Hormone Panel
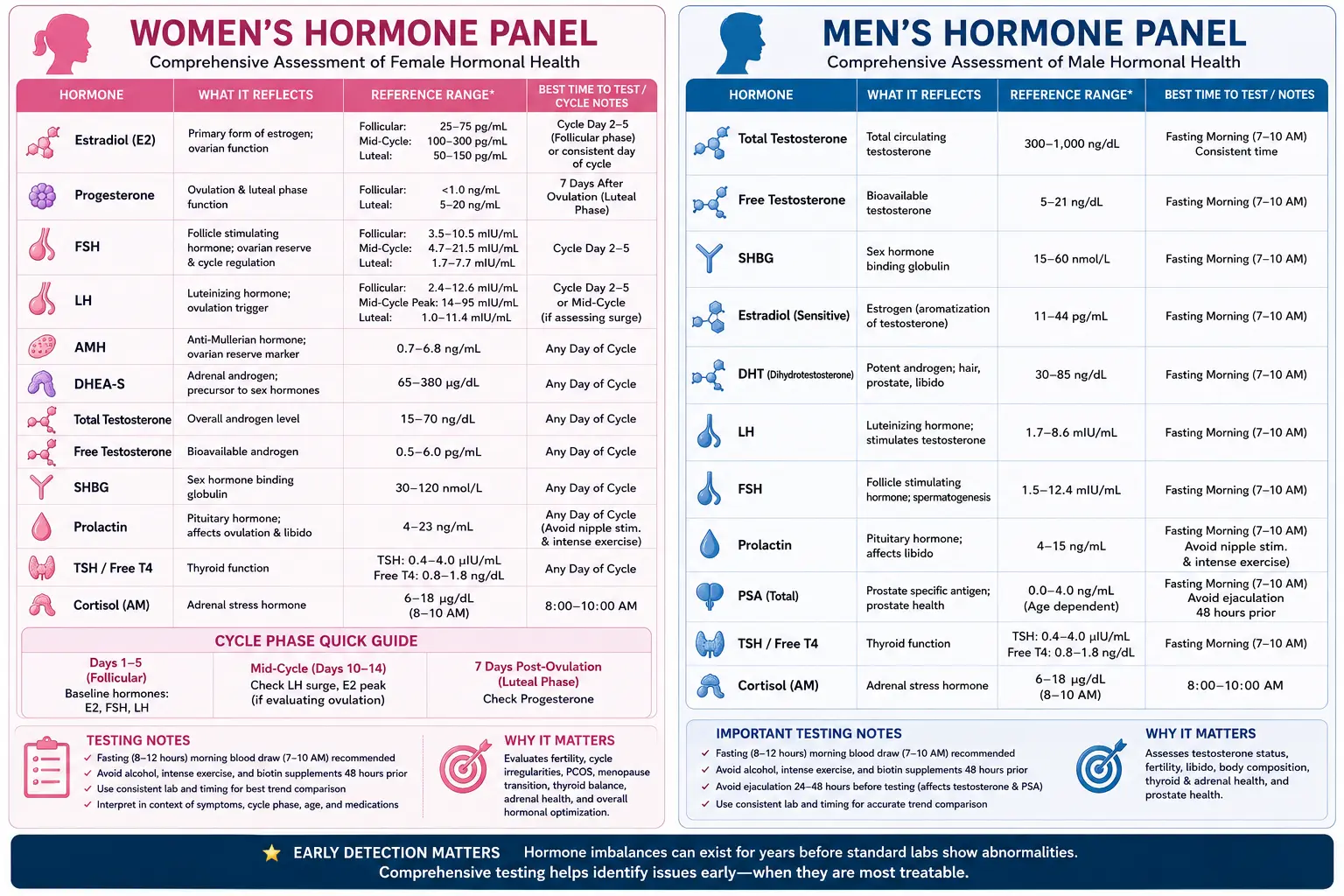
Table of Contents
- Why Comprehensive Hormone Testing Matters
- Cortisol: The Stress Hormone
- DHEA-S: The Youth Hormone
- Testosterone: Total and Free
- Estradiol (E2) and Estrogen Metabolites
- Progesterone and Luteal Phase Testing
- Estrogen-to-Progesterone Ratio and Estrogen Dominance
- FSH and LH: Fertility and Menopause Markers
- Fasting Insulin as a Metabolic Health Marker
- IGF-1: Growth Hormone Marker
- Prolactin
- When to Test Hormones
- Symptoms of Hormonal Imbalance
- Natural Hormone Support Strategies
- Bioidentical vs. Synthetic Hormones
- Cautions and Considerations
- Featured Videos
Why Comprehensive Hormone Testing Matters
Hormones are the chemical messengers that govern virtually every physiological process in the body, from metabolism and energy production to mood, sleep, reproduction, and tissue repair. In functional medicine, we recognize that hormonal imbalances rarely occur in isolation. A disruption in one hormonal pathway almost always affects others, creating a cascade of symptoms that can be difficult to untangle without thorough laboratory evaluation.
Standard conventional panels often check only one or two hormones in isolation, such as TSH for thyroid function or total testosterone for male hormone status. This narrow approach frequently misses subclinical imbalances, fails to evaluate hormone metabolites, and overlooks the critical interplay between the adrenal, thyroid, and gonadal axes. A comprehensive hormone panel provides a systems-level view of endocrine function, enabling practitioners to identify root causes rather than merely managing symptoms.
Comprehensive hormone testing is especially valuable for patients experiencing:
- Persistent fatigue despite adequate sleep
- Unexplained weight gain or difficulty losing weight
- Mood disturbances including anxiety, depression, and irritability
- Low libido or sexual dysfunction
- Menstrual irregularities, PMS, or perimenopausal symptoms
- Hair loss, acne, or other androgen-related skin changes
- Insomnia or disrupted sleep architecture
- Brain fog, poor concentration, and memory difficulties
- Muscle wasting or inability to build lean mass
- Bone density loss or early osteoporosis markers
By evaluating the full hormonal landscape, we can design targeted, individualized treatment protocols that address the underlying dysfunction rather than simply replacing a single hormone.
Cortisol: The Stress Hormone
Cortisol, produced by the adrenal glands under direction of the hypothalamic-pituitary-adrenal (HPA) axis, is arguably the most important hormone to evaluate in any comprehensive panel. Often called the "stress hormone," cortisol influences blood sugar regulation, immune function, inflammatory response, blood pressure, and the sleep-wake cycle. Chronic stress — whether physical, emotional, or environmental — can profoundly dysregulate cortisol production over time.
Morning Cortisol (Serum)
A single fasting morning cortisol drawn between 7:00 and 9:00 AM provides a snapshot of peak cortisol output. Optimal functional ranges are typically between 10 and 18 mcg/dL. Values below 10 may suggest adrenal insufficiency or HPA axis suppression, while values above 20 may indicate a heightened stress response or Cushing-like physiology. However, a single morning draw has significant limitations because cortisol naturally fluctuates throughout the day.
Four-Point Salivary Cortisol
The four-point salivary cortisol test is far more informative than a single serum draw. Saliva samples are collected at four intervals throughout the day — morning upon waking, late morning, afternoon, and evening before bed. This maps the diurnal cortisol curve, which should show a robust morning peak followed by a gradual decline throughout the day, reaching its lowest point at night. Common dysfunctional patterns include:
- Flat curve (low throughout the day): Often seen in advanced HPA axis dysfunction, sometimes referred to as "adrenal fatigue" or burnout
- Inverted curve (low morning, high evening): Associated with insomnia, nighttime anxiety, and disrupted circadian rhythm
- Elevated throughout the day: Indicative of chronic unresolved stress or high-cortisol states
- Blunted morning rise with afternoon crash: Common in individuals with poor sleep quality or chronic fatigue
DUTCH Test (Dried Urine Test for Comprehensive Hormones)
The DUTCH test represents the most advanced method for evaluating cortisol. It measures not only free cortisol at multiple time points but also cortisol metabolites (tetrahydrocortisol, tetrahydrocortisone), the cortisol-to-cortisone ratio (reflecting 11-beta-HSD enzyme activity), and cortisone levels. This allows practitioners to distinguish between true low cortisol production and excessive cortisol clearance — a distinction that fundamentally changes the treatment approach. The DUTCH test also includes melatonin metabolites, providing additional insight into circadian function.
