Gout
1. Overview
Gout is a common and intensely painful form of inflammatory arthritis caused by the deposition of monosodium urate (MSU) crystals in joints and surrounding tissues. It results from chronic hyperuricemia, defined as a serum urate level exceeding 6.8 mg/dL, the saturation point at which urate crystallizes under physiological conditions. Gout is one of the oldest recognized diseases in medical history, frequently referred to as the "disease of kings" due to its historical association with rich diets and alcohol consumption.
The disease follows a well-characterized clinical course progressing through distinct stages: asymptomatic hyperuricemia, acute gouty arthritis, intercritical periods, and chronic tophaceous gout. Acute attacks typically present as sudden-onset, excruciating monoarticular arthritis, most classically affecting the first metatarsophalangeal (MTP) joint, a presentation known as podagra. Without appropriate management, gout can progress to chronic, destructive arthropathy with formation of tophi, subcutaneous deposits of urate crystals.
Gout is classified among the crystal arthropathies and is the most common inflammatory arthritis in men. Unlike many rheumatic diseases, gout is one of the few forms of arthritis for which the underlying cause is thoroughly understood and for which curative therapy exists through sustained urate-lowering treatment. The disease is strongly associated with metabolic syndrome, cardiovascular disease, chronic kidney disease, and premature mortality.
2. Epidemiology
Gout is the most prevalent inflammatory arthritis worldwide, affecting an estimated 41 million people globally. In the United States, the prevalence is approximately 3.9% of adults (9.2 million individuals), with incidence rates that have been rising steadily over the past several decades. The disease disproportionately affects men, with a male-to-female ratio of approximately 3-4:1, though this gap narrows after menopause when women lose the uricosuric protective effect of estrogen.
Peak incidence in men occurs between ages 40 and 60, while in women, gout typically develops after age 60. Prevalence increases markedly with age, reaching 12% in men over 80 years old. There are notable racial and ethnic disparities: gout prevalence is higher among Pacific Islander populations (including Maori and Samoan peoples), where rates can exceed 10%, as well as among African American men compared to Caucasian men. The increasing global prevalence is attributed to rising rates of obesity, metabolic syndrome, hypertension, chronic kidney disease, and dietary changes including greater consumption of fructose-sweetened beverages.
3. Pathophysiology
Uric Acid Metabolism
Uric acid is the end product of purine metabolism in humans. Unlike most mammals, humans lack the enzyme uricase (urate oxidase), which converts uric acid to the more soluble allantoin. This evolutionary loss of uricase function results in baseline serum urate levels that are substantially higher in humans than in other species. Purines are derived from both endogenous sources (cellular turnover, de novo synthesis) and exogenous dietary intake. Approximately two-thirds of daily uric acid elimination occurs via renal excretion, with the remaining one-third eliminated through intestinal uricolysis by gut bacteria.
Crystal Formation and Deposition
When serum urate levels exceed 6.8 mg/dL, the supersaturation threshold, MSU crystals begin to form and deposit in articular and periarticular tissues. Crystal formation is favored by lower temperatures (explaining the predilection for peripheral joints), lower pH, mechanical stress, and the presence of certain connective tissue components including cartilage and synovium. Crystals may be present in joints for years before triggering an acute inflammatory response.
Inflammatory Cascade
The acute gouty attack is triggered when MSU crystals are recognized by the innate immune system. Resident macrophages and dendritic cells phagocytose the crystals, leading to activation of the NLRP3 inflammasome, a critical intracellular sensing platform. Inflammasome activation triggers caspase-1 mediated processing and release of interleukin-1 beta (IL-1beta), a potent pro-inflammatory cytokine that initiates a cascade of neutrophil recruitment, vasodilation, and intense local inflammation. The resulting massive neutrophilic infiltrate produces the cardinal signs of acute gouty arthritis: erythema, warmth, swelling, and exquisite tenderness.
