Hot Flashes & Night Sweats: Vasomotor Symptoms
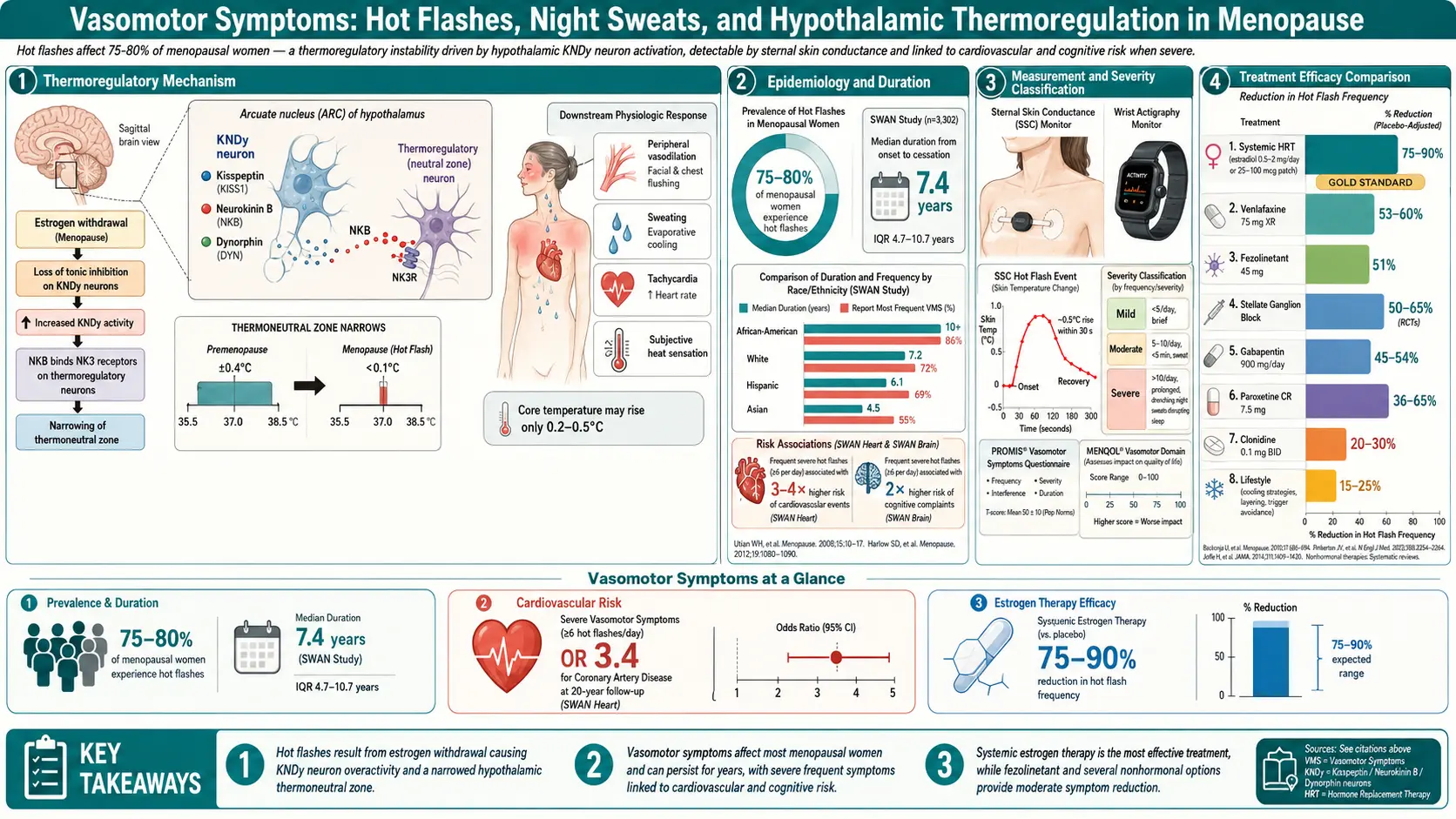
Hot flashes and night sweats — doctors call them vasomotor symptoms (VMS) — are the single most recognizable feature of menopause. They are also the most treatable. If you are drenching your pajamas at 3 a.m., fanning yourself through a work meeting, or avoiding turtlenecks you used to love, you are in very large company: roughly 75% of women going through menopause experience vasomotor symptoms, and 30–50% rate them as moderate to severe. This article walks through what is happening in your brain, how long it typically lasts, what actually works to stop the flashes, and what quietly does not work despite being heavily marketed.