HPA Axis Dysfunction
The term "adrenal fatigue," while widely used in integrative medicine, is more accurately described as HPA axis dysfunction. The adrenal glands themselves rarely fail; instead, the signaling between the hypothalamus, pituitary, and adrenals becomes dysregulated after prolonged stress exposure. This distinction matters because treatment must address the brain-adrenal communication pathway, not simply support the adrenal glands in isolation. Effective interventions include stress reduction techniques, sleep optimization, blood sugar stabilization, and targeted adaptogenic herbs.
DHEA-S: The Youth Hormone
Dehydroepiandrosterone sulfate (DHEA-S) is the most abundant steroid hormone in the human body and serves as a precursor to both testosterone and estrogen. Often called the "youth hormone," DHEA-S peaks in the mid-twenties and gradually declines with age, dropping by approximately 2% per year after age 30. By age 70, most individuals have only 10-20% of their peak DHEA-S levels remaining.
DHEA-S is a valuable marker of adrenal reserve and overall vitality. In functional medicine, we consider it an important biomarker of biological aging. Low DHEA-S levels are associated with:
- Chronic fatigue and reduced stress resilience
- Depression and cognitive decline
- Decreased immune function
- Reduced bone density
- Loss of lean muscle mass
- Decreased libido
- Accelerated aging
Optimal functional ranges for DHEA-S vary by age and sex. For women aged 30-50, optimal levels are generally between 150 and 350 mcg/dL. For men in the same age range, optimal levels are typically between 250 and 500 mcg/dL. When DHEA-S is chronically low alongside disrupted cortisol patterns, it suggests that the adrenal glands have shifted their pregnenolone "steal" toward cortisol production at the expense of DHEA — a hallmark of prolonged stress.
DHEA-S supplementation should be approached carefully and only under practitioner guidance, as it can convert to androgens and estrogens in ways that are not always predictable. Monitoring downstream hormone levels is essential when supplementing.
Testosterone: Total and Free
Testosterone is critical for both men and women, though it is often overlooked in female patients. In men, testosterone supports muscle mass, bone density, red blood cell production, libido, mood stability, and cognitive function. In women, testosterone plays an essential role in libido, energy, muscle tone, bone health, and overall sense of well-being — albeit at much lower circulating levels than in men.
Total Testosterone
Total testosterone measures all circulating testosterone, including the fraction bound to sex hormone-binding globulin (SHBG) and albumin, as well as the small percentage that circulates freely. Conventional lab ranges are notoriously broad. For men, a "normal" range might span from 250 to 1100 ng/dL, yet a man at 280 ng/dL may be profoundly symptomatic. Functional optimal ranges for men are typically 500-900 ng/dL. For premenopausal women, optimal total testosterone is generally between 40 and 70 ng/dL.
Free Testosterone
Free testosterone — the unbound, bioavailable fraction — is arguably more clinically relevant than total testosterone. Only about 1-3% of total testosterone circulates in the free form. A patient may have a "normal" total testosterone level but a low free testosterone due to elevated SHBG, which effectively binds testosterone and renders it unavailable to tissues.
The SHBG Factor
Sex hormone-binding globulin (SHBG) is a protein produced primarily by the liver that binds testosterone, estradiol, and other sex steroids, reducing their bioavailability. Elevated SHBG is commonly caused by:
- Oral contraceptive use (one of the most potent SHBG elevators)
- Hyperthyroidism
- Liver disease
- Aging
- Low-calorie diets and excessive exercise
Conversely, low SHBG is associated with insulin resistance, obesity, hypothyroidism, and polycystic ovary syndrome (PCOS). Understanding SHBG levels is essential for interpreting total testosterone and estradiol values accurately. Without measuring SHBG, clinicians risk missing the full picture of hormone bioavailability.
Estradiol (E2) and Estrogen Metabolites
Estradiol (E2) is the most potent and prevalent estrogen in premenopausal women. It is essential for bone health, cardiovascular protection, brain function, skin elasticity, and reproductive health. In men, small amounts of estradiol are necessary for bone density and brain health, but excess estradiol (often from aromatization of testosterone in adipose tissue) can contribute to gynecomastia, water retention, mood disturbances, and increased cardiovascular risk.
