Sleep, Exercise, and Lifestyle for Chronic Pain
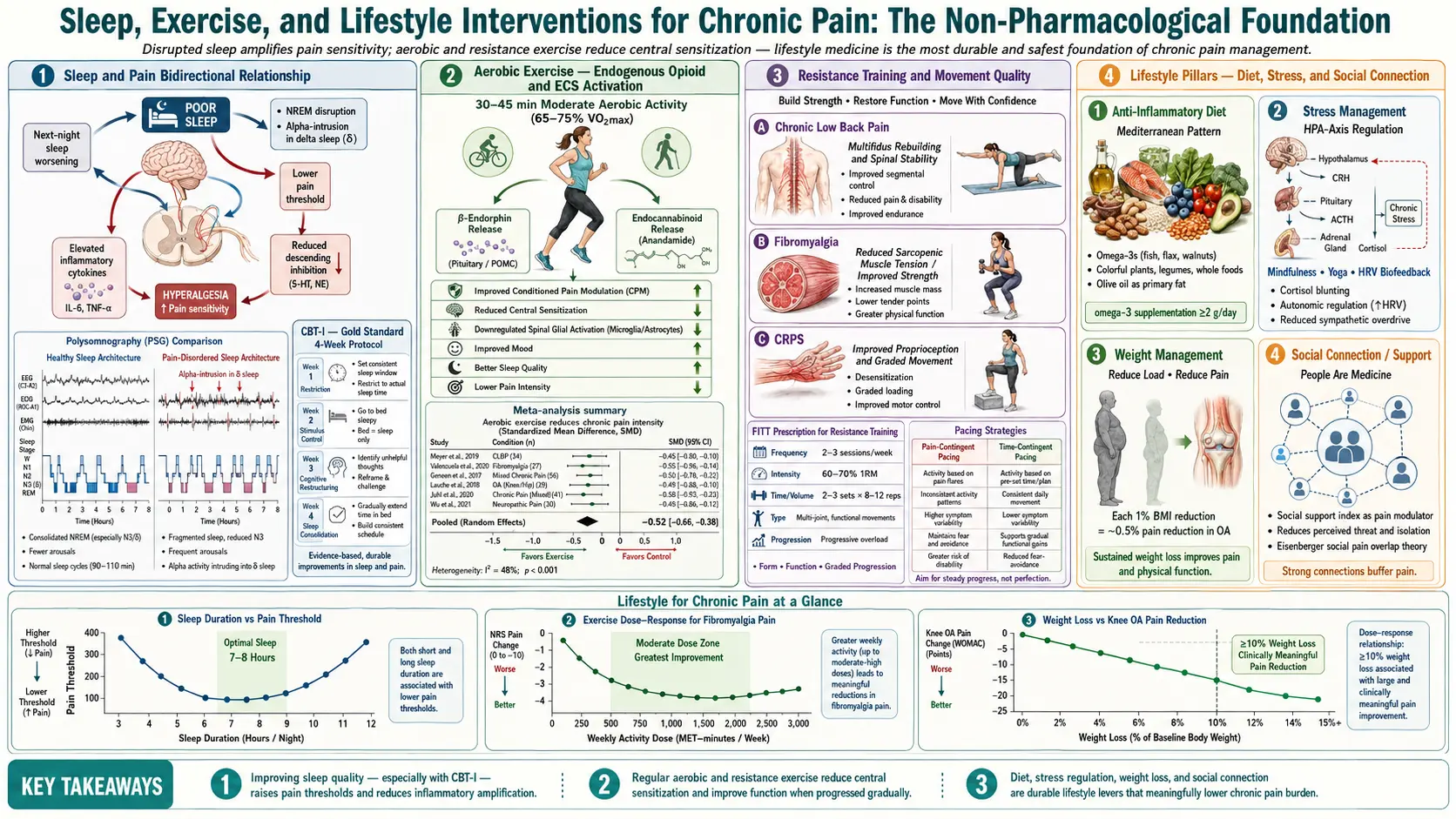
Table of Contents
- Why Lifestyle Matters More Than You Think
- The Sleep-Pain Loop — One Bad Night Costs You the Next Day
- CBT for Insomnia (CBT-I) — The Evidence-Based Fix
- Sleep Hygiene — What Actually Helps and What Doesn't
- Exercise — The 150-Minute Threshold and Why Low Doses Still Count
- Strength Training — Twice a Week, Non-Negotiable
- Pacing and the 80/20 Rule — Breaking the Boom-Bust Cycle
- Mediterranean Eating — Lowering Background Inflammation
- Weight, Knee OA, and the 1-to-4 Ratio
- Smoking and Alcohol — Two Habits That Feed Pain
- Stress, Nervous System, and Pain Volume
- Social Connection, Loneliness, and Meaning
- Putting It Together — A Realistic Week
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
Why Lifestyle Matters More Than You Think
If you have lived with chronic pain for more than a few months, you have probably been told to "eat better, sleep more, and exercise." Maybe your doctor said it in thirty seconds on the way out of the room. Maybe a well-meaning relative said it over dinner. Either way, it probably felt dismissive — as if your pain were the result of bad habits rather than a real medical condition.