Resolution of Inflammation
Acute gout attacks are characteristically self-limiting, typically resolving within 7-14 days even without treatment. Resolution involves neutrophil apoptosis, macrophage-mediated clearance of apoptotic cells (efferocytosis), and the production of anti-inflammatory mediators including transforming growth factor beta (TGF-beta), interleukin-10, and specialized pro-resolving mediators. The formation of neutrophil extracellular traps (NETs) also contributes to inflammation resolution by aggregating and degrading pro-inflammatory cytokines.
4. Etiology and Risk Factors
Genetic Factors
- ABCG2 gene variants — encoding a urate transporter in the kidney and gut; dysfunctional variants account for approximately 10% of all gout cases
- SLC2A9 (GLUT9) polymorphisms — major renal urate transporter; variants are the strongest genetic determinant of serum urate levels
- SLC22A12 (URAT1) variants — encoding the primary renal urate reabsorption transporter
- Familial predisposition — heritability of serum urate levels estimated at 40-70%
- Rare monogenic disorders — Lesch-Nyhan syndrome (HGPRT deficiency), PRPS superactivity, familial juvenile hyperuricemic nephropathy
Dietary and Lifestyle Factors
- High-purine foods — organ meats (liver, kidney, sweetbreads), red meat, shellfish, anchovies, sardines
- Alcohol consumption — particularly beer (high in guanosine) and spirits; wine has a lesser effect
- Fructose-sweetened beverages — fructose metabolism increases purine degradation and uric acid production
- Obesity — increases uric acid production and decreases renal excretion; body mass index is strongly correlated with serum urate
- Dehydration — reduces renal urate clearance
Medical Conditions
- Chronic kidney disease — reduced urate excretion; present in 20-40% of gout patients
- Metabolic syndrome — insulin resistance reduces renal urate excretion
- Hypertension — associated with reduced renal blood flow and urate excretion
- Heart failure — impaired renal perfusion and use of loop diuretics
- Myeloproliferative and lymphoproliferative disorders — increased cell turnover elevates purine load
- Psoriasis — high cell turnover increases purine metabolism
- Organ transplantation — related to calcineurin inhibitor (cyclosporine) use
Medications
- Thiazide and loop diuretics — most common drug-induced cause of hyperuricemia
- Low-dose aspirin — inhibits renal urate excretion at doses below 2 g/day
- Cyclosporine — reduces renal urate clearance; gout affects up to 10-15% of transplant recipients on cyclosporine
- Pyrazinamide and ethambutol — anti-tuberculosis drugs that impair urate excretion
- Niacin (nicotinic acid) — competes with uric acid for renal tubular excretion
5. Clinical Presentation
Acute Gouty Arthritis
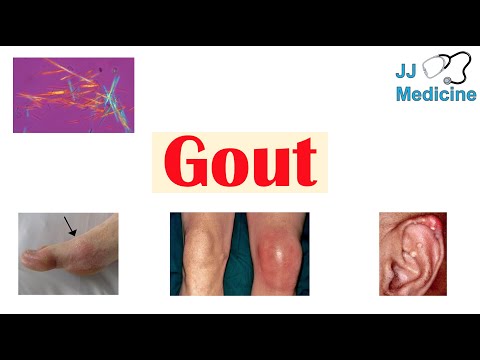
The hallmark of gout is the acute flare, characterized by sudden onset of severe, excruciating joint pain that typically reaches maximum intensity within 6-12 hours. The affected joint becomes intensely erythematous, swollen, warm, and exquisitely tender, often so painful that even the weight of a bedsheet is intolerable (the classic "bedsheet sign"). Attacks frequently begin at night and may be preceded by prodromal tingling or discomfort. The first metatarsophalangeal joint (podagra) is the most commonly affected site, involved in approximately 50% of first attacks and 70-90% of patients at some point. Other commonly involved joints include the ankle, midfoot, knee, wrist, fingers, and elbow.