Conventional testing typically measures only total serum estradiol, but this provides an incomplete picture. Estrogen metabolism follows three primary pathways, each producing distinct metabolites with different biological activities:
- 2-hydroxyestrone (2-OH-E1): Generally considered the "protective" metabolite, associated with lower cancer risk and anti-proliferative effects
- 4-hydroxyestrone (4-OH-E1): A potentially genotoxic metabolite that can form DNA-damaging quinones if not properly detoxified through methylation
- 16-alpha-hydroxyestrone (16-alpha-OH-E1): A proliferative metabolite that, when elevated relative to 2-OH-E1, may increase the risk of estrogen-sensitive conditions
The DUTCH test is particularly valuable for evaluating estrogen metabolites and the methylation of 4-OH-E1 to 4-methoxy-E1. Poor methylation of the 4-OH pathway is a clinically actionable finding that can be addressed with targeted nutritional support, including methylated B vitamins, magnesium, and cruciferous vegetable compounds such as DIM (diindolylmethane) and sulforaphane.
For premenopausal women, estradiol should ideally be tested during the follicular phase (day 3-5 of the menstrual cycle) for baseline evaluation, or during the mid-luteal phase (day 19-22) when assessing it in relation to progesterone.
Progesterone and Luteal Phase Testing
Progesterone is a calming, neuroprotective hormone that is critical for fertility, pregnancy maintenance, mood stability, sleep quality, and bone health. It serves as a natural counterbalance to estrogen, and its decline is one of the earliest hormonal changes in perimenopause — often beginning years before estrogen levels fall.
In cycling women, progesterone is produced primarily by the corpus luteum after ovulation. It rises sharply in the second half of the menstrual cycle (the luteal phase) and drops just before menstruation. For this reason, progesterone must be tested at the correct time to be meaningful:
- Serum progesterone: Ideally drawn 5-7 days after confirmed ovulation, typically around day 19-22 of a 28-day cycle. Optimal mid-luteal progesterone is generally above 10-15 ng/mL for fertility and above 8 ng/mL for general well-being.
- DUTCH test progesterone metabolites: Measures pregnanediol, the primary progesterone metabolite in urine, providing insight into total progesterone production over the testing period rather than a single-point-in-time snapshot.
Low progesterone is extremely common and is associated with:
- PMS (especially anxiety, irritability, and breast tenderness)
- Luteal phase deficiency and difficulty conceiving
- Recurrent early miscarriage
- Insomnia and disrupted sleep (progesterone enhances GABA receptor activity)
- Heavy or irregular menstrual bleeding
- Perimenopausal symptoms including hot flashes and night sweats
Anovulatory cycles — cycles in which ovulation does not occur — result in minimal progesterone production despite apparently normal menstrual bleeding. These cycles become increasingly common in the years leading up to menopause and are a major contributor to estrogen dominance symptoms.
Estrogen-to-Progesterone Ratio and Estrogen Dominance
The concept of estrogen dominance, originally described by Dr. John Lee, refers to a state in which estrogen activity is excessive relative to progesterone — regardless of whether estrogen levels are absolutely elevated. A woman can have low estrogen levels and still be estrogen dominant if her progesterone levels are even lower. This relative imbalance is one of the most common hormonal patterns encountered in clinical practice.
The estrogen-to-progesterone ratio is calculated by comparing estradiol (in pg/mL) to progesterone (in ng/mL) during the mid-luteal phase. In saliva testing, the optimal progesterone-to-estradiol ratio is approximately 200:1 to 300:1. An imbalanced ratio — even with individually "normal" hormone levels — can produce significant symptoms.
Factors that contribute to estrogen dominance include:
- Anovulatory cycles: No ovulation means no corpus luteum and negligible progesterone production
- Chronic stress: Cortisol production can "steal" pregnenolone from the progesterone pathway
- Xenoestrogen exposure: Environmental chemicals (BPA, phthalates, pesticides, parabens) that mimic estrogen activity
- Poor estrogen detoxification: Sluggish liver phase I/II detoxification or impaired gut elimination (constipation, dysbiosis) allowing estrogen recirculation via beta-glucuronidase activity
- Excess adipose tissue: Fat cells produce estrogen via the aromatase enzyme
- Oral contraceptive use: Synthetic estrogens suppress natural progesterone production