It is not that simple. Chronic pain is a real neurological condition involving sensitized pathways in your spinal cord and brain (see Central Sensitization). But the research over the last fifteen years has shown something important: the day-to-day inputs your nervous system receives — sleep, movement, food, stress, connection — directly turn the pain volume knob up or down. Not metaphorically. Measurably. In laboratory studies, a single night of poor sleep can drop your pain threshold the next morning. A twelve-week aerobic exercise program can shift the brain's descending pain-inhibition network. Losing one pound can take four pounds of load off a bad knee.
This article is not about "trying harder." It is about the specific, measurable lifestyle levers that every major pain guideline — NICE, the American College of Physicians, the CDC — now ranks alongside or above medications for long-term outcomes. These are not alternatives to medical care. They are the foundation on which medication, injections, and therapy actually work.
The Sleep-Pain Loop — One Bad Night Costs You the Next Day
Sleep and pain are locked in a two-way relationship. Pain wakes you up and fragments your sleep. Poor sleep, in turn, lowers your pain threshold the next day. The loop tightens over weeks and months until many patients cannot tell which came first.
The clearest laboratory evidence comes from Michael Smith's lab at Johns Hopkins. In controlled studies, healthy volunteers deprived of sleep for one night showed a measurable next-day increase in pain sensitivity — lower thresholds to heat, pressure, and cold — along with reduced activity in the brain's descending pain-inhibition pathway. In other words, your built-in pain brakes work worse after a bad night. Fragmented sleep (multiple awakenings) was even more pain-provoking than simply short sleep.
Population studies echo this. In long-term cohorts, people with chronic insomnia are roughly twice as likely to develop chronic widespread pain within the following five to ten years, independent of depression and anxiety. Among people who already have pain, sleep disturbance predicts worse pain trajectories more reliably than the initial pain severity itself.
The practical implication is uncomfortable but empowering: fixing sleep is not optional — it is a core pain treatment. If you have been chasing sleep with over-the-counter aids, alcohol, or cannabis and waking up groggy and still in pain, you are treating the symptom while leaving the underlying sleep architecture broken.
CBT for Insomnia (CBT-I) — The Evidence-Based Fix
Cognitive Behavioral Therapy for Insomnia (CBT-I) is the first-line treatment endorsed by the American College of Physicians, the American Academy of Sleep Medicine, the European Sleep Research Society, and essentially every major guideline body. In head-to-head trials against prescription sleep medications (zolpidem, eszopiclone, trazodone), CBT-I matches or beats the drugs in the short term and clearly wins at 6 and 12 months because its effects persist after treatment ends. Drugs stop working the night you stop taking them.
CBT-I is not sleep hygiene. It is a structured, usually 4-to-8-session protocol combining:
- Sleep restriction — temporarily compressing your time in bed to match your actual sleep time, which consolidates fragmented sleep. Counterintuitive and brutal in week one. Transformative by week three.
- Stimulus control — the bed is only for sleep and sex. No reading, no phone, no worrying. If you are awake more than twenty minutes, get up.
- Cognitive restructuring — identifying and defusing the catastrophic sleep thoughts that keep you awake ("if I don't sleep now I'll be destroyed tomorrow").
- Relaxation training and sleep education — how sleep actually works, why naps sabotage nighttime sleep, how caffeine lingers.
For people with comorbid chronic pain and insomnia — the typical situation — CBT-I also improves the pain, not just the sleep. Randomized trials in fibromyalgia, osteoarthritis, and chronic low back pain populations have all shown pain reductions alongside sleep gains. This is why the current guideline for a patient presenting with "I can't sleep because of my pain" is CBT-I first, medication second.
How to access it. Finding a CBT-I therapist is harder than finding a prescriber, but it is worth it. Options:
- In-person — psychologists trained in behavioral sleep medicine. Search the Society of Behavioral Sleep Medicine directory.