Intercritical Gout
Between acute attacks, patients enter symptom-free intervals known as intercritical periods. Early in the disease course, these intervals may last months to years. However, without urate-lowering therapy, the frequency of attacks typically increases and the intercritical periods shorten. Importantly, MSU crystals remain present in joints during intercritical periods, and subclinical inflammation persists even in the absence of overt symptoms.
Chronic Tophaceous Gout
With prolonged, untreated hyperuricemia, patients may develop tophi, which are firm, chalky, nodular deposits of MSU crystals surrounded by a chronic granulomatous inflammatory reaction. Tophi most commonly occur on the ears (helix), fingers, hands, elbows (olecranon bursa), knees, Achilles tendons, and feet. Large tophi can erode through the skin and discharge a white, chalky material. Tophaceous gout is associated with chronic joint destruction, deformity, and disability.
Systemic Features
- Fever — may occur during severe acute attacks, sometimes reaching 38-39 degrees Celsius
- Malaise and chills — mimicking septic arthritis or cellulitis
- Polyarticular involvement — occurs in approximately 10-15% of initial attacks; more common in elderly patients and women
- Periarticular inflammation — bursitis, tenosynovitis, and cellulitis-like presentations
6. Diagnosis
Definitive Diagnosis: Synovial Fluid Analysis
The gold standard for gout diagnosis is identification of negatively birefringent, needle-shaped MSU crystals under compensated polarized light microscopy in synovial fluid or tophus aspirate. Synovial fluid during an acute attack typically shows inflammatory characteristics with white blood cell counts of 10,000-100,000 cells/microL, predominantly neutrophils. Intracellular crystals (within neutrophils) confirm active crystal-induced inflammation.
Laboratory Studies
- Serum uric acid — elevated in most patients, though levels may be paradoxically normal or low during acute attacks (urate is consumed by inflammation); serial measurements are more informative
- Complete blood count — leukocytosis common during acute flares
- Inflammatory markers — elevated ESR and CRP during acute attacks
- Renal function tests — to assess for comorbid chronic kidney disease
- 24-hour urine uric acid — to classify patients as overproducers (greater than 800 mg/day) versus underexcretors
- Lipid panel and fasting glucose — screening for metabolic syndrome
Imaging Studies
- Conventional radiography — may be normal early; classic findings in chronic gout include well-defined periarticular erosions with overhanging edges ("rat bite" erosions), preserved joint space until late disease, and soft tissue tophi with or without calcification
- Musculoskeletal ultrasound — can detect the "double contour sign" (hyperechoic enhancement of articular cartilage surface representing MSU crystal deposition), tophi, and joint effusions; sensitivity approximately 85% for established gout
- Dual-energy CT (DECT) — color-codes urate deposits based on differential X-ray attenuation; highly specific (approximately 95%) for MSU crystal detection; useful for detecting occult tophi and quantifying crystal burden
2015 ACR/EULAR Classification Criteria
The 2015 ACR/EULAR gout classification criteria use a scoring system incorporating clinical, laboratory, and imaging domains. Entry criterion requires at least one episode of joint swelling, pain, or tenderness. Sufficient criterion is detection of MSU crystals in symptomatic joint or tophus. If crystals are not demonstrated, a point-based system (threshold score of 8 or greater out of 23) considers pattern of joint involvement, characteristics of the episode, time course, clinical evidence of tophi, serum urate level, synovial fluid analysis, and imaging evidence. These criteria have a sensitivity of 92% and specificity of 89%.