Estrogen dominance symptoms include heavy periods, fibroids, endometriosis, fibrocystic breasts, PMS, water retention, weight gain around the hips and thighs, mood swings, headaches, and difficulty sleeping. Addressing estrogen dominance requires a multifaceted approach including supporting progesterone production, improving estrogen metabolism and elimination, reducing xenoestrogen exposure, and optimizing gut health.
FSH and LH: Fertility and Menopause Markers
Follicle-stimulating hormone (FSH) and luteinizing hormone (LH) are pituitary gonadotropins that regulate reproductive function in both men and women. Their levels provide important information about ovarian reserve, fertility potential, and menopausal status.
FSH
FSH stimulates the growth and maturation of ovarian follicles in women and spermatogenesis in men. In women, FSH is best tested on day 3 of the menstrual cycle (early follicular phase). Elevated day-3 FSH (above 10-12 mIU/mL) suggests diminished ovarian reserve, meaning the pituitary is working harder to stimulate the ovaries. FSH levels above 25-30 mIU/mL on two separate occasions are generally consistent with menopause. In men, elevated FSH may indicate primary testicular failure, while low FSH can suggest pituitary dysfunction.
LH
LH triggers ovulation in women and stimulates testosterone production in men via the Leydig cells. The LH-to-FSH ratio is clinically significant: in PCOS, the LH:FSH ratio is often elevated (greater than 2:1 or 3:1), reflecting dysregulated pituitary signaling and contributing to excess androgen production. A mid-cycle LH surge confirms ovulation has occurred, which is important for fertility assessment and for timing progesterone testing.
In perimenopausal women, FSH and LH levels can fluctuate dramatically from one cycle to the next. A single elevated reading does not definitively confirm menopause, and serial testing over several months may be necessary to establish a trend. Anti-Mullerian hormone (AMH) is an additional marker that provides a more stable assessment of ovarian reserve and is less cycle-dependent than FSH.
Fasting Insulin as a Metabolic Health Marker
While not traditionally classified as a "sex hormone," fasting insulin is arguably one of the most important markers to include in any comprehensive hormone panel. Insulin is a master regulatory hormone that profoundly influences sex hormone production, SHBG levels, inflammation, and overall metabolic health.
Fasting glucose alone is an inadequate marker of metabolic health. Blood glucose can remain normal for years while insulin levels climb progressively higher — a state known as compensatory hyperinsulinemia. By the time fasting glucose becomes elevated (indicating prediabetes or diabetes), insulin resistance has often been present for a decade or more.
Optimal fasting insulin levels in functional medicine are generally considered to be between 2 and 6 mIU/L. Levels above 8-10 mIU/L suggest insulin resistance, even when fasting glucose appears normal. Elevated insulin has widespread hormonal consequences:
- Increases ovarian androgen production: A key driver of PCOS and hormonal acne
- Lowers SHBG: Increasing free testosterone and estradiol bioavailability
- Increases aromatase activity: Promoting conversion of testosterone to estrogen, particularly in men with visceral adiposity
- Promotes fat storage: Especially in the abdominal region
- Drives inflammation: Through NF-kB and other inflammatory pathways
- Disrupts ovulation: Contributing to anovulatory cycles and infertility
The HOMA-IR (Homeostatic Model Assessment of Insulin Resistance) score, calculated from fasting glucose and fasting insulin, provides a useful quantitative measure of insulin resistance. A HOMA-IR below 1.5 is considered optimal, while values above 2.5 strongly suggest clinically significant insulin resistance.
IGF-1: Growth Hormone Marker
Insulin-like growth factor 1 (IGF-1) is the primary mediator of growth hormone (GH) activity in the body. Because growth hormone is released in pulsatile bursts (primarily during deep sleep), direct measurement of GH is impractical for routine assessment. IGF-1 serves as a stable surrogate marker that reflects overall growth hormone status.
IGF-1 is critical for tissue repair, muscle protein synthesis, bone remodeling, cognitive function, and cellular regeneration. Levels decline with age, and low IGF-1 is associated with:
- Loss of lean muscle mass and increased body fat
- Decreased bone density
- Reduced exercise capacity and recovery
- Cognitive decline and poor memory
- Thin skin and impaired wound healing
- Decreased sense of vitality and well-being
