Genitourinary Syndrome of Menopause & Vaginal Estrogen
If sex hurts, if you are peeing every two hours, if you keep getting UTIs, if you feel dry and raw and nobody in medicine seems to take it seriously — this page is for you. Genitourinary Syndrome of Menopause (GSM) is the most under-treated, over-tolerated problem in women's health. It is common, it is progressive, and it is fixable. The treatment is cheap, remarkably safe, and in most cases transforms symptoms within weeks. Primary care misses it constantly. This article explains what GSM is, the specific products and doses that work, the cost and insurance realities, and the nuances — including the breast cancer conversation — that shape the decision.
Table of Contents
- From "Vaginal Atrophy" to GSM — Why the Name Changed
- Prevalence and the Progressive Nature of GSM
- Symptoms — The Full List No One Tells You
- What the Exam Shows
- Vaginal Estrogen Formulations and Doses
- Non-Estrogen Prescription Options: DHEA and Ospemifene
- Safety, Breast Cancer, and the Aromatase Inhibitor Nuance
- How Long to Use It (Spoiler: Indefinitely)
- Cost, Insurance, and How to Actually Get It
- Non-Hormonal Moisturizers, Lubricants, and Pelvic Floor PT
- Laser and Radiofrequency Devices — The Evidence
- Why Primary Care Keeps Missing This
- Key Research Papers
- PubMed Topic Searches
- Connections
- Featured Videos
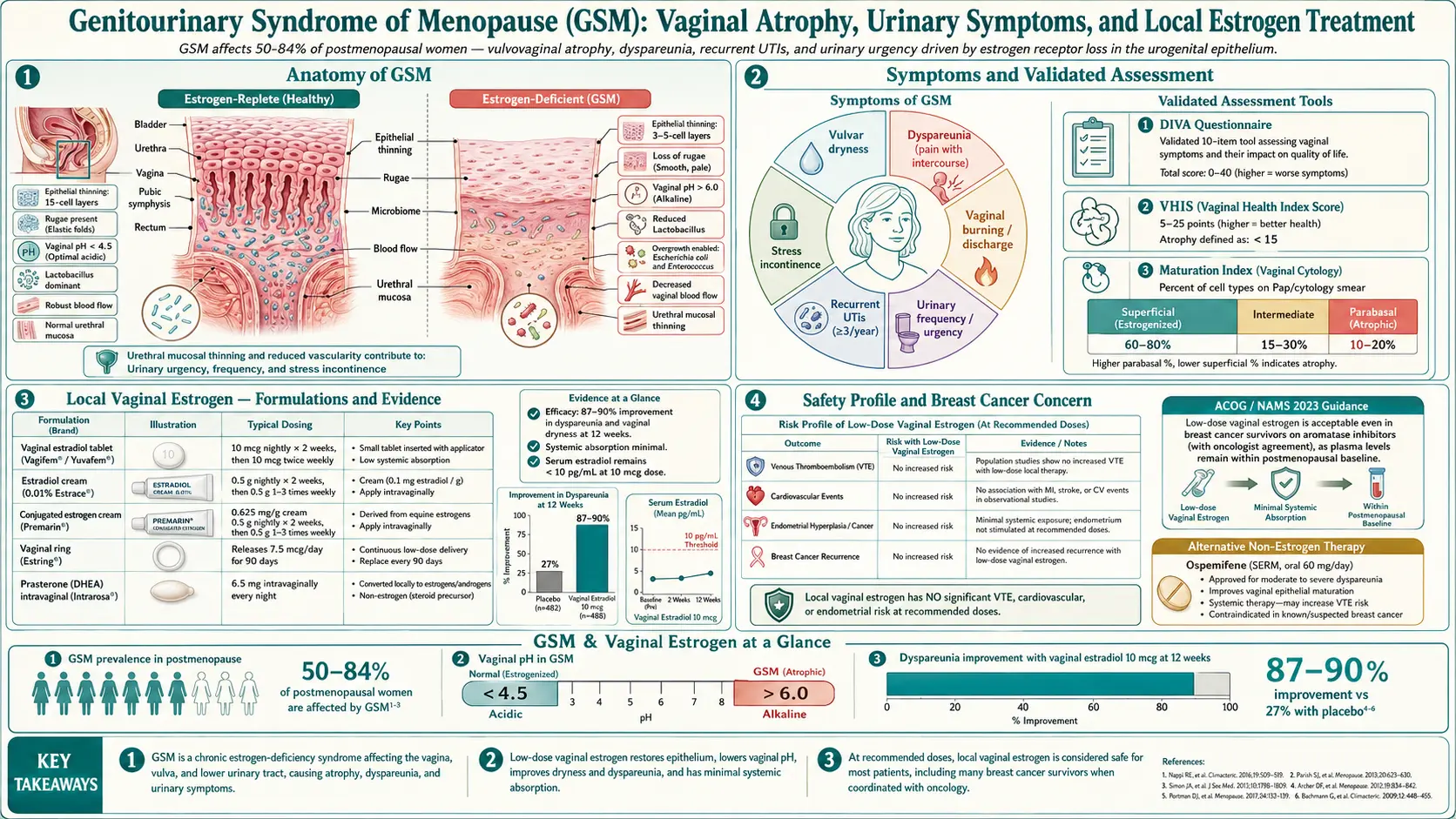
From "Vaginal Atrophy" to GSM — Why the Name Changed
For decades the textbook term was vulvovaginal atrophy (VVA) or "atrophic vaginitis." Both names were terrible. "Atrophy" sounds like shriveling; "vaginitis" implies infection. Neither captured the urinary half of the problem, and both were embarrassing enough to make patients and doctors sidestep the conversation.
In 2014, a joint consensus conference of the International Society for the Study of Women's Sexual Health (ISSWSH) and The North American Menopause Society (NAMS) published a new umbrella term: Genitourinary Syndrome of Menopause (GSM). The name reflects biology — estrogen receptors are densely packed not just in the vagina but in the vulva, urethra, bladder trigone, and pelvic floor. When estrogen drops, all of those tissues thin, lose collagen, lose blood flow, and change pH. Calling the problem "vaginitis" hid most of it.
The shift matters practically. If you describe "burning with urination" and "having to go every hour" to your doctor, you may be sent home with antibiotics and a clean urinalysis for the fifth time. If you describe the same symptoms as GSM, a menopause-literate clinician recognizes the pattern immediately and treats the estrogen deficit, not an imaginary infection.
