prescribed at 2.5-7.5 mg daily for 5 days beginning on cycle day 3, 4, or 5 of an induced or spontaneous cycle. Most patients respond at 2.5 or 5 mg. If three letrozole cycles do not produce pregnancy, the typical next steps are letrozole plus injectable gonadotropins, letrozole plus intrauterine insemination (IUI), or in vitro fertilization (IVF). See the infertility page for the full treatment ladder.
Diet and Exercise That Actually Work
There is no single “PCOS diet.” Multiple patterns work if they consistently lower insulin and are sustainable. The evidence-supported options:
- Low-glycemic-index — choose carbs that raise blood glucose slowly: lentils, beans, steel-cut oats, intact whole grains, most vegetables, lower-sugar fruits. Skip white bread, sugary drinks, and ultra-processed snacks.
- Mediterranean — olive oil, fish, nuts, legumes, vegetables, moderate whole grains, limited red meat. Repeatedly associated with improvements in insulin sensitivity and androgen levels in PCOS cohorts.
- DASH — originally developed for blood pressure, shares the Mediterranean framework and performs well in PCOS trials.
- Moderate-carb with adequate protein — typically 30-40% of calories from carbohydrate, 25-30% from protein (1.2-1.6 g/kg body weight), with the balance from fat. Protein preserves lean mass during weight loss and improves satiety.
Exercise is non-negotiable. The combination that moves the needle most reliably:
- Resistance training 2-3 times per week. Muscle is the body’s largest glucose sink; more muscle means better insulin disposal. See the exercise and muscle sensitivity page.
- Daily walking, particularly after meals. A 10-15 minute walk after dinner measurably lowers the post-meal glucose curve.
- Moderate cardio on top, 2-3 sessions per week, for cardiovascular fitness.
Sleep Apnea and Mental Health
PCOS carries a 5-30 times higher risk of obstructive sleep apnea (OSA) than the general female population, independent of weight. Untreated OSA drives insulin resistance, hypertension, and fatigue. If you have PCOS and any of the classic symptoms — loud snoring, observed apneas, morning headaches, unrefreshing sleep, daytime sleepiness — ask for a home sleep study. CPAP treatment of diagnosed OSA improves insulin sensitivity within weeks.
Mental health deserves the same rigor. PCOS patients have roughly 3 times the rate of anxiety and depression of age-matched women without PCOS. This is not a character flaw or a reaction to appearance changes — it is a direct biological consequence of the hormonal milieu, and it responds to the same treatments (therapy, SSRIs/SNRIs, exercise) that work in the general population. Screen yourself honestly and ask for help if the answer is yes.
Endometrial Protection
Chronic anovulation means the uterine lining is exposed to estrogen without the regular shedding that progesterone triggers after ovulation. Over years, that unopposed estrogen increases the risk of endometrial hyperplasia and endometrial cancer. The risk is real and meaningful — several-fold higher than baseline — and it is preventable.
If you are not trying to conceive, you need some form of progestin coverage. The standard options:
- A combined oral contraceptive (daily progestin exposure)
- A levonorgestrel IUD (Mirena, Kyleena, Liletta) — placed once, good for 5-8 years, delivers progestin locally to the endometrium with minimal systemic effect
- Cyclic oral progesterone (e.g., medroxyprogesterone 10 mg for 10 days every 1-3 months) to induce a withdrawal bleed — sometimes called a “progestin challenge”
If you are not on any of these, you should be having a withdrawal bleed at least every 3 months. Longer gaps without any of the above is a conversation to have with your gynecologist sooner rather than later.
Fertility Pathway and Pregnancy Risks
The typical PCOS fertility ladder looks like this:
- Lifestyle and IR treatment first — 3-6 months of diet, exercise, metformin, and/or inositol. A substantial minority resume ovulation on this alone and conceive without further intervention.
- Letrozole ovulation induction — typically 3 cycles
- Letrozole plus IUI — adds intrauterine insemination if ovulation is happening but not resulting in pregnancy
- Gonadotropins — injectable FSH, usually with IUI or IVF
- IVF — high success rates in PCOS because ovarian reserve is generally excellent, though ovarian hyperstimulation syndrome (OHSS) risk is elevated and requires careful protocol choice
Once pregnant, PCOS patients have elevated rates of gestational diabetes, preeclampsia, and preterm birth. None of these is inevitable, but they justify earlier and more frequent screening than a standard pregnancy: an early glucose tolerance test (often at 12-16 weeks instead of the standard 24-28), blood pressure vigilance, and in many cases continuation of metformin through pregnancy (which appears safe based on current evidence).