Optimal IGF-1 levels generally fall between 150 and 250 ng/mL for adults, though age-adjusted reference ranges should be considered. Very high IGF-1 levels (above 300 ng/mL) may warrant further evaluation, as chronically elevated IGF-1 has been associated with increased risk of certain cancers due to its growth-promoting effects.
Natural strategies to optimize IGF-1 include high-intensity interval training, adequate protein intake, deep restorative sleep (growth hormone is released primarily during slow-wave sleep), and minimizing alcohol consumption, which suppresses GH secretion.
Prolactin
Prolactin is a pituitary hormone best known for its role in lactation, but it has far-reaching effects beyond breastfeeding. Elevated prolactin (hyperprolactinemia) can suppress gonadotropin-releasing hormone (GnRH), leading to decreased FSH and LH production and, consequently, reduced sex hormone levels in both men and women.
Symptoms of elevated prolactin include:
- Irregular or absent menstrual periods in women
- Galactorrhea (milk discharge unrelated to breastfeeding)
- Decreased libido in both sexes
- Erectile dysfunction in men
- Infertility
- Headaches and visual changes (if caused by a pituitary adenoma)
Common causes of elevated prolactin include pituitary microadenomas (prolactinomas), certain medications (especially antipsychotics, SSRIs, and proton pump inhibitors), hypothyroidism (elevated TRH stimulates prolactin release), chronic stress, and nipple stimulation. Mild elevations can also result from the stress of the blood draw itself, so mildly elevated results should be confirmed with a repeat test drawn under calm conditions.
Normal prolactin levels are generally below 20 ng/mL in women and below 15 ng/mL in men. Levels above 100 ng/mL strongly suggest a prolactinoma and warrant MRI imaging of the pituitary gland.
When to Test Hormones
The timing of hormone testing is critical for obtaining accurate and clinically meaningful results. Incorrect timing is one of the most common reasons for misleading laboratory values.
Time of Day
- Cortisol: Morning fasting draw between 7:00-9:00 AM for serum; four-point collection at waking, noon, late afternoon, and bedtime for salivary testing
- Testosterone: Must be drawn before 10:00 AM, as levels peak in early morning and can drop by 30-40% later in the day
- Fasting insulin and glucose: After a 10-12 hour overnight fast
- Prolactin: Morning draw, ideally after a period of rest (stress and physical activity can transiently elevate levels)
- IGF-1: Can be drawn at any time of day (relatively stable)
Menstrual Cycle Timing for Women
- Day 3 (early follicular phase): FSH, LH, estradiol — baseline ovarian function assessment
- Day 12-14 (periovulatory): LH surge confirmation for ovulation timing
- Day 19-22 (mid-luteal phase): Progesterone, estradiol, estrogen-to-progesterone ratio — confirms ovulation and evaluates luteal phase adequacy
- DUTCH test: Collected during the mid-luteal phase (day 19-22) for the most comprehensive hormonal snapshot
Special Considerations
- Women on oral contraceptives will have suppressed natural hormone production; testing should ideally occur after at least 3 months off hormonal contraception to reflect true endogenous levels
- Perimenopausal women with irregular cycles may need to use ovulation predictor kits to time mid-luteal testing accurately
- Postmenopausal women and men can test at any point in the month, though morning draws remain important for cortisol and testosterone
- Patients using bioidentical hormone therapy should test at trough levels (immediately before the next dose) to assess baseline status
Symptoms of Hormonal Imbalance
Hormonal imbalances manifest through a wide array of symptoms that often overlap with other conditions, making laboratory testing essential for accurate diagnosis. The following symptom patterns are commonly associated with specific hormonal disruptions:
Fatigue and Low Energy
Persistent fatigue is the single most common complaint associated with hormonal imbalance. Cortisol dysregulation (both high and low), low thyroid function, low testosterone, low progesterone, and insulin resistance can all contribute to debilitating fatigue. Characterizing the pattern of fatigue (morning vs. afternoon, constant vs. fluctuating) helps direct the laboratory workup.
Weight Gain and Metabolic Dysfunction
Unexplained weight gain, particularly around the midsection, is frequently driven by insulin resistance, elevated cortisol, low testosterone, estrogen dominance, or hypothyroidism. Patients who "do everything right" with diet and exercise yet cannot lose weight often have an underlying hormonal component that must be addressed.
Mood Changes
Anxiety, depression, irritability, and emotional volatility are hallmark symptoms of hormonal imbalance. Low progesterone is strongly associated with anxiety (due to its GABA-enhancing effects), while low testosterone and estradiol deficiency are linked to depressive symptoms. Cortisol dysregulation affects neurotransmitter production and can mimic or exacerbate mood disorders.
