HRT Risks: Breast Cancer, Clots, and Stroke
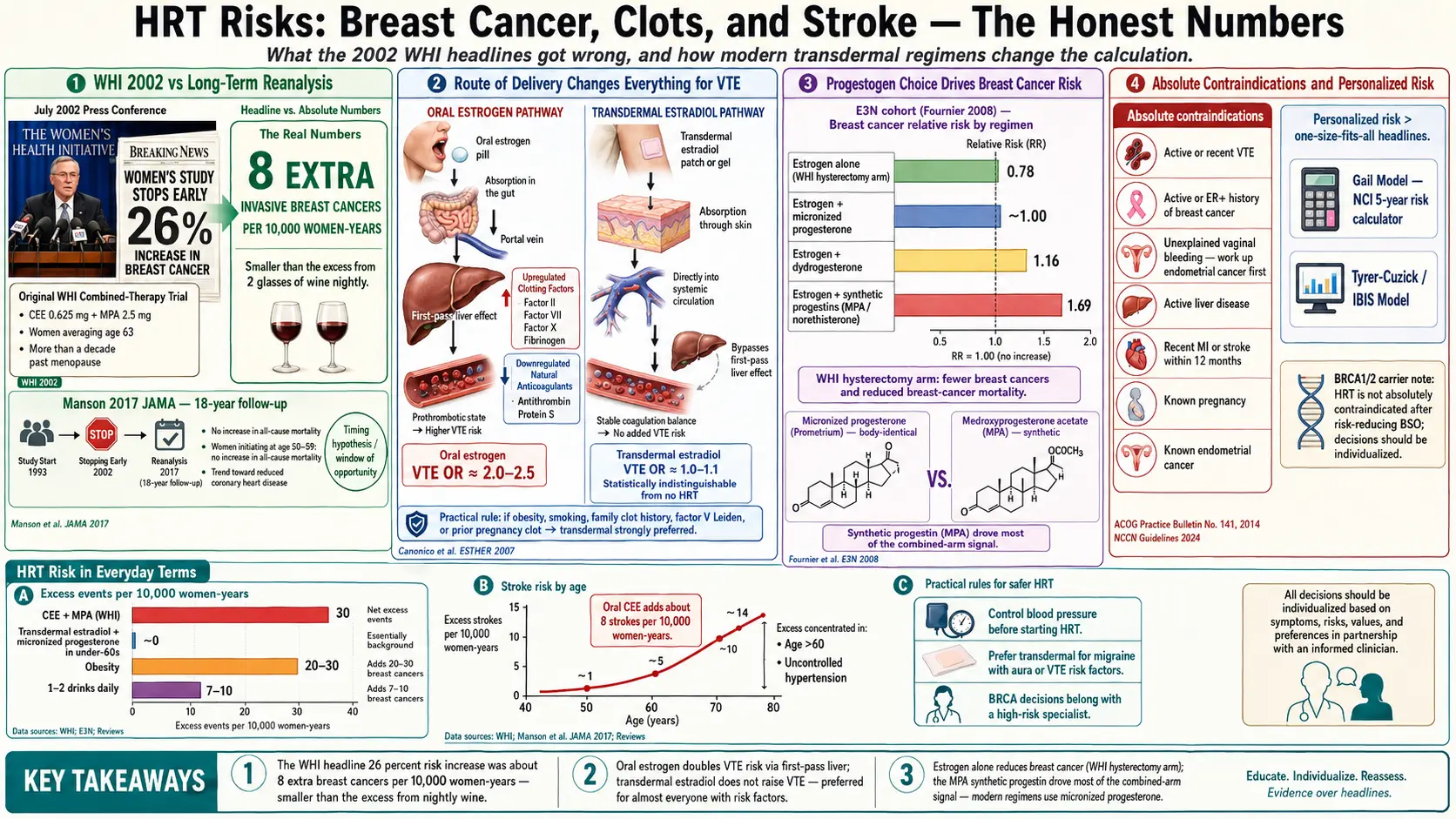
Almost everything most women think they know about hormone therapy risk comes from a single July 2002 press conference — and most of it was wrong, or at least wildly oversimplified. The Women's Health Initiative (WHI) investigators halted the combined estrogen-plus-progestin arm of their trial early, announced a "26% increase in breast cancer," and set off a panic that caused millions of women to throw their patches in the trash overnight. Prescriptions for HRT in the United States fell by more than half within two years. What almost nobody reported at the time was the absolute number: 8 extra breast cancer cases per 10,000 women per year. That is smaller than the excess breast-cancer risk from drinking two glasses of wine a night.