Key Research Papers
Foundational publications on the insulin resistance / PCOS axis, the Rotterdam diagnostic consensus, and the major drug trials that shape modern management. Each citation links to the full text via DOI.
- Dunaif A, Segal KR, Futterweit W, Dobrjansky A. Profound Peripheral Insulin Resistance, Independent of Obesity, in Polycystic Ovary Syndrome. Diabetes. 1989;38(9):1165–1174.
- Dunaif A. Insulin Resistance and the Polycystic Ovary Syndrome: Mechanism and Implications for Pathogenesis. Endocrine Reviews. 1997;18(6):774–800.
- Stepto NK, Cassar S, Joham AE, et al. Women with Polycystic Ovary Syndrome Have Intrinsic Insulin Resistance on Euglycaemic-Hyperinsulaemic Clamp. Human Reproduction. 2013;28(3):777–784.
- Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group. Revised 2003 Consensus on Diagnostic Criteria and Long-Term Health Risks Related to Polycystic Ovary Syndrome. Human Reproduction. 2004;19(1):41–47.
- Teede HJ, Tay CT, Laven JJE, et al. Recommendations from the 2023 International Evidence-Based Guideline for the Assessment and Management of Polycystic Ovary Syndrome. Journal of Clinical Endocrinology & Metabolism. 2023;108(10):2447–2469.
- Unfer V, Facchinetti F, Orrù B, Giordani B, Nestler J. Myo-inositol Effects in Women with PCOS: A Meta-Analysis of Randomized Controlled Trials. Endocrine Connections. 2017;6(8):647–658.
- Legro RS, Brzyski RG, Diamond MP, et al. Letrozole versus Clomiphene for Infertility in the Polycystic Ovary Syndrome (PPCOS II). New England Journal of Medicine. 2014;371(2):119–129.
- Reaven GM. Banting Lecture 1988. Role of Insulin Resistance in Human Disease. Diabetes. 1988;37(12):1595–1607.
- Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis Model Assessment: Insulin Resistance and Beta-Cell Function from Fasting Plasma Glucose and Insulin Concentrations in Man (HOMA). Diabetologia. 1985;28(7):412–419.
- Petersen MC, Shulman GI. Mechanisms of Insulin Action and Insulin Resistance. Physiological Reviews. 2018;98(4):2133–2223.
- Knowler WC, Barrett-Connor E, Fowler SE, et al. Reduction in the Incidence of Type 2 Diabetes with Lifestyle Intervention or Metformin (Diabetes Prevention Program). New England Journal of Medicine. 2002;346(6):393–403.
- Saltiel AR, Olefsky JM. Inflammatory Mechanisms Linking Obesity and Metabolic Disease. Journal of Clinical Investigation. 2017;127(1):1–4.
Live PubMed Searches
Live PubMed queries that update as new papers are indexed.
- PubMed: insulin resistance and PCOS
- PubMed: lean PCOS and IR
- PubMed: metformin and PCOS
- PubMed: myo-inositol in PCOS
- PubMed: GLP-1 agonists and PCOS
- PubMed: letrozole and ovulation induction
- PubMed: spironolactone in PCOS
- PubMed: PCOS and sleep apnea
- PubMed: PCOS and endometrial cancer
- PubMed: PCOS and gestational diabetes
Connections
- Fasting Insulin and HOMA-IR Testing
- Infertility
- Exercise and Muscle Insulin Sensitivity
- Metformin, Berberine and GLP-1s
- Hashimoto's Thyroiditis
- Insulin Resistance
- Polycystic Ovary Syndrome
- Reversal Protocol: Diet and Fasting
- Endometriosis
- Obesity
- Inositol
- GLP-1 Receptor Agonists
- Acne
- Testosterone
- Gestational Diabetes
- Fasting Insulin
- Diabetes
- NAFLD MASLD Connection