Libido and Sexual Function
Low libido in both men and women is frequently related to low free testosterone, though elevated prolactin, low DHEA-S, HPA axis dysfunction, and estrogen-progesterone imbalance all play contributing roles. In men, erectile dysfunction may be an early sign of low testosterone or elevated estradiol. In women, vaginal dryness and painful intercourse often accompany declining estradiol levels in perimenopause.
Sleep Disturbances
Insomnia and disrupted sleep are often driven by an inverted cortisol curve (high evening cortisol), low progesterone (which promotes GABA-mediated relaxation), declining melatonin production, or blood sugar dysregulation during the night. Waking between 2:00 and 4:00 AM is a classic pattern of nocturnal hypoglycemia or cortisol dysregulation.
Natural Hormone Support Strategies
Before considering hormone replacement therapy, there are numerous evidence-based natural strategies that can significantly improve hormonal balance. These foundational interventions should form the basis of any hormone optimization protocol.
Sleep Optimization
Sleep is the single most powerful hormone-regulating behavior. Growth hormone, testosterone, and melatonin are produced primarily during deep sleep, while cortisol regulation depends on consistent circadian rhythm. Prioritize 7-9 hours of quality sleep, maintain a consistent sleep-wake schedule, minimize blue light exposure after sunset, keep the bedroom cool and dark, and address any underlying sleep apnea — a condition that profoundly disrupts testosterone and growth hormone production.
Stress Management
Chronic psychological stress is one of the most destructive forces in hormonal health. Effective stress management techniques include meditation, deep breathing exercises, yoga, time in nature, journaling, and setting appropriate boundaries. Heart rate variability (HRV) training using biofeedback devices can measurably improve vagal tone and reduce HPA axis activation. Even 10-15 minutes of daily mindfulness practice has been shown to lower cortisol levels and improve DHEA-S production.
Exercise
Regular physical activity is essential for hormonal health, but the type, intensity, and duration matter significantly:
- Resistance training: The most potent natural stimulus for testosterone and growth hormone production in both men and women. Compound movements (squats, deadlifts, bench press) using moderate-to-heavy loads are optimal.
- High-intensity interval training (HIIT): Stimulates growth hormone release and improves insulin sensitivity. Should be limited to 2-3 sessions per week to avoid overtraining.
- Moderate aerobic exercise: Improves cortisol regulation and insulin sensitivity. Walking, cycling, and swimming are excellent choices.
- Excessive endurance exercise: Chronic overtraining (particularly long-distance running) can suppress testosterone, elevate cortisol, disrupt menstrual cycles, and promote HPA axis dysfunction. More is not always better.
Adaptogenic Herbs
Adaptogenic herbs have a long history of use in traditional medicine systems for supporting the stress response and hormonal balance:
- Ashwagandha (Withania somnifera): One of the most well-studied adaptogens. Clinical trials demonstrate reductions in cortisol, improvements in thyroid function (particularly T4 to T3 conversion), enhanced testosterone levels, and improved sleep quality. Typical dosing: 300-600 mg of a standardized root extract (KSM-66 or Sensoril) daily.
- Rhodiola rosea: Supports mental performance under stress, reduces cortisol, and improves fatigue. Particularly helpful for cognitive symptoms of HPA axis dysfunction. Typical dosing: 200-400 mg of a standardized extract daily.
- Maca (Lepidium meyenii): Supports libido, energy, and hormonal balance without directly altering serum hormone levels. Gelatinized maca is better tolerated. Typical dosing: 1,500-3,000 mg daily.
- Holy basil (Ocimum sanctum): Reduces cortisol and blood sugar, supports a calm stress response. Typical dosing: 300-600 mg of a standardized extract daily.
- Vitex agnus-castus (Chasteberry): Specifically supports progesterone production by modulating pituitary LH secretion. Well-studied for PMS, luteal phase deficiency, and hyperprolactinemia. Typical dosing: 20-40 mg of a standardized extract daily, taken in the morning.
DIM for Estrogen Metabolism
Diindolylmethane (DIM) is a compound derived from the digestion of indole-3-carbinol (I3C), found in cruciferous vegetables such as broccoli, cauliflower, cabbage, and Brussels sprouts. DIM promotes the 2-hydroxylation pathway of estrogen metabolism, favoring the production of the protective 2-OH-E1 metabolite over the proliferative 16-alpha-OH-E1 and potentially genotoxic 4-OH-E1 metabolites.