This page walks through what modern re-analysis of the WHI data actually shows, how risk varies enormously depending on when you start HRT, what kind of estrogen you take, and how you deliver it. It also covers who truly should not take HRT. The goal is not to minimize risk — blood clots and strokes are real and can kill — but to give you numbers you can compare to the risks you already accept every day.
Table of Contents
- The 2002 WHI Story — and What Went Wrong With the Headlines
- The Timing Hypothesis: Age at Initiation Matters Enormously
- The Estrogen-Alone Surprise
- Blood Clots (VTE): Route of Delivery Changes Everything
- Stroke Risk: Oral Estrogen, Age, and Baseline Risk
- Which Progestogen? Micronized Progesterone vs MPA
- Absolute Contraindications — Who Should Not Take HRT
- Personalizing Your Risk: Gail, Family History, BRCA
- Putting HRT Risk in Everyday Terms
- Key Research Papers
- PubMed Topic Searches
- Connections
- Featured Videos
The 2002 WHI Story — and What Went Wrong With the Headlines
The Women's Health Initiative was the largest randomized trial ever run in menopausal women — more than 27,000 participants across two arms. The estrogen-plus-progestin arm used conjugated equine estrogens (CEE, Premarin) 0.625 mg daily plus medroxyprogesterone acetate (MPA, Provera) 2.5 mg daily. The average participant was 63 years old at enrollment — more than a decade past the average age of menopause.
On July 9, 2002, the data safety monitoring board stopped the trial after 5.2 years. The headline number: a hazard ratio of 1.26 for invasive breast cancer — a "26% increase." What that actually meant in absolute terms:
- Placebo group: about 30 invasive breast cancers per 10,000 women per year.
- CEE + MPA group: about 38 invasive breast cancers per 10,000 women per year.
- Excess risk: roughly 8 extra cases per 10,000 women-years, or 0.08% per year.
Stated that way, the number is real but small. For comparison: the excess breast-cancer risk from drinking one alcoholic drink per day is roughly 7–10 extra cases per 10,000 women-years. The excess risk from a BMI over 30 is larger still. The story that got told in 2002 — "HRT causes breast cancer" — conflated a modest absolute increase in one specific regimen (oral CEE + oral MPA, started at an average age of 63) with all hormone therapy in all women.
There were also increases in coronary heart disease events and stroke in that arm, plus increased VTE — all real, all in older women started late, all using oral conjugated equine estrogens and a synthetic progestin that modern prescribers rarely use anymore.
The Timing Hypothesis: Age at Initiation Matters Enormously
The long-term follow-up analysis published by JoAnn Manson and colleagues in JAMA in 2017 tracked WHI participants for 18 years. When results were broken down by age at initiation, the picture changed dramatically:
- Women who started HRT between ages 50 and 59 (the group most like real-world new initiators) showed no increase in all-cause mortality, and hints of reduced mortality on several endpoints.
- Women who started within 10 years of menopause onset showed a trend toward reduced coronary heart disease.
- The harms clustered in women who started after age 60 or more than a decade past menopause — precisely the women already accumulating atherosclerotic plaque, in whom introducing estrogen may destabilize existing lesions.
