Gut-Thyroid Axis & Intestinal Permeability
If you have Hashimoto's and a lifetime of bloating, reflux, constipation, or a diagnosis of IBS you never really accepted, you already suspect what the research is finally confirming: your gut and your thyroid are running on the same wire. Fix one and the other improves. Ignore one and the other stalls, no matter how perfectly your TSH is dialed in.
The gut-thyroid axis is not alternative-medicine hand-waving. It is conventional physiology: the intestinal lining controls what reaches your immune system, the microbiome converts a meaningful fraction of T4 to T3, stomach acid determines whether your levothyroxine is absorbed, and a breached gut barrier is now a documented feature of most autoimmune disease. The practical question is not whether your gut matters. It is which levers actually move the needle.
This article walks through the mechanisms in plain language, then gives you a real protocol — what to eat, what to supplement, what to test, and how to time your thyroid medication so it actually gets into your bloodstream.
Table of Contents
- How Gut Bacteria Convert and Absorb Thyroid Hormone
- Intestinal Permeability, LPS, and Zonulin
- Th17 Polarization — Why Dysbiosis Drives Autoimmunity
- Why Celiac and Hashimoto's Travel Together
- SIBO as a Thyroid Trigger and Medication Blocker
- H. pylori, Low Stomach Acid, and Thyroid Antibodies
- The Yeast/Candida Debate
- A Practical Gut-Healing Protocol: Remove, Repair, Repopulate
- Levothyroxine Timing, PPIs, and What Actually Blocks Absorption
- Stool Testing — What Is Worth Paying For
- Key Research Papers
- PubMed Topic Searches
- Connections
- Featured Videos
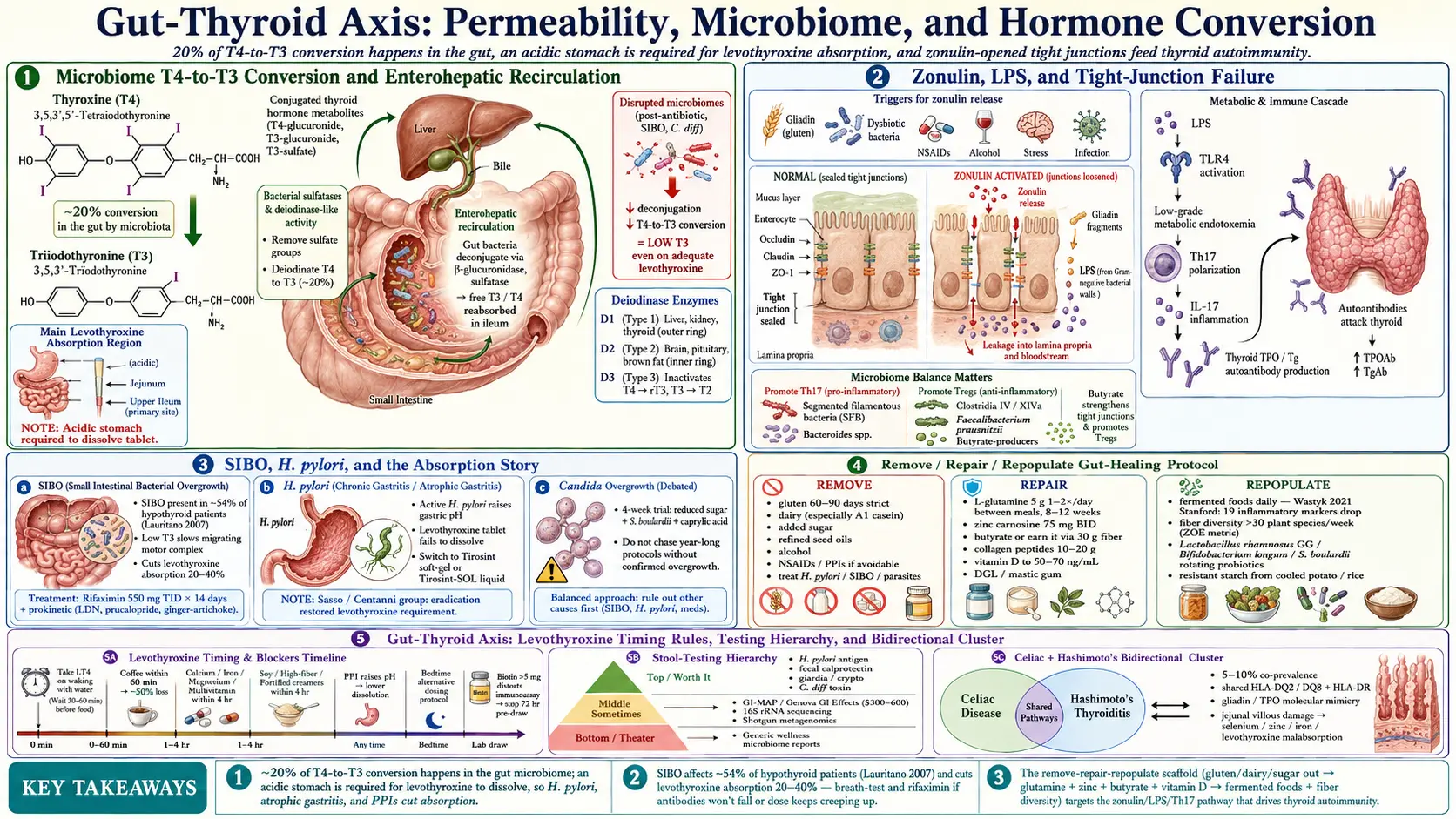
How Gut Bacteria Convert and Absorb Thyroid Hormone
Your thyroid mostly produces T4, the storage form of thyroid hormone. The active form your cells actually use is T3, and the bulk of that conversion happens in the liver and peripheral tissues via deiodinase enzymes. What is less widely known is that your gut microbiome converts roughly 20% of T4 to T3 through bacterial sulfatases and deiodinase-like activity. Patients with severely disrupted microbiomes — after long antibiotic courses, in SIBO, or after C. diff — often run low-T3 labs even on adequate levothyroxine.
The gut also houses a reservoir of conjugated thyroid hormone in bile. Healthy gut bacteria deconjugate these metabolites so they can be reabsorbed, a loop called enterohepatic recirculation. Constipation, dysbiosis, or bile-acid problems leak that reservoir, and free T3 trends lower than TSH would predict.