DIM supplementation (typically 100-200 mg daily) is particularly beneficial for individuals with estrogen dominance, a history of estrogen-sensitive conditions, or unfavorable estrogen metabolite ratios on DUTCH testing. It is often combined with calcium-D-glucarate (which inhibits beta-glucuronidase and supports estrogen elimination through the gut) and sulforaphane from broccoli sprout extract (which upregulates phase II liver detoxification).
Additional Nutritional Support
- Zinc: Essential for testosterone production, immune function, and thyroid hormone conversion. Dosing: 15-30 mg daily with food.
- Magnesium: Supports cortisol regulation, progesterone production, insulin sensitivity, and sleep. Magnesium glycinate or threonate are preferred forms. Dosing: 200-400 mg daily.
- Vitamin D3: Functions as a prohormone and is essential for testosterone production, immune regulation, and mood. Optimal serum 25(OH)D levels are 50-70 ng/mL.
- Omega-3 fatty acids: Reduce inflammation, support hormone receptor sensitivity, and improve cell membrane fluidity. Dosing: 2-3 grams of combined EPA/DHA daily.
- B vitamins (methylated): Support estrogen detoxification, neurotransmitter synthesis, and adrenal function. Methylfolate (5-MTHF) and methylcobalamin are preferred forms for individuals with MTHFR polymorphisms.
Bioidentical vs. Synthetic Hormones
When natural interventions are insufficient to restore hormonal balance, hormone replacement therapy (HRT) may be warranted. In functional and naturopathic medicine, bioidentical hormones are generally preferred over synthetic alternatives.
Bioidentical Hormones
Bioidentical hormones are molecularly identical to the hormones produced by the human body. They are typically derived from plant precursors (soy or wild yam) and are modified in a laboratory to match the exact molecular structure of endogenous hormones such as estradiol, progesterone, testosterone, and DHEA. Because they are structurally identical to human hormones, they bind to hormone receptors in the same manner and are metabolized through the same enzymatic pathways.
Common bioidentical hormone preparations include:
- Micronized progesterone (Prometrium): FDA-approved oral bioidentical progesterone
- Transdermal estradiol patches, gels, or creams: Deliver estradiol directly into the bloodstream, bypassing first-pass liver metabolism
- Testosterone creams or pellets: Compounded bioidentical testosterone for men and women
- Compounded hormone preparations: Custom formulations created by compounding pharmacies to meet individual patient needs, including combinations like Biest (a blend of estriol and estradiol)
Synthetic Hormones
Synthetic hormones, such as medroxyprogesterone acetate (Provera) and conjugated equine estrogens (Premarin), have molecular structures that differ from human hormones. These structural differences lead to altered receptor binding, different metabolic pathways, and a different side-effect profile. The Women's Health Initiative (WHI) study, which raised significant concerns about HRT and cardiovascular and breast cancer risk, used synthetic hormones — not bioidentical ones. Many practitioners and researchers argue that the findings of the WHI should not be extrapolated to bioidentical hormone therapy.
Route of Administration Matters
The route of hormone delivery significantly affects safety and efficacy. Transdermal (through the skin) estrogen delivery is generally preferred over oral administration because it:
- Avoids first-pass hepatic metabolism
- Does not increase clotting factor production (reducing thromboembolism risk)
- Does not elevate SHBG as dramatically as oral estrogen
- Does not increase triglycerides or C-reactive protein
- Provides more stable hormone levels throughout the day
Similarly, oral micronized progesterone has a well-established safety profile and offers neuroprotective and sleep-promoting benefits that synthetic progestins do not provide.
Cautions and Considerations
While comprehensive hormone testing and optimization are powerful tools, several important cautions must be observed:
- Never self-prescribe hormones: Hormone replacement therapy, even with bioidentical preparations, should always be supervised by a qualified healthcare practitioner. Unsupervised hormone use can lead to serious adverse effects, including increased cancer risk, cardiovascular events, and worsening of underlying conditions.
- Test before you treat: Empiric hormone supplementation without baseline laboratory evaluation is irresponsible practice. Always establish baseline levels and monitor regularly during treatment.