- Digital CBT-I — evidence-based app-delivered programs (SHUTi, Sleepio, Somryst) have shown results nearly matching in-person therapy in randomized trials. Somryst is FDA-cleared and sometimes insurance-covered.
- VA patients — the VA has rolled out CBT-I across its system and it is one of the most-accessed therapies in VA mental health.
- Books — "Quiet Your Mind and Get to Sleep" (Carney & Manber) walks you through a self-guided version.
Sleep Hygiene — What Actually Helps and What Doesn't
"Sleep hygiene" is the term for the checklist of habits around sleep: cool room, dark room, consistent bedtime, no caffeine after noon, no screens before bed. The uncomfortable truth is that sleep hygiene alone rarely fixes established insomnia. Trials repeatedly show it underperforms CBT-I. But the habits still matter as a foundation, especially for people whose sleep is borderline rather than severely broken.
Worth doing:
- Consistent wake time, seven days a week. This anchors your circadian rhythm more powerfully than bedtime does. Sleeping in on weekends resets your body clock later every time.
- Cool, dark, quiet room. Target around 65–68°F (18–20°C). Blackout curtains. White noise or earplugs if needed.
- Morning daylight. 10–20 minutes of outdoor light within an hour of waking is the strongest circadian signal available. More effective than any supplement.
- Caffeine cutoff by noon. Caffeine's half-life is 5–6 hours; a 3 p.m. coffee still has meaningful levels in your system at midnight.
- No large meals within three hours of bed. Reflux and digestion both fragment sleep.
Common pitfalls that actively backfire:
- Going to bed earlier because you slept badly. This increases time-in-bed-awake, which trains your brain that bed means wakefulness. Counterintuitively, a slightly later bedtime usually helps.
- Alcohol as a sleep aid. It knocks you out but destroys deep and REM sleep in the second half of the night. You wake unrefreshed.
- Long daytime naps. If you have insomnia, a nap longer than twenty minutes after 2 p.m. steals from your nighttime sleep drive.
- Clock-watching. Turn the clock away. Knowing it is 3:47 a.m. never helped anyone fall back asleep.
- Over-the-counter diphenhydramine (Benadryl, ZzzQuil). Strongly anticholinergic. Disrupts deep sleep architecture, causes next-day fog, and is linked to cognitive decline in older adults. Avoid.
Exercise — The 150-Minute Threshold and Why Low Doses Still Count
Exercise is the single most evidence-backed non-drug intervention for nearly every chronic pain condition studied — low back pain, knee osteoarthritis, fibromyalgia, neck pain, chronic widespread pain. Cochrane reviews across all of these conclude the same thing: movement wins.
The current consensus target, from WHO and nearly every pain guideline, is 150 minutes of moderate aerobic activity per week, ideally spread across most days. "Moderate" means you can talk but not sing — roughly a brisk walk, easy bike, or slow jog. That works out to about 30 minutes five days a week, or 22 minutes every day.
If 150 minutes sounds impossible right now, do not skip exercise because you cannot hit the target. The dose-response curve is not linear. The biggest gains, per minute invested, come at the low end — moving from zero to 30 minutes a week of walking is statistically a bigger jump in pain and function than moving from 150 to 250. People who do any exercise have measurably better outcomes than people who do none.
A realistic ramp for someone starting from near-zero:
- Week 1–2. 5–10 minutes of easy walking, most days. That is it. You are teaching your nervous system that movement is safe, not building cardiovascular fitness yet.
- Week 3–4. 15 minutes, five days a week.
- Week 5–8. Add a "long day" of 20–25 minutes, keeping the others at 15.
- Week 9–12. Work toward 30 minutes, five days a week — the 150-minute target.
This is the same structure as the "Couch to 5K" running apps, but you can substitute anything — walking, swimming, stationary bike, elliptical, recumbent bike. The modality matters less than the consistency. Swimming and aquatic exercise are gold-standard for anyone with knee, hip, or back arthritis because water offloads joint pressure by 75–90 percent.
Expect a paradox in the first two to four weeks: some pages, some days, your pain may briefly worsen. This is not damage — it is a sensitized nervous system recalibrating. If you start at a low enough dose and hold steady, the flare-ups settle within a few weeks and pain begins to decrease. The patients who fail are almost always the ones who did too much on day one, spiked their pain, and quit.