Prevalence and the Progressive Nature of GSM
Studies put GSM prevalence at roughly 50–80% of postmenopausal women. The range is wide because women under-report. In symptom surveys that name the specific problems (dryness, painful sex, urinary urgency), rates cluster near the top of that range.
Two critical differences from hot flashes (vasomotor symptoms, VMS):
- GSM is progressive. Without treatment, symptoms get worse over time, not better. Year five postmenopause is usually worse than year one.
- GSM does not "burn out." Hot flashes typically wane after 7–10 years (though a substantial minority of women have VMS for life). GSM does not fade with time. An 80-year-old has more atrophy than she did at 55.
Translation: if you have GSM and do nothing, you will be worse in five years than you are now. This is the single most important message primary care fails to deliver.
Symptoms — The Full List No One Tells You
GSM is not just "dryness." Patients are often relieved to see the whole picture written down:
Vulvovaginal symptoms:
- Dryness, even when aroused.
- Dyspareunia — pain with penetrative sex, ranging from a raw sandpaper sensation to sharp tearing.
- Itching or burning of the vulva, sometimes mistaken for chronic yeast infection.
- Loss of labial fullness; the labia minora may shrink or fuse.
- Narrowing and shortening of the vaginal canal.
- Spotting or bleeding after intercourse (postcoital bleeding). Always worth mentioning to a clinician the first time, to rule out other causes, but the most common reason in postmenopausal women is friable atrophic tissue.
- Reduced lubrication, loss of arousal, decreased orgasm intensity.
Urinary symptoms:
- Urgency — the sudden, can't-wait feeling.
- Frequency — voiding more than every 2 hours in the day.
- Dysuria — burning with urination, often with a sterile urine culture.
- Nocturia — waking more than once a night to pee.
- Recurrent urinary tract infections, especially the pattern of true UTIs two to four times a year.
- Stress incontinence that worsens postmenopause.
Any combination of these, especially the recurrent-UTI pattern in a postmenopausal woman, should trigger a GSM evaluation before another round of antibiotics.
What the Exam Shows
A clinician trained to look for GSM will see a combination of:
- Pale, thin vaginal walls with loss of the normal ridges (rugae).