- Monitor downstream metabolites: Simply replacing a hormone without monitoring its metabolites can create new imbalances. For example, testosterone supplementation can increase estradiol through aromatization, and DHEA supplementation can unpredictably increase androgens or estrogens.
- Address root causes first: Hormone imbalances are often symptoms of deeper issues — chronic stress, poor sleep, blood sugar dysregulation, gut dysfunction, environmental toxin exposure, or nutrient deficiencies. Hormone replacement without addressing these underlying drivers provides only temporary relief.
- Lab values are not the whole picture: Clinical symptoms must be correlated with laboratory findings. A patient with "normal" lab values who is profoundly symptomatic deserves further investigation, while an asymptomatic patient with a mildly out-of-range value may not require aggressive intervention.
- Consider contraindications: Hormone therapy — particularly estrogen — is contraindicated or requires extreme caution in individuals with a history of hormone-sensitive cancers (breast, ovarian, endometrial, prostate), active liver disease, unexplained vaginal bleeding, or a history of blood clots or stroke. A thorough personal and family health history is essential before initiating any hormone therapy.
- Beware of over-the-counter hormone products: DHEA, pregnenolone, and melatonin are available without a prescription in many countries. Their accessibility does not make them risk-free. These are potent hormones that can create downstream imbalances when used without monitoring.
- Compounding pharmacy quality varies: Not all compounding pharmacies maintain the same standards of quality and potency. Use only pharmacies that follow USP (United States Pharmacopeia) compounding standards and undergo third-party testing.
- Periodic reassessment is essential: Hormonal needs change with age, stress levels, and life circumstances. Regular retesting (every 3-6 months initially, then annually once stable) ensures that treatment remains appropriate and safe.
Comprehensive hormone testing, when performed correctly and interpreted in the context of the whole patient, is one of the most valuable tools available in functional medicine. It provides actionable data that can guide targeted interventions — from lifestyle modifications and nutritional support to carefully monitored bioidentical hormone therapy — that restore balance, vitality, and quality of life.
Research Papers and References
The following are curated PubMed literature searches covering the evidence base for hormone panel testing, interpretation, and clinical management. Each link opens a live, filtered PubMed query so the results stay current as new studies are indexed.
- Comprehensive hormone testing — PubMed literature search
- Testosterone free total bioavailable — PubMed literature search
- Estradiol measurement — PubMed literature search
- DUTCH hormone test — PubMed literature search
- Progesterone luteal phase — PubMed literature search
- Estrogen dominance — PubMed literature search
- Cortisol diurnal rhythm — PubMed literature search
- DHEA-S aging — PubMed literature search
- IGF-1 growth hormone — PubMed literature search
- Thyroid hormones and sex hormones — PubMed literature search
External Authoritative Resources
- MedlinePlus — Laboratory Tests Reference
- Lab Tests Online — Patient-Oriented Lab Test Reference (AACC)
- StatPearls / NCBI Bookshelf — Clinical Laboratory Reference
Connections
- Testosterone
- DHEA-S
- Cortisol Test
- Thyroid Panel
- Fasting Insulin
- PCOS
- Menopause & HRT
- Perimenopause
- Adrenal Fatigue
- Insulin Resistance
- Anxiety
- Depression
- Fatigue
- Insomnia
- Magnesium
- Perimenopause Symptom Tracker
- Stress Management
- Estradiol Formulations
Featured Videos
Hormones: Are Your Hormones Normal? What Do your Lab Numbers Mean?

What Hormones to Test and When | FSH, LH, Estradiol, AMH, and More

Hormone Testing 101

Mistakes to avoid with hormone testing for acne.

Testosterone blood test explained, and your other male hormones.

What type of hormone testing is best for you?

Hormone Testing Methods: Blood, Urine or Saliva? | Dr. Breen Explains

Supercharge your Testosterone by Optimizing this 1 Blood Test | SHBG Sex hormone Binding Globulin

DUTCH Hormone Test FAQs: Everything You Need to Know

I Want a Full Hormone Panel! Why Won’t My Provider Order it? Listener Question HealthiHer Ep #92

Female Hormone blood tests explained - from periods to pregnancy to menopause

WOMEN'S HORMONES Blood Test Results Explained: Menopause & Fertility | Dr. Natalie Crawford

DUTCH Test Explained - Advanced Hormone Testing With Esther Blum

Female Blood Panel Hormonal Blood Testing

Vibrant's Hormone Zoomer - Report Walkthrough

How Can I Get My Hormones Checked For Imbalance?