7. Treatment
Acute Flare Management
- Colchicine — most effective when initiated within 12-36 hours of attack onset; current low-dose regimen (1.2 mg followed by 0.6 mg one hour later) is as effective as high-dose with fewer side effects; gastrointestinal toxicity is the main limitation
- NSAIDs — indomethacin (50 mg three times daily) has been traditionally used, but naproxen (500 mg twice daily) and other NSAIDs are equally effective; use full anti-inflammatory doses and continue until flare resolves
- Corticosteroids — oral prednisone (30-40 mg/day for 5-7 days), intra-articular injection (especially for monoarticular involvement), or intramuscular triamcinolone for patients who cannot take oral medications; preferred in patients with renal impairment or NSAID contraindications
- IL-1 inhibitors — anakinra (100 mg subcutaneously daily for 3 days) or canakinumab (150 mg single subcutaneous dose) for refractory cases; canakinumab is FDA-approved for gout in some regions
- ACTH (corticotropin) — 40-80 IU intramuscularly or subcutaneously; stimulates endogenous cortisol and has direct anti-inflammatory effects via melanocortin receptors
Urate-Lowering Therapy (ULT)
- Allopurinol — first-line xanthine oxidase inhibitor; start low (100 mg/day, or 50 mg/day in CKD stage 3 or greater) and titrate every 2-4 weeks to target serum urate below 6 mg/dL; maximum dose 800 mg/day; requires HLA-B*5801 screening before initiation (mandatory in Southeast Asian, African American, and Korean populations due to risk of severe hypersensitivity)
- Febuxostat — selective xanthine oxidase inhibitor; 40-80 mg/day; does not require dose adjustment for mild-moderate renal impairment; FDA boxed warning regarding cardiovascular risk based on CARES trial (though subsequent FAST trial showed no increased risk)
- Probenecid — uricosuric agent; 500 mg twice daily up to 2 g/day; requires adequate renal function (eGFR greater than 50 mL/min) and absence of nephrolithiasis or uric acid overproduction
- Lesinurad — selective URAT1 inhibitor; 200 mg/day used only in combination with a xanthine oxidase inhibitor; available as fixed-dose combination with allopurinol (Duzallo)
- Pegloticase — pegylated recombinant uricase; 8 mg IV infusion every 2 weeks; reserved for severe refractory tophaceous gout unresponsive to conventional ULT; can dramatically reduce tophus burden; risk of infusion reactions and anaphylaxis; loss of efficacy with development of anti-drug antibodies
Anti-Inflammatory Prophylaxis During ULT Initiation
Initiating or adjusting ULT frequently triggers gout flares due to crystal mobilization. Concurrent anti-inflammatory prophylaxis is recommended for at least 3-6 months (ACR recommends continuing for 3-6 months after reaching target serum urate). Options include low-dose colchicine (0.6 mg once or twice daily) or low-dose NSAID (naproxen 250 mg twice daily). For patients who cannot tolerate either, low-dose prednisone (5-10 mg/day or less) may be used.
Treat-to-Target Strategy
Current guidelines from the ACR (2020) and EULAR (2016) endorse a treat-to-target approach aiming for a serum urate level below 6 mg/dL (360 micromol/L) in most patients, and below 5 mg/dL in patients with tophi, chronic arthropathy, or frequent flares. Maintaining serum urate below the crystallization threshold promotes gradual dissolution of existing MSU crystal deposits and prevents new crystal formation, ultimately leading to cessation of flares and regression of tophi.
8. Complications
- Chronic tophaceous arthropathy — destructive joint erosions, deformity, and functional disability from persistent MSU crystal deposition
- Uric acid nephrolithiasis — occurs in 10-25% of gout patients; uric acid stones account for approximately 10% of all kidney stones
- Urate nephropathy — chronic tubulointerstitial damage from medullary urate crystal deposition; historically a major cause of renal failure in gout, now rare with modern treatment
- Cardiovascular disease — gout is an independent risk factor for myocardial infarction, stroke, heart failure, and cardiovascular mortality; hyperuricemia promotes endothelial dysfunction, oxidative stress, and vascular inflammation
- Chronic kidney disease — bidirectional relationship; both a cause and consequence of hyperuricemia
- Metabolic syndrome — gout patients have markedly elevated rates of obesity, hypertension, dyslipidemia, insulin resistance, and type 2 diabetes
- Septic arthritis — must be excluded in every acute monoarthritis presentation; gout and septic arthritis can coexist in the same joint
- Disability and impaired quality of life — recurrent flares and chronic pain significantly impact work productivity and daily functioning
- Carpal tunnel syndrome and tendon rupture — from tophaceous deposits compressing nerves or weakening tendons
9. Prognosis
Gout has an excellent prognosis when properly treated, as it is one of the few rheumatic diseases for which the underlying pathophysiology can be fully addressed. With sustained urate-lowering therapy achieving target serum urate levels below 6 mg/dL, acute flares cease within 1-2 years and tophi dissolve completely over 1-5 years depending on their size and the degree of serum urate reduction achieved.