- Petechiae or small abrasions from minor friction.
- A vaginal pH above 5.0 (normal premenopausal is 3.5–4.5). A simple pH strip at the bedside is diagnostic.
- Shortened vaginal length, narrowed introitus.
- Labial involution.
- Urethral caruncle or prolapsed, red urethral mucosa at the meatus.
You do not need all of these findings to have GSM. Symptoms alone — especially dyspareunia plus an elevated pH — are enough to start treatment.
Vaginal Estrogen Formulations and Doses
Vaginal estrogen is the gold standard treatment. It works because it restores estrogen receptors in the exact tissue that is symptomatic, with almost no drug reaching the bloodstream. Multiple formulations exist — any of them can work, and preference is usually about delivery method and cost.
Estradiol Cream (Estrace)
- Dose: 0.5–1 g intravaginally. Loading phase: nightly for 2 weeks. Maintenance: 2–3 nights per week.
- Pros: Cream lets you spread a small amount on the vulva and urethra too — helpful for burning and urinary symptoms.
- Cost: Generic estradiol cream runs $50–$200 per 42.5 g tube, which lasts many weeks at maintenance doses. GoodRx and manufacturer coupons often bring it to $30–$60.
Conjugated Equine Estrogen Cream (Premarin)
- Dose: 0.5–1 g intravaginally, same 2-week load then 2–3 times weekly.
- Note: Derived from pregnant mare urine; effective but usually more expensive than generic estradiol. Many clinicians default to Estrace.
Estradiol Vaginal Tablet (Vagifem / Yuvafem) and Softgel Insert (Imvexxy)
- Dose: 10 mcg tablet or insert, nightly for 2 weeks, then twice weekly.
- Pros: Pre-measured, no drip, no mess. The single most popular option for women who dislike cream.
- Cost: Brand Vagifem runs $200–$350 per 24-pack; generic estradiol tablets (Yuvafem) are often $50–$120 with coupons. Imvexxy is usually brand-priced.
Estradiol Vaginal Ring (Estring)
- Dose: One soft silicone ring inserted like a diaphragm and left in place for 90 days, then replaced.
- Pros: Absolutely the lowest-effort option — four insertions a year. Not felt during sex by most couples (and easy to remove temporarily). Excellent for women with dexterity or memory issues.
- Cost: $300–$600 per ring retail; typically one of the better-covered products on insurance because cost-per-month is modest.
- Note: Do not confuse with Femring, which is a high-dose systemic estradiol ring used for VMS and does require progestogen if you have a uterus. Estring is the low-dose local ring.
Non-Estrogen Prescription Options: DHEA and Ospemifene
Prasterone / DHEA Vaginal Insert (Intrarosa)
- Dose: 6.5 mg vaginal insert, once nightly (no maintenance step-down).
- Mechanism: DHEA is converted locally inside vaginal cells into both estrogen and testosterone. Serum hormone levels remain in the postmenopausal range.
- When useful: Women who do not want an estrogen label on their prescription, or who find that estrogen alone does not fully restore comfort (the local testosterone contribution seems to help some patients).
- Cost: $250–$500 per month retail; manufacturer copay cards can reduce this substantially. Insurance coverage varies.
Ospemifene (Osphena) — Oral SERM
- Dose: 60 mg orally once daily with food.
- Mechanism: Selective estrogen receptor modulator — acts like an estrogen on vaginal tissue but behaves differently at breast and other sites.
- Indication: Moderate to severe dyspareunia from GSM, in women who cannot or will not use vaginal products.
- Trade-offs: Oral route means measurable systemic exposure. Small increase in VTE (blood clot) risk. Some women get hot flashes on it. Not first-line when a vaginal option is acceptable.
- Cost: Roughly $200–$450 per month retail.
Safety, Breast Cancer, and the Aromatase Inhibitor Nuance
The systemic absorption from low-dose vaginal estrogen is extremely small. Across every formulation at standard maintenance doses, serum estradiol stays within the normal postmenopausal range. For this reason:
- Vaginal estrogen does NOT require progestogen coverage in women with a uterus, at standard low doses. This is a formal position of NAMS and ACOG. If you hear otherwise, the clinician is extrapolating inappropriately from systemic HRT guidance.
