Reverse T3 and Low-T3 Syndrome in Hashimoto's
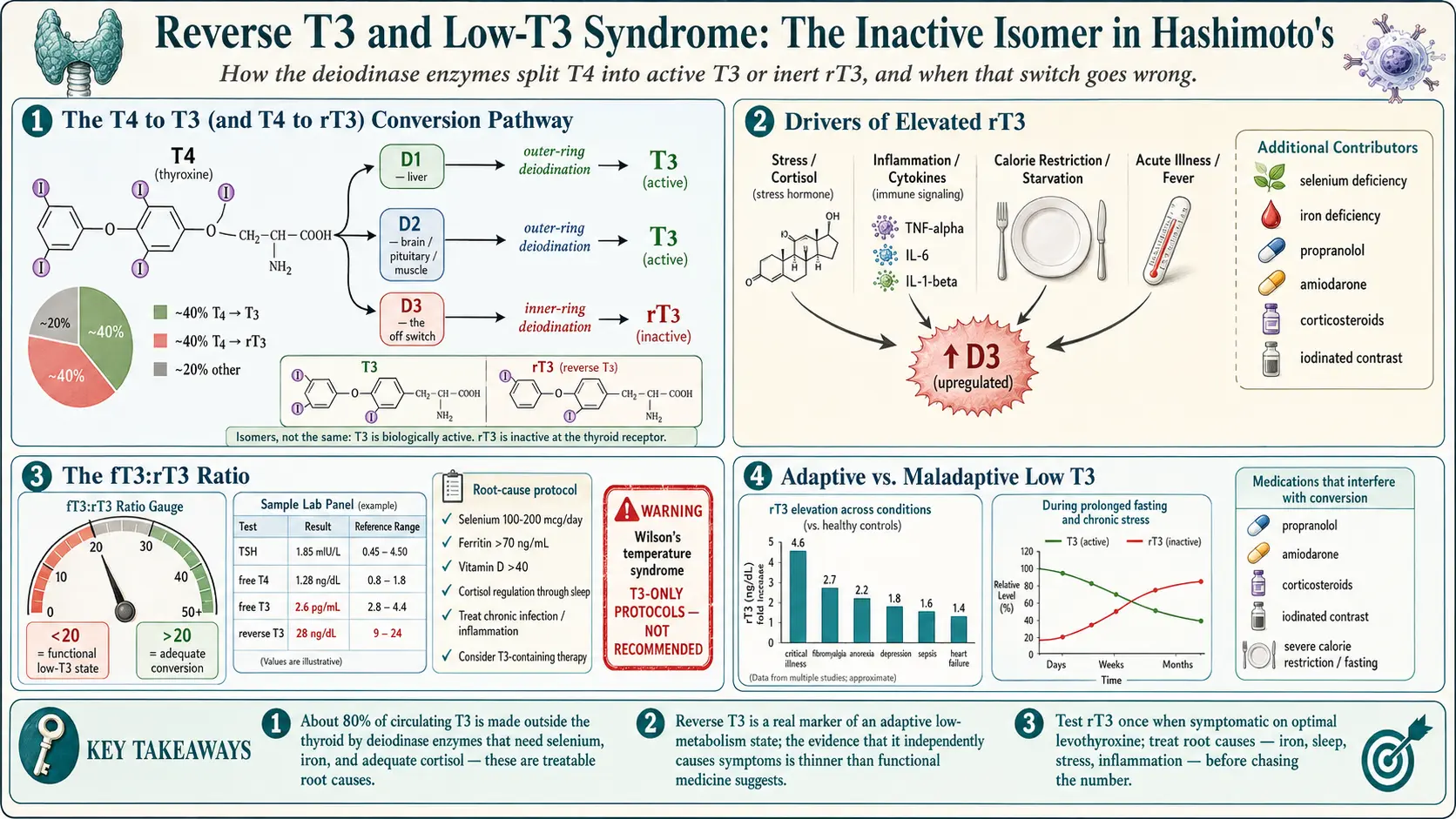
Table of Contents
- The T4 → T3 (and T4 → rT3) Conversion Pathway
- Reverse T3 — The Inactive Isomer
- Why rT3 Matters — If It Matters
- The Mainstream View
- The Functional / Integrative View
- What the Evidence Actually Shows
- When to Test rT3
- Causes of Elevated rT3
- Low T3 Syndrome (Nonthyroidal Illness Syndrome)
- Functional Protocols for Lowering rT3
- Cortisol and Thyroid
- Iron and Ferritin as a Driver
- Leptin, Dieting, and Metabolic Adaptation
- Chronic Inflammation and Conversion
- A Practical Approach for a Patient with High rT3 Symptoms
- When rT3 Isn't the Problem
- The Wilson's Syndrome Warning
- What's Reasonable
- Key Research Papers
- Connections
- Featured Videos
The T4 → T3 (and T4 → rT3) Conversion Pathway
Your thyroid gland mostly makes T4 (thyroxine). T4 is essentially a prohormone — it has four iodine atoms attached to a tyrosine backbone, and on its own it does very little inside a cell. What your tissues actually use is T3 (triiodothyronine), which is T4 with one iodine surgically removed from the outer ring. T3 binds the thyroid hormone receptor inside the cell nucleus and switches on the genes that set your metabolic rate, body temperature, heart rate, cognition, and roughly every other energy-dependent process in the body. About 80% of circulating T3 is made outside the thyroid, in peripheral tissues, by cutting that one iodine off T4.