Table of Contents
- What a Hot Flash Actually Is
- The Neurobiology: KNDy Neurons and a Narrowed Thermoregulatory Zone
- How Long Will This Last?
- Triggers — What Sets Flashes Off
- Measuring What You Have — Diaries and Severity
- First-Line Treatment: Estradiol
- Second-Line: Fezolinetant (Veozah)
- SSRIs, SNRIs, Gabapentin, and Oxybutynin
- Stellate Ganglion Block and Menopause CBT
- Lifestyle Strategies That Actually Help
- What Does Not Work
- Cost Reality Check
- Key Research Papers
- PubMed Topic Searches
- Connections
- Featured Videos
What a Hot Flash Actually Is
A hot flash is a sudden, involuntary heat-dissipation event. It typically starts with a wave of warmth rising up the chest, neck, and face, peaks within 1–3 minutes, and is followed by flushing of the skin, sweating, and often a chill as the body overcools. Most women also notice a racing heart and a kind of internal alarm that comes with it. At night, the same physiology produces night sweats — drenching episodes that wake you up, force a change of pajamas or sheets, and fragment sleep.
What you are feeling is real and measurable. During a flash, skin blood flow can increase tenfold, skin temperature rises 1–7°C, and core body temperature drops slightly afterward as the body dumps heat. This is the same machinery your body uses on a hot day at the beach — it is just being triggered at the wrong times, for the wrong reasons.
The Neurobiology: KNDy Neurons and a Narrowed Thermoregulatory Zone
For decades, hot flashes were blamed vaguely on "low estrogen." The modern explanation is much more specific, and it is the foundation for a new class of drugs.
Deep in the hypothalamus sits a small cluster of cells called KNDy neurons — named for the three signaling molecules they release: kisspeptin, neurokinin B, and dynorphin. These neurons normally receive a calming estrogen signal. When estrogen falls during menopause, KNDy neurons lose that brake and become hypertrophied and hyperactive. They pour out neurokinin B, which activates the NK3 receptor (NK3R) on nearby temperature-regulating neurons in the median preoptic nucleus.
The practical result is that your brain's thermoneutral zone — the narrow internal-temperature range in which you feel neither hot nor cold — collapses from roughly 0.4°C wide down to almost nothing. A tiny rise in core temperature, the kind that used to be invisible, now punches straight through the "too hot" threshold, and your hypothalamus slams down on the cooling response: vasodilation, sweating, flushing. Moments later it overshoots, triggers the "too cold" threshold, and you shiver.
This is why hot flashes are not a personality flaw or a sign of anxiety — they are a neurological misfire. And it is why the newest non-hormonal drug, fezolinetant, works: it simply blocks the NK3 receptor that KNDy neurons are screaming into.
How Long Will This Last?
Longer than most women are told. The landmark Study of Women's Health Across the Nation (SWAN) followed more than 3,000 women for over a decade and found that the median total duration of frequent vasomotor symptoms is 7.4 years, and the median duration after the final menstrual period is about 4.5 years. Plenty of women land on either side of that median. Key patterns:
- African-American women have the longest median duration — over 10 years in SWAN.
- Women whose symptoms start early, in perimenopause before periods stop, tend to have the longest total courses.
- Women whose symptoms start only after the last period often have the shortest, averaging 3–4 years.
- Japanese-American and Chinese-American women in SWAN reported the shortest durations, though cultural reporting differences may play a role.
The older line that "you just have to wait a couple of years and it will pass" is, for most women, wrong. If symptoms are disrupting your sleep, work, or relationships, waiting it out is a clinical choice, not a default — and it is not the choice most specialists now recommend.