Absorption of oral levothyroxine happens primarily in the jejunum and upper ileum. It requires an acidic stomach to dissolve the tablet and an intact small-bowel mucosa to cross into the bloodstream. Anything that disrupts either end — H. pylori, atrophic gastritis, PPIs, celiac villous flattening, SIBO — can cut absorption by 20–40% and push your TSH up even though your dose has not changed.
Intestinal Permeability, LPS, and Zonulin
"Leaky gut" was a fringe term ten years ago and is now a measurable phenomenon published in mainstream journals. The single-cell layer lining your small intestine is sealed by tight junctions — protein complexes that decide what passes through. A protein called zonulin, discovered by Alessio Fasano at Harvard, is the main human regulator that opens those junctions. Elevated serum zonulin correlates with increased permeability in celiac disease, type 1 diabetes, Hashimoto's, and several other autoimmune conditions.
When the barrier loosens, bacterial fragments from your gut — especially lipopolysaccharide (LPS), a component of Gram-negative bacterial cell walls — leak into your bloodstream. LPS is a potent immune trigger. Even low-grade "metabolic endotoxemia" activates toll-like receptor 4 (TLR4) on immune cells and drives systemic inflammation. In Hashimoto's, that inflammation both damages thyroid tissue directly and trains your immune system to treat thyroid proteins (TPO, Tg) as enemies.
Triggers that open zonulin and widen the junctions include gliadin (from wheat gluten), certain gut bacteria, chronic stress, NSAIDs, alcohol, and intestinal infections. The cascade is: trigger → zonulin release → tight junctions loosen → LPS and undigested proteins cross → immune activation → autoimmune flare. This is why gluten elimination helps some Hashimoto's patients even when they test negative for celiac — gliadin still opens zonulin in most humans; they simply do not have the HLA-DQ2/DQ8 genes to mount a full celiac attack.
Th17 Polarization — Why Dysbiosis Drives Autoimmunity
Your immune system has several flavors of T-helper cells. Th17 cells, which produce the cytokine IL-17, are the ones implicated in Hashimoto's, psoriasis, ankylosing spondylitis, and inflammatory bowel disease. Healthy guts keep Th17 in balance with regulatory T cells (Tregs), which calm the immune response.