This is the "timing hypothesis" or "window of opportunity." It is now accepted by the North American Menopause Society (now The Menopause Society), the Endocrine Society, the International Menopause Society, and the British Menopause Society. Their consensus: for symptomatic women under 60 and within 10 years of menopause, the benefits of HRT generally outweigh the risks. After 60 or beyond 10 years, the calculation tilts less favorably, and the decision becomes much more individual.
The Estrogen-Alone Surprise
The WHI ran a second arm for women who had already had a hysterectomy, so they did not need a progestogen to protect the uterine lining. These 10,739 women received CEE 0.625 mg daily alone or placebo. The results, published and updated through 2020:
- Invasive breast cancer decreased in the estrogen-alone group: hazard ratio about 0.78, translating to roughly 7 fewer cases per 10,000 women-years.
- Breast cancer mortality was also reduced (hazard ratio around 0.60 in long-term follow-up).
This finding, almost never reported in the mainstream press, strongly suggests that the synthetic progestin MPA — not estrogen itself — drove most of the breast-cancer signal in the combined arm. Estrogen without MPA, in hysterectomy women, actually appears breast-protective.
Whether that protection extends to women who still have a uterus and must take some progestogen is the central question of modern HRT practice. The answer depends heavily on which progestogen you choose — see section 6 below.
Blood Clots (VTE): Route of Delivery Changes Everything
Venous thromboembolism (VTE) — deep vein thrombosis and pulmonary embolism — is the most clear-cut risk of systemic HRT. But it is almost entirely a risk of oral estrogen, not transdermal.
Here is why. When you swallow an estrogen pill, the hormone is absorbed through the gut and travels through the portal vein directly to the liver in high concentration. The liver responds by upregulating production of clotting factors (fibrinogen, factor VII, factor X) and down-regulating anticoagulants (antithrombin, protein S). A patch or gel delivers estrogen through the skin into the general circulation, so the liver only sees physiologic bloodstream concentrations — not a concentrated first-pass hit.
The ESTHER case-control study (Canonico et al., 2007) and subsequent meta-analyses found:
- Oral estrogen: odds ratio for VTE roughly 2.0 to 2.5 vs non-users. Baseline VTE rate in healthy postmenopausal women is about 1–2 per 1,000 per year, so oral estrogen adds roughly 1–2 extra VTE events per 1,000 women-years.
- Transdermal estradiol (patch or gel): odds ratio 1.0 to 1.1 — statistically indistinguishable from no HRT.
This is one of the clearest risk-reduction choices in hormone therapy. For any woman with even modestly elevated VTE risk — obesity, smoking, family history of clot, factor V Leiden, prior pregnancy clot, planned surgery — transdermal delivery is strongly preferred. Many clinicians now default to transdermal for everyone regardless of baseline risk.
Stroke Risk: Oral Estrogen, Age, and Baseline Risk
Stroke risk on HRT follows a similar pattern but is more sensitive to age and cardiovascular baseline. WHI found an increased ischemic stroke risk on oral CEE: roughly 8 extra strokes per 10,000 women-years, concentrated in women over 60 and those with untreated hypertension.
Observational data on transdermal estradiol suggest no meaningful increase in stroke at doses of 50 mcg/day or lower. Higher-dose patches (100 mcg) do appear to carry some excess risk, though the absolute numbers remain small in women under 60.
Practical implications:
- Control blood pressure before starting HRT. Uncontrolled hypertension is the single biggest amplifier of stroke risk on estrogen.
- Prefer transdermal over oral if you have migraine with aura, prior TIA, atrial fibrillation, or other stroke risk factors.
- After age 60 or 10 years post-menopause, the stroke calculus shifts — this is where the "window of opportunity" argument has real weight.
Which Progestogen? Micronized Progesterone vs MPA
If you still have a uterus, you need a progestogen alongside systemic estrogen to prevent endometrial hyperplasia and endometrial cancer. The choice of progestogen dramatically changes breast cancer risk.