Triggers — What Sets Flashes Off
Once your thermoneutral zone is narrow, small perturbations tip you over. Common triggers that patients identify in diaries:
- Caffeine — especially afternoon coffee; raises core temperature and sympathetic tone.
- Alcohol — red wine is the most frequent single offender; causes skin vasodilation and disrupts sleep architecture.
- Spicy food — capsaicin activates the same heat-sensing receptors (TRPV1) that the hypothalamus uses.
- Stress and anxiety — sympathetic surges raise core temperature directly.
- Warm rooms, hot showers, heated car seats, heavy blankets.
- Sugar crashes and skipped meals — blood-sugar swings trigger adrenaline, which triggers flashes.
- Tight, synthetic, or non-breathable clothing.
Identifying your personal triggers will not cure VMS, but it usually cuts frequency 15–30% for motivated women — enough to matter if your baseline is moderate.
Measuring What You Have — Diaries and Severity
Before you walk into a clinician's office, spend 7–14 days on a hot flash diary. You can do this on paper or in any notes app. For each episode log:
- Time of day
- Severity (mild = warm and aware, moderate = sweating, severe = drenching and interrupts activity)
- Trigger if you noticed one
- Whether it woke you up
This gives you two numbers that doctors use and that trial protocols use: daily frequency and moderate-to-severe frequency. The FDA requires at least seven moderate-to-severe hot flashes per day (or 50/week) as the entry threshold for VMS drug trials, which is useful calibration: if you are hitting that number, you are squarely in the population where every treatment below has been proven to work.
First-Line Treatment: Estradiol
If you have no contraindications, systemic estradiol is the most effective treatment for vasomotor symptoms that exists. Randomized trials consistently show 75–90% reduction in hot flash frequency and severity — numbers no non-hormonal option matches.
Key points for the VMS context (full details are in the Estradiol Formulations article):
- Transdermal is preferred over oral for most women. Patches (0.025–0.1 mg/day), gels, and sprays bypass the liver and carry a lower risk of blood clots and stroke than oral estradiol — an important distinction that the old WHI headlines glossed over.
- Starting doses: 0.05 mg/day patch is a standard starting point for moderate-to-severe VMS; mild VMS may respond to 0.025 mg/day.
- Relief timeline: many women feel the difference within 2–4 weeks; full effect takes 8–12 weeks.
- Progesterone is mandatory if you still have a uterus, to protect the endometrial lining. See the micronized progesterone article.
- Contraindications: active or recent breast cancer, active clot disease, active liver disease, unexplained vaginal bleeding, pregnancy. See the risks article for the nuanced picture.
Second-Line: Fezolinetant (Veozah)
For women who cannot or will not take estrogen, the major news of 2023 was fezolinetant — a first-in-class NK3 receptor antagonist that targets the KNDy-neuron pathway described above. The FDA approved it in May 2023 after the SKYLIGHT trial program.
- Dose: 45 mg orally once daily. No dose titration.
- Efficacy: roughly 50–65% reduction in moderate-to-severe VMS frequency by week 12, versus about 30% for placebo. Onset is within the first 1–2 weeks — faster than most SSRIs.
- Mechanism: blocks neurokinin B binding at NK3R, restoring a wider thermoneutral zone without touching estrogen pathways.
- Monitoring: baseline liver function tests, then at 3, 6, and 9 months. Elevated liver enzymes in a small minority led the FDA to add a boxed warning in 2024; most elevations are reversible on stopping.
- Not for women with: cirrhosis, severe kidney impairment, or concurrent strong CYP1A2 inhibitors (fluvoxamine, ciprofloxacin).
For a breast-cancer survivor or a woman with a strong family history who has been told hormones are off the table, fezolinetant is the most important new option in thirty years.
SSRIs, SNRIs, Gabapentin, and Oxybutynin
Before fezolinetant, these were the backbone of non-hormonal therapy. They still are, for women who cannot afford fezolinetant or who have liver concerns.