Specific gut bacteria shift this balance. Segmented filamentous bacteria and certain Bacteroides species push Th17 up. Clostridia cluster IV and XIVa, plus Faecalibacterium prausnitzii, push Tregs up. A gut dominated by the wrong species tilts your immune system toward chronic Th17-driven inflammation — the exact biology driving thyroid autoantibody production. Short-chain fatty acids (especially butyrate), produced when healthy bacteria ferment fiber, are the key molecule that induces Tregs. A fiber-poor, low-diversity diet starves these bugs and the brake on autoimmunity comes off.
Why Celiac and Hashimoto's Travel Together
Around 5–10% of Hashimoto's patients have celiac disease — roughly four to ten times the general-population rate. Conversely, Hashimoto's is the single most common non-GI autoimmune disease in celiac patients. The reasons are stacked:
- Shared HLA risk genes. HLA-DQ2 and HLA-DQ8 predispose to celiac; overlapping HLA-DR alleles predispose to Hashimoto's.
- Molecular mimicry. Segments of gliadin resemble thyroid peroxidase (TPO). Antibodies raised against gliadin can cross-react with thyroid tissue.
- Malabsorption. Untreated celiac damages the jejunal villi where selenium, zinc, iron, vitamin D, and levothyroxine are absorbed. The resulting deficiencies further impair thyroid function (see the minerals article).
- Shared zonulin biology. Both conditions run high serum zonulin and high permeability.
Every patient with Hashimoto's should be screened for celiac before starting a gluten-free diet (the antibodies normalize off gluten and you lose your diagnostic window). Ask for tissue transglutaminase IgA (tTG-IgA) plus total IgA. If positive or equivocal, a gastroenterologist will confirm with duodenal biopsy. If negative and you still want to trial gluten-free for Hashimoto's symptom control, that is a legitimate n-of-1 experiment — see the AIP & gluten elimination article.
SIBO as a Thyroid Trigger and Medication Blocker
Small intestinal bacterial overgrowth (SIBO) is a disproportionately common companion of hypothyroidism. One frequently cited study found roughly 54% of hypothyroid patients had a positive lactulose breath test for SIBO, compared to 5–20% in controls. The mechanism is straightforward: low T3 slows small-bowel motility, the migrating motor complex (MMC) — the cleansing wave that sweeps bacteria downstream between meals — weakens, and bacteria creep upward into the jejunum where they do not belong.
Once established, SIBO hits the thyroid back three ways:
- Absorption interference. Overgrown bacteria deconjugate bile acids, damage the brush border, and physically compete for drug uptake. Levothyroxine absorption can fall 20–40%, which looks on paper like your dose is no longer working.
- Immune activation. SIBO elevates LPS and drives chronic low-grade inflammation that feeds Hashimoto's antibody production.
- Conversion disruption. Disordered bacteria mean disordered peripheral T4-to-T3 conversion.
If your Hashimoto's antibodies will not come down, your levothyroxine dose keeps creeping up, or your gut is loud with bloating within 90 minutes of eating, ask for a lactulose or glucose breath test. Treatment is usually rifaximin 550 mg three times daily for 14 days (add neomycin for methane-dominant SIBO), followed by a prokinetic at bedtime (low-dose naltrexone, prucalopride, or ginger-artichoke extract) to restart the MMC. Full details are in the SIBO overview.
H. pylori, Low Stomach Acid, and Thyroid Antibodies
Active Helicobacter pylori infection is associated with higher TPO antibody titers and with levothyroxine malabsorption. The Sasso group in Italy published a landmark study showing that treating H. pylori in patients with unexplained rising TSH restored normal levothyroxine requirements. The mechanism is partly molecular mimicry (H. pylori antigens share sequence with thyroid proteins) and partly mechanical: chronic H. pylori causes atrophic gastritis, which flattens acid-producing parietal cells and raises gastric pH.
Levothyroxine needs an acidic stomach to dissolve. When pH rises — from H. pylori, atrophic gastritis, or PPIs — a tablet can sit intact in the stomach long enough to leave via the small bowel without being absorbed. Patients in this situation often do better on a liquid or soft-gel levothyroxine (Tirosint, Tirosint-SOL), which bypasses the dissolution step. This is a documented workaround your endocrinologist should know about.