The enzymes that do the cutting are called deiodinases, and there are three of them:
- D1 (deiodinase type 1) — mostly in liver, kidney, and thyroid itself. Produces T3 for circulation. Relatively non-selective; it can also clear rT3.
- D2 (deiodinase type 2) — in brain, pituitary, brown fat, muscle, placenta. Produces T3 locally for the tissue that needs it. This is why your pituitary can “see” thyroid status even when circulating T3 is low: D2 quietly makes its own T3 from T4 inside pituitary cells.
- D3 (deiodinase type 3) — the “off switch.” Instead of removing the outer-ring iodine (producing active T3), D3 removes an inner-ring iodine, producing reverse T3 (rT3), which is biologically inactive. D3 also degrades T3 into T2.
Deiodinase activity is tissue-specific. Your liver, brain, and muscle can each be doing different things at the same moment — one tissue activating T4 into T3, another shunting it into rT3. This matters because a single blood draw measures the circulating pool, not what is happening inside your muscle or your hypothalamus. When people say they feel hypothyroid despite “normal labs,” part of the answer may be tissue-level conversion that the blood test can't see.
Free T3 — not total T3, not T4, and not TSH — is the hormone actually doing the work. Only about 0.3% of circulating T3 is free (unbound from thyroxine-binding globulin); the rest is stuck to carrier proteins and metabolically invisible. This is why free T3 and free T4 are the measurements functional physicians push for rather than the total values.
Reverse T3 — The Inactive Isomer
When D3 pulls an iodine off the inner ring of T4 instead of the outer one, the result is a mirror-image molecule: same molecular formula, same weight, inert at the T3 receptor. This is reverse T3. In a healthy, well-fed, unstressed person at rest, roughly 40% of T4 becomes T3 and 40% becomes rT3; the remaining 20% is further metabolized or excreted. Those fractions are not fixed — they shift with physiologic state.
D3, and therefore rT3, is favored by:
- Acute or chronic illness
- Fasting, aggressive calorie restriction, or starvation
- High cortisol (physical or emotional stress)
- Systemic inflammation
- Some medications
From an evolutionary standpoint the logic is obvious: when you are sick, injured, or starving, your body slows metabolism by shunting T4 away from active T3 and into inert rT3. It's a thrift mode. The question that drives this whole article is whether that thrift mode, switched on chronically, becomes a disease in its own right.
Why rT3 Matters — If It Matters
The functional-medicine argument is that rT3 doesn't just sit around doing nothing. According to this view, rT3 competes with T3 at the nuclear receptor and at cellular transporters — docking without activating, like a key that fits the lock but won't turn. High rT3 would then produce cellular hypothyroidism even when serum free T3 looks acceptable. Patients would feel cold, tired, foggy, and slow while their lab numbers reassure the endocrinologist that everything is fine.
This is the “rT3 dominance” hypothesis. It is attractive because it explains a real clinical frustration: people on levothyroxine with “perfect” TSH who still feel hypothyroid. Whether it is also biologically correct is a separate question, and one the evidence does not clearly answer.
The Mainstream View
Mainstream endocrinology — the American Thyroid Association (ATA), the European Thyroid Association, and most academic endocrinologists — considers rT3 a marker, not a mechanism. In their framework, an elevated rT3 reflects that something else is going on (illness, stress, starvation, a medication effect) and the right response is to treat that upstream cause. Giving thyroid hormone to drive rT3 down is, in the mainstream view, treating the thermometer rather than the fever.