Strength Training — Twice a Week, Non-Negotiable
Aerobic exercise is only half of the prescription. Every major guideline now recommends resistance (strength) training at least twice a week for chronic pain patients, particularly for osteoarthritis, low back pain, and fibromyalgia. The reasons are biomechanical and neurological:
- Stronger muscles offload joints. Quadriceps strength in knee OA, core strength in back pain, and grip strength in hand OA all independently predict better pain and function.
- Sarcopenia (age-related muscle loss) accelerates disability. Adults lose roughly 3–8 percent of muscle mass per decade after age thirty without resistance training.
- Strength training lowers central sensitization markers in fibromyalgia trials — not as dramatically as aerobic exercise, but additively.
You do not need a gym. Bodyweight squats, wall push-ups, sit-to-stand from a chair, step-ups, and resistance bands deliver most of the benefit. Two 20-to-30-minute sessions per week, covering major muscle groups (legs, back, chest, core), is enough. Progress by adding one rep, one set, or a small increment of resistance each week — the "progressive overload" principle.
If available, a referral to physical therapy for an initial 4–8 weeks is the best starting point. A PT can screen for form issues, match the program to your specific pain pattern, and get you comfortable with the movements before you continue independently.
Pacing and the 80/20 Rule — Breaking the Boom-Bust Cycle
One of the most damaging patterns in chronic pain is boom-bust cycling. On a good day, you feel better, so you catch up on everything — cleaning, errands, the yard, a long walk. You overdo it. The next two or three days you are flat on the couch, hurting worse than before. Over months, your overall activity decreases because the crashes last longer than the boom days earn.
The alternative is called pacing. The core idea is simple: do a consistent 80 percent of what you think you can do on a good day, every day. Leave the last 20 percent in the tank. The shorthand is the "80/20 rule of pacing."
Concretely:
- Find your baseline. Pick one activity (walking, standing, gardening). Time how long you can do it on an average day before pain forces a break. Take 80 percent of that.
- Stick to that 80 percent even on good days. This is the hardest part. You will feel like you are "wasting" a good day. You are not — you are breaking the cycle.
- Use time, not pain, as the stop signal. If your baseline is 20 minutes of standing, stand for 20 minutes and sit down, even if you still feel okay.
- Increase baseline by 10 percent per week once stable. Slow, steady, boring — and effective.
Pacing is the operational counterpart to graded exercise. It is also the heart of programs for ME/CFS (see Chronic Fatigue Syndrome), where pacing is even more critical because post-exertional malaise can last days or weeks. For fibromyalgia, pacing combined with graded aerobic exercise is evidence-based (see Exercise Pacing and Graded Movement).
Mediterranean Eating — Lowering Background Inflammation
Diet is not a cure for chronic pain. But the long-term pattern of what you eat influences systemic inflammation, body weight, insulin sensitivity, and gut health — all of which feed into pain.
The dietary pattern with the strongest evidence for pain and general health outcomes is the Mediterranean diet: olive oil as the primary fat, abundant vegetables and fruit, whole grains, beans and lentils, fish two or more times a week, moderate dairy (especially yogurt and cheese), limited red meat, nuts and seeds daily, and red wine only in moderation if at all.
In trials, Mediterranean-style eating lowers C-reactive protein (CRP) — a general marker of systemic inflammation — by 15–30 percent over 6–12 months. In osteoarthritis and rheumatoid arthritis cohorts, adherence to this pattern is associated with lower pain scores and better function. In migraine, it reduces frequency. The PREDIMED trial demonstrated cardiovascular and metabolic benefits that compound over years.
The specific anti-inflammatory components that matter most:
- Omega-3 fatty acids — fatty fish (salmon, sardines, mackerel) two to three times a week, or 1–2 g daily EPA+DHA from supplements.
- Extra-virgin olive oil — oleocanthal, a compound in EVOO, has ibuprofen-like effects on the same COX enzymes.
- Polyphenols from colorful vegetables, berries, green tea, dark chocolate.
- Fiber — 25–35 g/day feeds gut bacteria that produce short-chain fatty acids, which are systemically anti-inflammatory.
What to cut back on:
- Ultra-processed foods. In cohort studies, high intake is independently associated with more chronic pain and worse depression — probably through inflammation, obesity, and gut dysbiosis.
- Added sugar. Drives weight gain, insulin resistance, and CRP.
- Industrial seed oils used in large amounts for frying. The omega-6 to omega-3 ratio in most Western diets is wildly skewed.
Beware elimination diets marketed as pain cures — gluten-free, dairy-free, nightshade-free — unless you have a specific condition (celiac disease, true food allergy) justifying them. For most chronic pain patients, restrictive diets add stress and nutritional gaps without improving pain.