- There is no measured increase in breast cancer risk in observational data, including large analyses of the Women's Health Initiative observational cohort (Crandall 2018).
- There is no measured increase in endometrial cancer, VTE, stroke, or cardiovascular events at standard doses.
For women with a history of breast cancer, the picture is more nuanced but still reassuring:
- ACOG and NAMS both endorse vaginal estrogen as an option for breast cancer survivors with bothersome GSM that has not responded to non-hormonal measures — after a conversation with their oncologist.
- For women on tamoxifen, vaginal estrogen is generally considered compatible (tamoxifen blocks any estrogen effect at breast tissue).
- The genuinely debated group is women on aromatase inhibitors (anastrozole, letrozole, exemestane). These drugs depend on keeping systemic estradiol as close to zero as possible. Even tiny bumps from vaginal estrogen are a theoretical concern. The data we have (including the low-dose ring and tablet) show minimal serum rise, but the decision is individualized. Many oncologists now accept low-dose vaginal estrogen for AI patients with severe GSM; others prefer a non-hormonal or DHEA trial first. This is a conversation, not a blanket rule.
How Long to Use It (Spoiler: Indefinitely)
Because GSM is progressive and estrogen deficiency never reverses on its own, treatment is typically lifelong. Symptoms return within weeks to months of stopping. This is not a failure — it is how the biology works. Using a vaginal insert twice a week at age 75 is as appropriate as it was at age 55.
There is no cumulative-dose ceiling, no "I should take a break" rule, and no expiration on benefit. The safety data out to 10+ years remain reassuring.
Cost, Insurance, and How to Actually Get It
Cost is the single biggest barrier after clinician ignorance. Practical tactics:
- Use GoodRx or SingleCare for generic estradiol cream and generic estradiol tablets. The cash discount often beats the insurance copay.
- Ask for generic first. Brand Vagifem and Estrace are unnecessary when generic estradiol cream or generic vaginal tablets work identically.
- Manufacturer copay cards exist for Imvexxy, Intrarosa, Osphena, and Estring. Search the brand name + "savings card."
- Appeal denials. If insurance rejects the ring, your clinician can document that cream/tablets were tried first or contraindicated. Most denials flip on appeal.
- Compounding pharmacies offer estradiol creams at sometimes lower cost, but quality and dose consistency are variable. FDA-approved products are preferred when affordable.
- Telehealth menopause clinics (Midi, Alloy, Evernow, Winona, and others) can prescribe vaginal estrogen in most U.S. states for a flat consult fee, which is often the fastest route if your primary care is unhelpful.
Non-Hormonal Moisturizers, Lubricants, and Pelvic Floor PT
These are worth using even on vaginal estrogen, and are the mainstay for women who cannot use hormones.
Vaginal Moisturizers (Regular Use)
- Hyaluronic acid suppositories — the best of the non-hormonal options. Revaree (Bonafide) inserts one suppository every 2–3 days. Studies show meaningful symptom improvement. ~$55 for a month.
- Replens — a bioadhesive polycarbophil gel, one applicator every 2–3 days. $15–$25 for a month's supply.
- Coconut oil — inexpensive short-term moisturizer applied externally to the vulva. Fine for occasional use; not a substitute for a vaginal moisturizer for internal symptoms, and it can degrade latex condoms.
Lubricants (Used During Sex)
Separate from moisturizers. Use generously.
- Water-based (Slippery Stuff, Sliquid H2O) — condom-safe, washes off. Can dry out mid-session; reapply.
- Silicone-based (Uberlube, Pjur) — longer-lasting, excellent for dyspareunia. Do not use with silicone toys.
- Avoid glycerin-heavy lubes (can worsen yeast) and warming/flavored products (irritants).
Pelvic Floor Physical Therapy
A massively under-used resource. A pelvic floor PT can address the hypertonic (tight, guarded) pelvic floor that develops after months of painful sex, treat urinary urgency, and guide vaginal dilator use to rebuild a narrowed canal. Insurance usually covers it under a standard PT benefit. One or two visits plus a home program often produces outsized gains on top of vaginal estrogen.
Laser and Radiofrequency Devices — The Evidence
Fractional CO2 lasers (MonaLisa Touch, FemiLift) and radiofrequency devices (ThermiVa) have been heavily marketed as "non-hormonal GSM fixes." The short version:
- Small uncontrolled studies show symptom improvement.