The ATA does not recommend routine rT3 testing in the workup of hypothyroidism or in the monitoring of treatment. The clinical guidelines explicitly state that rT3 is not useful for managing hypothyroid patients. Insurance often refuses to cover it outside of research protocols or ICU settings.
The mainstream position is not paranoia. Reverse T3 levels bounce around with minor illnesses, recent meals, sleep, and blood-draw timing. Interpreting a single number as a dominant pathology is, at best, aspirational. Most endocrinologists who ignore rT3 are acting on the published evidence, not dismissing patients.
The Functional / Integrative View
Functional and integrative practitioners often order rT3 alongside TSH, free T4, free T3, TPO antibodies, and thyroglobulin antibodies. They typically calculate the free T3 to reverse T3 ratio (fT3:rT3) as a proxy for conversion efficiency. A commonly cited cutoff:
- fT3:rT3 ratio > 20 — considered adequate conversion (when both values are in ng/dL or both in pg/mL after unit conversion).
- Ratio < 20 — interpreted as poor peripheral conversion or an effective low-T3 state.
Protocols then target lowering rT3 by fixing conversion — addressing stress, cortisol, iron, selenium, inflammation, and in some cases switching from levothyroxine to a T3-containing therapy. It is worth stating clearly: the 20 cutoff is a convention, not a validated threshold. The studies linking the ratio to clinical outcomes are small and heterogeneous.
What the Evidence Actually Shows
The honest summary: rT3 elevation correlates with a long list of conditions — critical illness, major depression, chronic fatigue syndrome, fibromyalgia, post-surgical recovery, anorexia nervosa, sepsis, heart failure, cirrhosis. The correlation is robust. What remains uncertain is causality: does high rT3 cause the symptoms, or does the underlying illness cause both the symptoms and the high rT3?
Randomized trials of T3 supplementation in nonthyroidal illness have been largely negative for hard outcomes. When investigators give T3 to ICU patients with low T3 and high rT3, mortality and length of stay generally do not improve, and in some studies adverse events (arrhythmias) increase. This is a strong signal that, at least in acute illness, the low-T3 state is adaptive and overriding it is not helpful.
Whether the same logic applies to a chronically-stressed outpatient with Hashimoto's, a normal TSH, and persistent fatigue is unknown. No large randomized trial has answered that question, and it is unlikely to be funded. The clinical reality sits in that data gap.
When to Test rT3
A reasonable, middle-ground indication: consider testing rT3 once if you are symptomatic despite apparently optimal TSH and free T4, particularly if fatigue, weight gain, cold intolerance, and cognitive slowness have persisted for months on levothyroxine. The goal is not to chase a number but to decide whether a conversion problem could be part of the picture — and to focus attention on the upstream drivers.
Retesting rT3 repeatedly is rarely useful. The value moves with the last night's sleep, a recent cold, or a stressful week. Treat it as an occasional snapshot, not a running dashboard.
Causes of Elevated rT3
Before ascribing symptoms to “rT3 dominance,” walk through the real drivers. Most of them are more productive to treat than the number itself.
- Acute illness — respiratory infection, surgery, trauma, sepsis. Sick euthyroid / nonthyroidal illness syndrome is the textbook cause.
- Chronic stress and HPA dysregulation — sustained high cortisol shifts conversion toward rT3 and suppresses TSH.
- Calorie restriction and extreme diets — prolonged low-carbohydrate or very-low-calorie dieting drops leptin, which drops T3.
- Selenium deficiency — D1 and D2 are selenoenzymes; without selenium, conversion is impaired.
- Low ferritin / iron deficiency — iron is required for thyroid peroxidase activity and indirectly supports peripheral conversion.
- Systemic inflammation — cytokines (TNF-α, IL-6, IL-1β) upregulate D3.
- Chronic infections — Lyme, reactivated viruses, chronic sinusitis, and dental abscesses keep the inflammatory dial turned up.
- Medications — beta-blockers (propranolol especially), amiodarone, corticosteroids, iodinated contrast dye, and some antiepileptics all interfere with conversion.
- Pregnancy — rT3 rises normally in pregnancy due to placental D3. This is not pathologic.
- Heavy metals — mercury and lead interfere with selenoenzyme function; the clinical significance in typical outpatients is debated.