Testing: ask for the H. pylori stool antigen or the urea breath test (blood antibody tests are cheaper but cannot distinguish past from active infection). If positive, standard eradication is a 14-day quadruple therapy (PPI + bismuth + two antibiotics). Re-test at least four weeks after finishing therapy.
The Yeast/Candida Debate
Gut Candida overgrowth is a genuine phenomenon in immunocompromised patients, after heavy antibiotic courses, or in uncontrolled diabetes. Whether it drives Hashimoto's in otherwise healthy adults is contested. The integrative-medicine community treats it aggressively; mainstream gastroenterology is skeptical because the diagnostic tests (stool culture, organic acid urine markers) have mediocre specificity.
A reasonable middle position: if you have a plausible history (prolonged antibiotics, recurrent vaginal or oral thrush, visible white tongue coating, strong carbohydrate cravings), a 4-week trial of reduced added sugar, a rotating antifungal (caprylic acid, undecylenic acid, or prescription nystatin for gut-limited exposure), and a probiotic such as Saccharomyces boulardii is cheap and low-risk. If nothing changes in four weeks, drop it and move on. Do not fall down a year-long "candida protocol" rabbit hole unless a stool test or biopsy actually confirms overgrowth.
A Practical Gut-Healing Protocol: Remove, Repair, Repopulate
Integrative gastroenterology collapses gut work into three phases. They overlap in practice, but the scaffold is useful.
1. Remove
- Gluten — 60–90 day strict trial at minimum. If you test celiac-positive, it is forever.
- Dairy — especially conventional A1 casein; many Hashimoto's patients tolerate goat, sheep, or A2 cow dairy.
- Added sugar and refined seed oils — both feed dysbiosis and drive Th17 inflammation.
- Alcohol — opens tight junctions directly.
- Unnecessary NSAIDs and PPIs — both damage barrier integrity.
- Treat identified pathogens — H. pylori, SIBO, parasites if found.
2. Repair
- L-glutamine: 5 g powder in water, 1–2 times daily between meals, 8–12 weeks. Glutamine is the preferred fuel of enterocytes and supports tight-junction protein synthesis.
- Zinc carnosine: 75 mg twice daily with food. Studied specifically for gastric and small-bowel mucosal repair; also supports thyroid enzyme function.
- Butyrate: either as a capsule (tributyrin or sodium butyrate, 300–600 mg twice daily) or, preferably, earned from eating 30+ g of fiber daily. Butyrate fuels colonocytes, tightens barrier, and induces Tregs.
- Collagen peptides / bone broth: 10–20 g daily. Glycine and proline are building blocks for mucosal repair.
- Vitamin D: correct to a serum 25(OH)D of 50–70 ng/mL. Vitamin D directly regulates tight-junction gene expression and Treg development.
- Deglycyrrhizinated licorice (DGL) or mastic gum for gastritis-type symptoms.
3. Repopulate
- Fermented foods daily: real sauerkraut, kimchi, kefir (if dairy tolerated), yogurt, miso. A 2021 Stanford trial showed 10 weeks of fermented foods increased microbiome diversity and lowered 19 inflammatory markers — a bigger effect than a high-fiber arm of the same study.
- Fiber diversity > fiber quantity: aim for 30+ plant species per week (Tim Spector's ZOE number). Different fibers feed different bacteria.
- Targeted probiotics: Lactobacillus rhamnosus GG, Bifidobacterium longum, and Saccharomyces boulardii have the most data. A rotating probiotic is more useful than the same strain forever.
- Resistant starch: cooled potatoes, green-tip bananas, cooked-and-cooled rice. Feeds butyrate producers.
Levothyroxine Timing, PPIs, and What Actually Blocks Absorption
Getting your thyroid medication into your bloodstream is a solved problem if you follow the rules. Most patients do not.
- Take it on an empty stomach, 60 minutes before food or coffee. Coffee alone can cut absorption by up to 50%. Water only in that window.
- Separate from calcium, iron, magnesium, and multivitamins by 4 hours. These minerals bind levothyroxine into an unabsorbable complex.