The French E3N cohort (Fournier et al., 2008) followed 80,377 postmenopausal women for about 8 years and compared breast cancer incidence by HRT regimen:
- Estrogen + micronized progesterone (bio-identical, Prometrium): relative risk about 1.00 — essentially no increase in breast cancer over non-users during the study window.
- Estrogen + dydrogesterone: relative risk about 1.16 — a small increase.
- Estrogen + synthetic progestins (including MPA, norethisterone): relative risk about 1.69 — substantially higher.
Longer-term follow-up does show some signal with micronized progesterone after 5+ years of use, so the protection is not absolute — but the difference between bio-identical progesterone and MPA is large and consistent across cohorts. This is why essentially every modern menopause guideline now prefers micronized progesterone (or the progesterone-releasing intrauterine system) over older synthetic progestins for women starting HRT today. See the companion progesterone vs progestins article for dosing details.
Absolute Contraindications — Who Should Not Take HRT
These are the situations where systemic HRT is genuinely off-limits. Vaginal estrogen for GSM is a separate question with a different safety profile — see the GSM article.
- Active or recent venous thromboembolism (DVT or pulmonary embolism). A personal history of unprovoked VTE is a strong contraindication to oral estrogen; transdermal may be considered case-by-case with hematology input.
- Active breast cancer or a history of estrogen-receptor-positive breast cancer. Oncology generally advises against systemic HRT; non-hormonal options exist for hot flashes (see below).
- Unexplained vaginal bleeding. Must be worked up and the cause identified before any HRT — hormone therapy can mask endometrial cancer.
- Active liver disease with significantly abnormal liver function. Oral estrogen is metabolized hepatically; transdermal may be acceptable in stable mild disease, but check with hepatology.
- Recent myocardial infarction or stroke (within roughly 12 months).
- Known or suspected pregnancy.
- Estrogen-dependent tumors beyond breast (e.g., known endometrial cancer).
Relative contraindications — where a careful conversation with a knowledgeable menopause clinician is needed — include: migraine with aura, uncontrolled hypertension, gallbladder disease, familial hypertriglyceridemia, BRCA1/2 mutation, and dense breast tissue with strong family history.
Personalizing Your Risk: Gail, Family History, BRCA
The population-average numbers above become much more useful when you translate them to your specific risk profile. The two most common tools:
- Gail Model (NCI Breast Cancer Risk Assessment Tool). Free online. Uses age, race, age at menarche, age at first live birth, number of first-degree relatives with breast cancer, number of prior breast biopsies, and presence of atypia. Outputs a 5-year and lifetime breast cancer risk estimate. A 5-year risk above 1.67% is considered elevated — the threshold at which the FDA approved tamoxifen for prevention. If your baseline 5-year risk is 1.0%, adding the WHI excess (~0.08% per year, ~0.4% over 5 years on CEE+MPA) gives roughly 1.4%. Perspective matters.
- Tyrer-Cuzick (IBIS) Model. More sophisticated than Gail; incorporates breast density, BMI, and more detailed family history. Often preferred in high-risk clinics.
- BRCA1/2 carriers. Lifetime breast cancer risk is already 55–72% for BRCA1 and 45–69% for BRCA2. HRT is not absolutely contraindicated after risk-reducing bilateral salpingo-oophorectomy (prophylactic ovary removal), and multiple studies suggest short-term HRT to the natural menopause age (~51) does not erase the risk-reduction benefit of the surgery. This decision belongs with a high-risk breast specialist.
Personal density reports, prior biopsy history showing atypical hyperplasia or lobular carcinoma in situ (LCIS), and first-degree relatives with premenopausal breast cancer are all features that should shift the conversation toward caution or non-hormonal options.
Putting HRT Risk in Everyday Terms
Numbers in isolation are meaningless. Consider what other risks look like per 10,000 women-years:
- CEE + MPA (WHI): ~8 extra breast cancers, ~7 extra coronary events, ~8 extra strokes, ~18 extra VTE events — offset by ~6 fewer colorectal cancers and ~5 fewer hip fractures. Net excess events roughly 30 per 10,000 per year.