- Aging and obesity — both are associated with modest rT3 elevation of uncertain importance.
Low T3 Syndrome (Nonthyroidal Illness Syndrome)
Nonthyroidal illness syndrome (NTIS), also called euthyroid sick syndrome or low T3 syndrome, is the well-characterized pattern seen in critically ill patients. In its classic form: free T3 drops, rT3 rises, TSH stays normal or slightly low, free T4 is usually normal until the illness is severe. In prolonged ICU illness all of these values eventually fall.
NTIS is now understood as an adaptive response — the body lowering metabolism to conserve energy during catastrophic illness. Multiple randomized trials in ICU populations have asked whether giving thyroid hormone (usually T3) improves outcomes. The results are consistently underwhelming, and in some subsets harmful. Current critical-care and endocrine society guidelines do not recommend treating NTIS with thyroid hormone in the ICU. Treat the underlying illness; the thyroid axis corrects itself as the patient recovers.
Outpatient “chronic NTIS” — the term some functional clinicians use for persistent fatigue with low fT3 and high rT3 in an ambulatory Hashimoto's patient — is a looser concept, not a formal diagnosis. Whether the same ICU data apply is genuinely unclear.
Functional Protocols for Lowering rT3
With the caveat that the evidence base is thin, the interventions below are at least biologically plausible and mostly low-risk:
- Address root causes first. Stress, sleep, infections, dieting patterns, and inflammation drive rT3 far more than anything on a supplement shelf. Start here.
- Stress and cortisol. Sleep 7–9 hours. Limit caffeine after noon. Reduce late-evening screen time. Structured meditation, yoga, or therapy. Adaptogens (ashwagandha, rhodiola) have modest evidence; they are not a substitute for the foundational work.
- Selenium. 100–200 mcg/day (upper safe limit around 400 mcg/day; higher doses are toxic). A Brazil nut or two a day is an option, though their selenium content is highly variable.
- Ferritin correction. Aim for ferritin > 70 ng/mL for thyroid purposes; iron supplementation guided by iron studies, not guesses.
- Gut healing. Treat small intestinal bacterial overgrowth, food reactivity, and celiac disease. See the AIP and gluten elimination article.
- Infection workup. If suspicion warrants, evaluate for chronic viral reactivation, Lyme, or dental sources.
- T3-containing therapy. Switching from levothyroxine to natural desiccated thyroid or adding liothyronine (T3) bypasses the conversion step entirely. See Levothyroxine vs NDT vs T3. This is a reasonable, evidence-tiered intervention with a willing clinician.
- T3-only “Wilson's temperature syndrome” protocols. Highly controversial, rejected by mainstream endocrinology, and associated with cardiac risk. Not recommended. See the warning section below.
Cortisol and Thyroid
The HPA (hypothalamic-pituitary-adrenal) axis and the HPT (thyroid) axis are tightly coupled. Chronically elevated cortisol suppresses TSH at the pituitary and favors D3 over D2 in peripheral tissues — producing a functional low-T3 picture even in people with an intact thyroid. Chronically low cortisol (true adrenal insufficiency) must be ruled out separately because it causes its own syndrome and changes how thyroid hormone is tolerated.
Some integrative practitioners order a four-point salivary cortisol test to map the daily curve. This is clinically reasonable, though interpretation is not fully standardized and insurance rarely covers it. “Adrenal fatigue” as a diagnosis is not recognized by endocrinology, but the underlying observation — that chronic stress produces measurable changes in cortisol rhythm — is real. Treat it with sleep, stress load, and lifestyle before turning to supplements.
Iron and Ferritin as a Driver
Iron deficiency is one of the most common, most fixable, and most-overlooked causes of persistent hypothyroid symptoms in Hashimoto's. Ferritin below 70 ng/mL can impair thyroid peroxidase function, reduce conversion, and produce fatigue, hair loss, cold intolerance, and breathlessness that look identical to undertreated hypothyroidism. Menstruating women, vegetarians, and long-time proton-pump-inhibitor users are at highest risk.
Check ferritin, iron saturation, and TIBC before assuming the problem is rT3. If ferritin is low, correcting it often resolves “rT3 symptoms” without touching the thyroid dose.
Leptin, Dieting, and Metabolic Adaptation
Leptin is the satiety hormone secreted by fat tissue. It also signals the hypothalamus to keep the thyroid axis running normally. When you restrict calories aggressively for weeks or months, leptin drops sharply — far more than the fat loss alone would predict — and the hypothalamus responds by dialing down T3 production. This is the mechanism behind “metabolic adaptation” or, colloquially, “metabolic damage” from chronic dieting.