- Separate from PPIs and H2 blockers. These raise stomach pH and reduce dissolution. If you must be on one, consider switching to Tirosint (soft-gel) or Tirosint-SOL (liquid), which do not require acid to dissolve.
- Separate from soy, high-fiber meals, and fortified coffee creamers by at least 4 hours.
- Bedtime dosing is an acceptable alternative. Take it at least 3–4 hours after your last meal, at least 1 hour after calcium or magnesium supplements. Some patients get more consistent TSH this way because their morning routine is variable.
- Biotin warning. Biotin supplements (>5 mg) distort TSH/T4/T3 assays on most immunoassay platforms and can make your labs look falsely off. Stop biotin for 72 hours before any thyroid draw.
If your TSH is unstable despite perfect timing, the likeliest culprits in order are: H. pylori, SIBO, celiac, a new PPI, a new calcium supplement, or a switch between generic manufacturers (bioequivalence is ±12.5%, which matters at narrow therapeutic windows). See the levothyroxine article for formulation differences.
Stool Testing — What Is Worth Paying For
Stool testing ranges from medically essential to expensive theater. A patient-facing breakdown:
- Worth it, covered by insurance: H. pylori stool antigen, fecal calprotectin (screens for IBD), giardia/cryptosporidium antigen if recent travel, C. difficile toxin if recent antibiotics, standard O&P if symptomatic.
- Sometimes worth it, often out of pocket: A comprehensive stool test with 16S/shotgun sequencing (GI-MAP, Genova GI Effects) — useful if you have been symptomatic for years, have tried basic interventions, and want to target dysbiosis more specifically. Expect $300–$600.
- Mostly theater: Generic microbiome "wellness" reports that give you percentages of bacteria without clinical decision rules. Interesting, not actionable.
- Breath tests (lactulose or glucose for SIBO, urea for H. pylori) are cheaper, more targeted, and usually a better first step than a full stool panel.
The honest truth: most Hashimoto's patients do not need a $500 stool test. They need to pull gluten for 90 days, take their levothyroxine correctly, screen for H. pylori and celiac, and add fermented foods and fiber. Reserve deep testing for after you have exhausted the basics.
Key Research Papers
- Fasano A. Zonulin, regulation of tight junctions, and autoimmune diseases. Ann N Y Acad Sci. 2012.
- Knéžević J, et al. Thyroid-gut-axis: How does the microbiota influence thyroid function? Nutrients. 2020.
- Centanni M, Gargano L, Canettieri G, et al. Thyroxine in goiter, Helicobacter pylori infection, and chronic gastritis. N Engl J Med. 2006. (Sasso collaboration)
- Lauritano EC, et al. Association between hypothyroidism and small intestinal bacterial overgrowth. J Clin Endocrinol Metab. 2007.
- Wastyk HC, et al. Gut-microbiota-targeted diets modulate human immune status. Cell. 2021. (Stanford fermented-foods trial)
- Fasano A. Intestinal permeability and its regulation by zonulin: diagnostic and therapeutic implications. Physiol Rev. 2011.
PubMed Topic Searches
For readers who want to go deeper, these PubMed queries return current peer-reviewed work on the gut-thyroid axis:
- Gut microbiome and thyroid hormone metabolism
- Intestinal permeability and Hashimoto's thyroiditis
- Zonulin and autoimmune thyroid disease
- SIBO, hypothyroidism, and levothyroxine absorption
- Helicobacter pylori and thyroid autoantibodies
- Celiac disease and Hashimoto's co-occurrence
- Th17, Tregs, and microbiota in autoimmunity
- Butyrate, intestinal barrier, and autoimmune regulation
- Proton pump inhibitors and levothyroxine absorption
- L-glutamine and intestinal barrier repair
Connections
- Hashimoto's Thyroiditis Overview
- AIP and Gluten Elimination
- Levothyroxine vs NDT vs T3
- Selenium, Zinc, Iron, and Iodine
- TSH, T4, and T3 Optimal Ranges
- TPO and TgAb Antibodies
- Reverse T3 and Low T3 Syndrome
- Pregnancy and Hashimoto's
- Hashimoto, POTS, and MCAS Overlap
- SIBO
- Celiac Disease
- Gut Healing
- Elimination Diet
- Glutamine
- Bloating
- Fermented Foods
- Thyroid Disorders