- Paroxetine 7.5 mg (Brisdelle) — the only SSRI FDA-approved specifically for VMS (2013). About 30–40% reduction in hot flashes versus placebo. The 7.5 mg dose is below antidepressant range and better tolerated. Do not combine with tamoxifen — paroxetine is a strong CYP2D6 inhibitor and reduces tamoxifen's active metabolite.
- Venlafaxine 37.5–75 mg (SNRI) — off-label but heavily used, especially in breast-cancer survivors on tamoxifen because it does not interfere. About 40–60% reduction. Start at 37.5 mg for a week, then up to 75 mg. Taper to stop — abrupt discontinuation causes a miserable flu-like syndrome.
- Escitalopram 10–20 mg — the best non-paroxetine SSRI option; MsFLASH trial data show about 47% reduction.
- Gabapentin 300–900 mg at bedtime — particularly useful for night sweats because it is sedating. Start at 300 mg nightly; can titrate up. Helpful when sleep disruption is the main complaint.
- Oxybutynin 2.5–5 mg twice daily — an anticholinergic originally for overactive bladder. About 60–75% reduction in frequency. The catch: recent studies link long-term anticholinergic exposure to cognitive decline, so it is usually reserved for shorter courses in women who cannot use other options.
- Clonidine 0.1 mg/day — older, modest efficacy, side effects (dry mouth, drop in blood pressure) usually put it last.
Stellate Ganglion Block and Menopause CBT
Stellate ganglion block (SGB) is an ultrasound-guided injection of local anesthetic into a cluster of sympathetic nerves in the neck. It was originally used for chronic pain. Small randomized trials in menopausal women show about a 50% reduction in moderate-to-severe hot flashes, lasting weeks to months per injection. It is not first-line, but it is a real option for women with severe refractory VMS — particularly breast-cancer survivors who cannot use hormones or systemic drugs. A pain-medicine anesthesiologist performs the procedure; it takes about 15 minutes.
Cognitive Behavioral Therapy for menopausal symptoms (CBT-Meno) is a structured 4–6 session protocol developed by Myra Hunter at King's College London. It does not make hot flashes stop, but it reliably reduces how bothersome they are — the subjective distress that actually drives the damage to sleep, work, and mood. Randomized trials show improvement comparable in effect size to low-dose paroxetine on bother scales. If your flashes are moderate but the anxiety around them is the worst part, this is a high-yield intervention.
Lifestyle Strategies That Actually Help
None of these replace the treatments above if you have moderate-to-severe VMS, but stacked together they make a real difference for many women:
- Layered clothing — a thin base layer you can peel off; natural fibers (cotton, linen, merino) over synthetics.
- Fan at the bedside — the single most recommended item in every menopause clinic. Many women use two: one oscillating, one pointed at the face.
- Cooling pillow and moisture-wicking sheets — bamboo or Tencel sheets, a gel or phase-change pillow. Chipgel pillows (the ones that feel cold to the touch) help more than they should for how cheap they are.
- Lower the bedroom thermostat to 65–68°F.
- Paced respiration — slow 6-breath-per-minute breathing at flash onset may shorten duration. Low-quality evidence, zero cost, zero side effects.
- Weight loss if overweight — the MsFLASH lifestyle trial showed modest VMS reduction with a 10% body-weight loss.
- Regular exercise — does not reliably reduce VMS frequency but improves sleep quality, mood, and insulin sensitivity, all of which amplify the benefit of anything else you do.
What Does Not Work (Despite Marketing)
Supplement aisles at Whole Foods and the Amazon search for "menopause" are dominated by products with poor or negative evidence. The honest list:
- Black cohosh — the most-studied herbal remedy. Results are genuinely mixed: some European trials show modest benefit, the large NIH HALT trial showed no benefit over placebo. Rare but real cases of liver injury have been reported. If you try it, a 3-month trial is reasonable, but do not expect estradiol-level relief.
- Soy isoflavones — large meta-analyses show a real but modest effect, in the 15–20% reduction range. Better than nothing, far weaker than any prescription option. S-equol (a specific isoflavone metabolite) may be slightly better in the subset of women who can produce it.
- Evening primrose oil — multiple randomized trials show no benefit over placebo for VMS. Save your money.