For a Hashimoto's patient who has spent years on very-low-calorie or very-low-carbohydrate diets, part of a persistent low-T3 picture may simply be a starved-leptin state. The treatment is not more thyroid hormone; it is adequate calories, adequate carbohydrate, and patience. Refeeding often restores conversion within weeks to months.
Chronic Inflammation and Conversion
Inflammatory cytokines — TNF-α, IL-6, IL-1β — directly upregulate D3 and downregulate D1. This is how infections, autoimmune flares, obesity-associated inflammation, and untreated gut disease can all produce the high-rT3 pattern. Treating the inflammation generally fixes the conversion problem without the need to intervene at the thyroid level. Relevant levers include an anti-inflammatory diet, treating autoimmune activity (the Hashimoto's itself), addressing gut pathology, managing sleep apnea, and losing visceral fat if applicable.
A Practical Approach for a Patient with High rT3 Symptoms
A sensible sequence, done with your clinician:
- Verify the basics. Ferritin > 70, vitamin D > 40 ng/mL, B12 in the upper half of the reference range, selenium intake adequate, zinc adequate.
- Rule out mimics. Obstructive sleep apnea, celiac disease, perimenopause, iron-deficiency anemia, major depression, chronic fatigue syndrome, POTS. See the Hashi/POTS/MCAS overlap article.
- Address cortisol patterns. Prioritize sleep quality and duration, reduce stress load where possible, consider a four-point salivary cortisol if the picture suggests dysregulation.
- Re-evaluate thyroid dosing. TSH in the upper half of the reference range with symptoms is commonly undertreatment. Free T4 mid-range with low free T3 suggests conversion is worth considering.
- Trial a T3-containing therapy with a willing clinician — either NDT or adding a small dose of liothyronine to levothyroxine.
- Re-check labs at 8–12 weeks. Look at symptoms first, ratios second, single numbers last.
When rT3 Isn't the Problem
Most symptomatic Hashimoto's patients with a “normal TSH” are not rT3 dominant. They are one of the following:
- Undertreated. A TSH between 2.0 and 4.5 is inside the reference range but often symptomatic. Many patients feel best with TSH between 0.5 and 2.0. See TSH/T4/T3 optimal ranges.
- Iron deficient. Check ferritin.
- Perimenopausal. Declining estrogen and progesterone mimic hypothyroidism symptom-for-symptom.
- Under-slept. Chronic sleep restriction produces cognitive slowness, weight gain, and cold intolerance independent of thyroid status.
- Carrying a separate diagnosis — chronic fatigue syndrome, POTS, depression, sleep apnea, celiac disease.
Chasing rT3 before ruling these out is a common way to spend years and thousands of dollars on supplements without feeling better.
The Wilson's Syndrome Warning
In the 1990s, E. Denis Wilson proposed “Wilson's temperature syndrome” — the claim that low basal body temperature, independent of lab values, reflects an rT3 problem and should be treated with T3-only protocols. Mainstream endocrinology, including the American Thyroid Association, formally rejected this diagnosis. The Florida Board of Medicine disciplined Dr. Wilson following the death of a patient on his protocol, and his medical license was suspended. He later surrendered it.
A subset of functional clinicians still use basal-temperature-guided T3-only protocols. The concerns are real: T3-only dosing causes marked free-T3 peaks and troughs, predisposing to atrial fibrillation, tachycardia, anxiety, insomnia, and bone loss, particularly in older patients or those with cardiac disease. If a clinician proposes a T3-only “temperature protocol” without thyroid labs, seek a second opinion. Legitimate T3 use is dose-titrated against free T3, TSH, symptoms, and heart rate — not a thermometer.
What's Reasonable
Putting it together:
- Test rT3 once, at baseline, if you remain symptomatic on apparently optimal levothyroxine.
- Calculate the free T3 to reverse T3 ratio for a rough sense of conversion, but don't treat the number as gospel.
- Treat root causes — iron, selenium, sleep, stress, inflammation, infection — before treating the ratio.
- If conversion really does look impaired and symptoms persist, discuss a T3-containing therapy with a clinician who titrates against labs, not temperature.
- Don't retest rT3 every few months. Retest free T3, free T4, and TSH.