- The best-designed randomized sham-controlled trials (e.g., the 2021 VeLVET and Li trials of fractional CO2 laser) show no significant difference from sham treatment at 6 months.
- The FDA issued a warning in 2018 about the marketing of "vaginal rejuvenation" devices for GSM, noting safety concerns and lack of robust evidence.
- Cost is $1,500–$3,000 for a three-session course, rarely covered by insurance.
Laser may eventually find a legitimate niche, particularly for breast cancer survivors who cannot use any hormone. For most women with GSM, spending that money on five years of vaginal estrogen is a dramatically better investment.
Why Primary Care Keeps Missing This
You are not imagining it. The pattern is real and has specific causes:
- Training gap. The average U.S. medical resident receives less than one hour of menopause education. Most graduating OB/GYNs and family physicians have never been shown how to counsel on vaginal estrogen.
- The WHI misread. The 2002 Women's Health Initiative headlines scared a generation of clinicians off all estrogen, including local low-dose vaginal estrogen that has nothing to do with WHI findings. Many clinicians still conflate the two.
- The FDA black-box label. Every vaginal estrogen product carries a class-wide boxed warning copied from systemic HRT, despite the evidence showing local therapy has none of those risks. Patients see the label and assume the worst.
- Time and discomfort. GSM requires a pelvic exam, a frank sexual history, and a 10-minute counseling conversation — not possible in a 15-minute visit.
- UTI reflex. Recurrent urinary symptoms get treated as bacterial infections even when cultures are negative, because antibiotics are a faster disposition than a menopause workup.
If your primary care cannot or will not treat GSM, you have options: a menopause-certified clinician (NAMS certification "MSCP" or "NCMP"), a telehealth menopause service, or a urogynecologist. Do not accept untreated GSM as normal aging.
Key Research Papers
- Portman DJ, Gass ML. Genitourinary syndrome of menopause: new terminology for vulvovaginal atrophy from ISSWSH and NAMS. Menopause. 2014;21(10):1063–1068. — the naming consensus paper.
- The 2020 genitourinary syndrome of menopause position statement of The North American Menopause Society. Menopause. 2020;27(9):976–992.
- Crandall CJ, et al. Breast cancer, endometrial cancer, and cardiovascular events in participants who used vaginal estrogen in the Women's Health Initiative Observational Study. Menopause. 2018;25(1):11–20. — the large reassuring safety cohort.
- Mitchell CM, et al. Efficacy of vaginal estradiol or vaginal moisturizer vs placebo for treating postmenopausal vulvovaginal symptoms: a randomized clinical trial. JAMA Intern Med. 2018.
- Li FG, et al. Effect of fractional carbon dioxide laser vs sham treatment on symptom severity in women with postmenopausal vaginal symptoms: a randomized clinical trial. JAMA. 2021;326(14):1381–1389. — the sham-controlled laser trial.
- ACOG Committee Opinion No. 659: The use of vaginal estrogen in women with a history of estrogen-dependent breast cancer. Obstet Gynecol. 2016;127(3):e93–e96.
PubMed Topic Searches
For current peer-reviewed literature:
- Genitourinary syndrome of menopause
- Vaginal estrogen safety and breast cancer
- Low-dose vaginal estradiol and systemic absorption
- Prasterone (DHEA) and dyspareunia
- Ospemifene for vulvovaginal atrophy
- Recurrent UTI and vaginal estrogen
- Fractional CO2 laser for vaginal atrophy (randomized trials)
- Hyaluronic acid vaginal moisturizers
- Aromatase inhibitors and vaginal estrogen
- Pelvic floor PT for dyspareunia
Connections
- Vasomotor Symptoms: Hot Flashes and Night Sweats
- Micronized Progesterone vs Synthetic Progestins
- Estradiol Formulations: Patch, Gel, Oral Routes
- HRT Risks: Breast Cancer, Clots and Stroke
- Testosterone Therapy for Women
- Non-Hormonal Options: SSRIs, Gabapentin, Fezolinetant
- Perimenopause Symptom Tracker and Hormonal Testing
- Bone Loss Prevention and HRT
- Perimenopause
- Menopause and HRT
- Hormone Panel
- DHEA-S
- Testosterone
- Urinary Tract Infections
- Depression
- Insomnia
- Cardiovascular Disease
- Osteoporosis