- Dong quai, red clover, wild yam cream, "bioidentical" compounded creams from pellet clinics — insufficient evidence, unregulated dosing, and for pellets, supraphysiologic estrogen and testosterone levels that carry real risks. FDA-approved transdermal estradiol achieves the same "bioidentical" profile safely and far more cheaply.
- Vitamin E 400 IU — trials show a statistically significant but clinically trivial reduction (about one flash per day). Not worth it.
- Acupuncture — large sham-controlled trials show benefit equal to sham acupuncture. The ritual may help; the needles do not appear to.
Cost Reality Check
Costs in the United States, approximate, as of the mid-2020s:
- Generic estradiol patch: $10–$40/month with insurance, often under $15 on GoodRx without.
- Micronized progesterone (generic): $15–$30/month.
- Paroxetine 7.5 mg (Brisdelle): branded is ~$200/month; generic paroxetine 10 mg cut in half is <$10.
- Venlafaxine generic: <$15/month.
- Gabapentin generic: <$10/month.
- Fezolinetant (Veozah): around $550–$650/month list; manufacturer copay cards can bring insured patients to $30/month but the cash price remains the big barrier. Insurance coverage has improved through 2024–2025 but still requires prior authorization at most plans.
- Stellate ganglion block: $500–$1,500 per procedure; usually covered for chronic-pain indications, inconsistently covered for VMS.
- CBT-Meno: often covered under mental-health benefits; self-guided workbook versions are free or under $30.
The cheapest effective regimen for most women is a generic estradiol patch plus generic micronized progesterone — under $50/month total, often under $25. That is not a marketing number; that is what the treatment actually costs.
Key Research Papers
- Johnson KA, Martin N, Nappi RE, et al. Efficacy and safety of fezolinetant in moderate-to-severe vasomotor symptoms associated with menopause (SKYLIGHT 1): a phase 3 randomised controlled study. Lancet. 2023.
- Stearns V, Beebe KL, Iyengar M, Dube E. Paroxetine controlled release in the treatment of menopausal hot flashes: a randomized controlled trial. JAMA. 2003;289(21):2827–2834.
- Loprinzi CL, Kugler JW, Sloan JA, et al. Venlafaxine in management of hot flashes in survivors of breast cancer: a randomised controlled trial. Lancet. 2000;356:2059–2063.
- The 2023 nonhormone therapy position statement of The North American Menopause Society. Menopause. 2023;30(6):573–590.
- Avis NE, Crawford SL, Greendale G, et al. Duration of menopausal vasomotor symptoms over the menopause transition (SWAN). JAMA Intern Med. 2015;175(4):531–539.
- Hunter MS, Ayers B, Smith M. The Hot Flush Behavior Scale and CBT for menopausal hot flashes. Menopause. 2011.
PubMed Topic Searches
For further reading, the following PubMed searches return current peer-reviewed work on vasomotor symptoms and their treatment:
- Fezolinetant and vasomotor symptoms
- KNDy neurons, menopause, and hot flashes
- Transdermal estradiol for vasomotor symptoms
- Paroxetine and hot flashes
- Venlafaxine for hot flashes in breast cancer survivors
- Gabapentin for menopausal night sweats
- Oxybutynin for vasomotor symptoms
- Stellate ganglion block for hot flashes
- CBT for menopausal hot flashes
- SWAN study and vasomotor symptom duration
- Black cohosh and hot flashes
Connections
- Estradiol Formulations: Patch, Gel, Oral Routes
- Micronized Progesterone vs Synthetic Progestins
- HRT Risks: Breast Cancer, Clots and Stroke
- Perimenopause Symptom Tracker and Hormonal Testing
- Perimenopause
- Testosterone Therapy for Women
- Genitourinary Syndrome of Menopause and Vaginal Estrogen
- Non-Hormonal Options: SSRIs, Gabapentin, Fezolinetant
- Bone Loss Prevention and HRT
- Ashwagandha
- Insomnia
- Menopause and HRT
- Anxiety
- Black Cohosh
- Hormone Panel
- Depression
- Cardiovascular Disease
- Sleep Hygiene