- Transdermal estradiol + micronized progesterone (modern regimen in women under 60): breast-cancer signal at or near background, no excess VTE, no meaningful stroke excess at 50 mcg dosing. Benefits on bone, vasomotor symptoms, and possibly cardiovascular mortality (per timing hypothesis) remain.
- Obesity (BMI ≥ 30): adds roughly 20–30 extra breast cancers per 10,000 postmenopausal women-years, plus large excesses in cardiovascular and diabetes-related events.
- One to two alcoholic drinks per day: adds 7–10 extra breast cancers per 10,000 women-years.
- Driving a car in the United States: roughly 1 fatal motor vehicle crash per 10,000 drivers per year.
- Combined oral contraceptives in a 30-year-old: VTE rate roughly 3–9 per 10,000 women-years above baseline — comparable to or higher than oral postmenopausal HRT.
This is the context the 2002 headlines left out. HRT risk is real and deserves informed consent. It is not categorically different from — and in many configurations is smaller than — risks women routinely accept with a glass of wine, a second helping at dinner, or a commute to work. The Menopause Society's 2022 position statement makes this point in clinical language: for symptomatic women under 60 and within 10 years of menopause, "the benefit-risk ratio is favorable for the treatment of bothersome vasomotor symptoms and for women at elevated risk of bone loss or fracture."
Key Research Papers
- Rossouw JE, et al. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the Women's Health Initiative randomized controlled trial. JAMA. 2002;288(3):321-333.
- Manson JE, et al. Menopausal hormone therapy and long-term all-cause and cause-specific mortality: the Women's Health Initiative randomized trials. JAMA. 2017;318(10):927-938.
- Canonico M, et al. Hormone therapy and venous thromboembolism among postmenopausal women: impact of the route of estrogen administration and progestogens: the ESTHER study. Circulation. 2007;115(7):840-845.
- Fournier A, Berrino F, Clavel-Chapelon F. Unequal risks for breast cancer associated with different hormone replacement therapies: results from the E3N cohort study. Breast Cancer Res Treat. 2008;107(1):103-111.
- The 2022 Hormone Therapy Position Statement of The North American Menopause Society. Menopause. 2022;29(7):767-794.
- Chlebowski RT, et al. Association of menopausal hormone therapy with breast cancer incidence and mortality during long-term follow-up of the Women's Health Initiative randomized clinical trials. JAMA. 2020;324(4):369-380.
PubMed Topic Searches
For further reading, these PubMed searches pull up current peer-reviewed work on HRT risk:
- WHI hormone therapy long-term follow-up
- Transdermal estradiol and venous thromboembolism
- Micronized progesterone and breast cancer risk
- HRT timing hypothesis and cardiovascular outcomes
- Estrogen-alone and breast cancer in hysterectomy women
- Hormone therapy and stroke risk by route
- E3N cohort, HRT, and breast cancer
- BRCA carriers, HRT, and risk-reducing oophorectomy
- The Menopause Society 2022 position statement
- Absolute vs relative risk communication in HRT
Connections
- Genitourinary Syndrome of Menopause and Vaginal Estrogen
- Micronized Progesterone vs Synthetic Progestins
- Estradiol Formulations: Patch, Gel, Oral Routes
- Testosterone Therapy for Women
- Vasomotor Symptoms: Hot Flashes and Night Sweats
- Non-Hormonal Options: SSRIs, Gabapentin, Fezolinetant
- Perimenopause Symptom Tracker and Hormonal Testing
- Bone Loss Prevention and HRT
- Perimenopause
- Cardiovascular Disease
- Menopause and HRT
- Hormone Panel
- Stroke
- Deep Vein Thrombosis
- Pulmonary Embolism
- Hypertension
- Depression
- Insomnia