- Remember that mainstream endocrinology is not ignoring rT3 out of bias — the evidence base really is thin, and the adaptive-response data from critical illness argues for humility.
Reverse T3 is neither the villain some functional sites describe nor the irrelevance some endocrinologists claim. It is a signpost. Read the signpost, look where it points, and treat what you find there.
Key Research Papers
Foundational and recent peer-reviewed publications on reverse T3, the deiodinase enzyme system, and non-thyroidal illness syndrome. Each citation links to the full text via DOI.
- Bianco AC, Salvatore D, Gereben B, Berry MJ, Larsen PR. Biochemistry, Cellular and Molecular Biology, and Physiological Roles of the Iodothyronine Selenodeiodinases. Endocrine Reviews. 2002;23(1):38–89.
- Peeters RP, Wouters PJ, Kaptein E, van Toor H, Visser TJ, Van den Berghe G. Reduced Activation and Increased Inactivation of Thyroid Hormone in Tissues of Critically Ill Patients. Journal of Clinical Endocrinology & Metabolism. 2003;88(7):3202–3211.
- Van den Berghe G. Non-Thyroidal Illness in the ICU: A Syndrome with Different Faces. Thyroid. 2014;24(10):1456–1465.
- De Groot LJ. Non-Thyroidal Illness Syndrome Is a Manifestation of Hypothalamic-Pituitary Dysfunction, and in View of Current Evidence, Should Be Treated with Appropriate Replacement Therapies. Critical Care Clinics. 2006;22(1):57–86.
- Boelen A, Kwakkel J, Fliers E. Beyond Low Plasma T3: Local Thyroid Hormone Metabolism During Inflammation and Infection. Endocrine Reviews. 2011;32(5):670–693.
- Hoermann R, Midgley JEM, Larisch R, Dietrich JW. Recent Advances in Thyroid Hormone Regulation: Toward a New Paradigm for Optimal Diagnosis and Treatment. Frontiers in Endocrinology. 2017;8:364.
- Bunevicius R, Kazanavicius G, Zalinkevicius R, Prange AJ Jr. Effects of Thyroxine as Compared with Thyroxine plus Triiodothyronine in Patients with Hypothyroidism. New England Journal of Medicine. 1999;340(6):424–429.
- Jonklaas J, Bianco AC, Bauer AJ, et al. Guidelines for the Treatment of Hypothyroidism (American Thyroid Association). Thyroid. 2014;24(12):1670–1751.
- Stockigt JR. Free Thyroid Hormone Measurement: A Critical Appraisal. Endocrinology and Metabolism Clinics of North America. 2001;30(2):265–289.
- Burmeister LA. Reverse T3 Does Not Reliably Differentiate Hypothyroid Sick Syndrome from Euthyroid Sick Syndrome. Thyroid. 1995;5(6):435–441.
- Dentice M, Marsili A, Zavacki A, Larsen PR, Salvatore D. The Deiodinases and the Control of Intracellular Thyroid Hormone Signaling During Cellular Differentiation. Biochimica et Biophysica Acta. 2013;1830(7):3937–3945.
- Gärtner R, Gasnier BCH, Dietrich JW, Krebs B, Angstwurm MWA. Selenium Supplementation in Patients with Autoimmune Thyroiditis Decreases Thyroid Peroxidase Antibodies Concentrations. Journal of Clinical Endocrinology & Metabolism. 2002;87(4):1687–1691.
Live PubMed Searches
Live PubMed queries that update as new papers are indexed.
- PubMed: reverse T3 clinical significance
- PubMed: nonthyroidal illness syndrome
- PubMed: free T3 / reverse T3 ratio
- PubMed: deiodinases D1/D2/D3
- PubMed: euthyroid sick syndrome
- PubMed: cortisol + T4-to-T3 conversion
- PubMed: ferritin/iron + thyroid conversion
- PubMed: selenium + deiodinases
Connections
- Levothyroxine vs NDT vs T3
- TSH, T4 & T3 Optimal Ranges
- AIP and Gluten Elimination
- Chronic Fatigue Syndrome
- Hashimoto, POTS & MCAS Overlap
- TPO and TgAb Antibodies
- Selenium, Zinc, Iron & Iodine
- Pregnancy and Hashimoto's
- Hashimoto's Thyroiditis
- Iron
- Selenium
- Iodine
- Fatigue
- Pantothenic Acid and Adrenal Health
- Celiac Disease
- POTS
- Tyrosine
- Heavy Metals