However, gout remains significantly undertreated globally. Studies consistently show that fewer than 50% of gout patients receive urate-lowering therapy, and among those treated, fewer than 40% achieve target serum urate levels. Untreated or poorly managed gout progresses to chronic tophaceous disease in approximately 12-35% of patients over 5-10 years, with increasing frequency and severity of flares, polyarticular involvement, and joint destruction.
Gout is associated with increased all-cause mortality, primarily driven by cardiovascular disease. A meta-analysis found that gout increases the risk of cardiovascular mortality by approximately 29% and all-cause mortality by 24%. Whether urate-lowering therapy reduces cardiovascular risk remains an active area of investigation.
10. Prevention
Prevention strategies for gout target both primary prevention (avoiding development of gout in individuals with hyperuricemia) and secondary prevention (preventing recurrent flares and disease progression):
- Dietary modification — limit intake of high-purine foods (organ meats, certain seafood), reduce alcohol consumption (especially beer), avoid fructose-sweetened beverages; increase consumption of low-fat dairy products, cherries, and coffee (all associated with lower urate levels)
- Weight management — achieving and maintaining a healthy body weight reduces serum urate levels; each unit decrease in BMI is associated with approximately 0.5 mg/dL decrease in serum urate
- Adequate hydration — maintaining urine output of at least 2 liters per day enhances renal urate excretion
- Medication review — consider alternatives to thiazide diuretics, low-dose aspirin, and other hyperuricemia-inducing medications when possible; losartan and calcium channel blockers have mild uricosuric effects and are preferred antihypertensives in gout patients
- Management of comorbidities — aggressive treatment of hypertension, diabetes, dyslipidemia, and chronic kidney disease
- Vitamin C supplementation — 500 mg/day has a modest uricosuric effect, reducing serum urate by approximately 0.5 mg/dL
- Adherence to urate-lowering therapy — lifelong therapy with regular monitoring of serum urate levels is essential for sustained disease control
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Research Papers
- Connections
- Featured Videos
Research Papers
Curated PubMed topic searches on Gout. Each link opens a live PubMed query so the result set stays current as new studies are indexed.
- PubMed topic search: Gout review pathophysiology
- PubMed topic search: Hyperuricemia gout
- PubMed topic search: Allopurinol gout trial
- PubMed topic search: Febuxostat gout cardiovascular
- PubMed topic search: Colchicine acute gout
- PubMed topic search: Gout dietary management
- PubMed topic search: Urate-lowering therapy gout
- PubMed topic search: Pegloticase refractory gout
- PubMed topic search: Dual-energy CT gout crystals
- PubMed topic search: Gout kidney disease
- PubMed topic search: ACR gout management guideline
- PubMed topic search: Tophaceous gout treatment
Connections
Featured Videos

Gout: Everything You Need to Know

Gout - causes, symptoms, diagnosis, treatment, pathology

Gout Attack Symptoms, Treatment, Diet, Pharmacology Nursing Lecture for NCLEX Explained

🔥What is Gout? TRUE Causes & Treatments! [Symptoms, Diet & Diagnosis]

🔥Top 7 Gout Foot & Big Toe HOME Treatments [+1 BIG SECRET]

Effective Treatments for Gout: The Ultimate Guide

Chronic Gout Treatment - The Second Phase of Treating Gout (6 of 6)

Gout Relief at Home is EASIER Than You Think!
Gout | Causes, Pathophysiology, Risk Factors (ex. Diet), Symptoms, Diagnosis, Treatment