Weight, Knee OA, and the 1-to-4 Ratio
For pain conditions involving weight-bearing joints — knee OA, hip OA, chronic low back pain in people with obesity — weight loss is one of the most powerful interventions available.
The landmark data come from the IDEA trial (Intensive Diet and Exercise for Arthritis), led by Stephen Messier and published in JAMA in 2013. In 454 overweight and obese adults with knee OA, the combination of diet-driven weight loss plus exercise was compared with diet alone, exercise alone, and usual care. The combined group lost on average 23 lb (10.6 kg) over 18 months and showed the largest improvements in pain, function, and walking speed. Imaging and gait analysis revealed something striking: each pound of body weight lost reduced the compressive load across the knee by roughly four pounds per step. A 20-pound loss took about 80 pounds of cumulative load off the knee with every stride.
Practical takeaways:
- Modest losses matter. 5–10 percent of body weight delivers most of the pain benefit. You do not need to reach an "ideal" BMI.
- Diet plus exercise beats either alone. Weight loss without strength training loses muscle and weakens the joint support. Exercise without diet change rarely produces enough weight loss to matter for severe knee OA.
- The newer GLP-1 medications (semaglutide, tirzepatide) are changing this conversation. For patients with obesity plus knee OA, the STEP-9 trial (2024) showed that semaglutide 2.4 mg produced meaningful weight loss and knee pain reduction over 68 weeks. These drugs are expensive and not universally appropriate, but they are now a legitimate option for discussion with your physician.
This is not about shaming body size. Plenty of people with chronic pain are not overweight. But if you have knee, hip, or lumbar OA and excess weight, the mechanical math is unforgiving — and unusually responsive to change.
Smoking and Alcohol — Two Habits That Feed Pain
Smoking is a chronic pain risk factor, full stop. It is associated with higher rates of chronic low back pain, fibromyalgia, rheumatoid arthritis, and post-surgical pain. The mechanisms are multiple: nicotine directly sensitizes pain pathways, smoking reduces blood flow to spinal discs, and tobacco smoke is pro-inflammatory across nearly every tissue. Smokers also metabolize some pain medications (including opioids) faster and get less relief per dose.
Nicotine is a tricky drug for pain because acute nicotine can temporarily reduce pain — which is why smokers often say a cigarette "takes the edge off." But chronically, nicotine upregulates pain signaling. The withdrawal-use cycle makes baseline pain worse. Patients who quit smoking, with adequate nicotine-replacement support during the transition, almost uniformly report improvement in pain over 3–12 months.
If you smoke and have chronic pain, quitting is arguably one of the highest-leverage interventions available. Combine nicotine replacement, varenicline or bupropion if indicated, and behavioral support (quitlines, apps, groups). Do not try to do it during a major pain flare — stabilize first, then quit.
Alcohol is more nuanced. Moderate intake is not strongly linked to chronic pain. But alcohol devastates sleep architecture: it shortens REM sleep, suppresses deep slow-wave sleep in the second half of the night, and fragments overall sleep. Because sleep is a core pain driver, any meaningful alcohol use (more than one drink a few times a week) commonly worsens pain through the sleep pathway.
Alcohol also interacts with many pain medications — benzodiazepines, opioids, gabapentinoids, and even over-the-counter acetaminophen (liver toxicity). If you are using alcohol to cope with pain or to sleep, it is one of the most common reversible causes of stalled treatment.
Stress, Nervous System, and Pain Volume
Chronic stress keeps the sympathetic nervous system (the fight-or-flight branch) persistently activated, which turns up the pain amplifier in the spinal cord and brain. Cortisol dysregulation, muscle guarding, shallow breathing, and reduced heart-rate variability all feed the pain loop. This is not psychological weakness — it is measurable physiology.
The evidence-based stress tools for pain:
- Mindfulness-Based Stress Reduction (MBSR) — 8-week group program developed by Jon Kabat-Zinn. Randomized trials in chronic low back pain, fibromyalgia, and mixed chronic pain show moderate but durable reductions in pain interference and catastrophizing.
- Paced breathing. 5–6 breaths per minute (roughly 5 seconds in, 5 seconds out) for 10 minutes daily raises heart-rate variability and downshifts the sympathetic nervous system.
- Yoga and tai chi. Both combine gentle movement, breath, and attention. Both have evidence for chronic low back pain, neck pain, and knee OA.
- Cognitive Behavioral Therapy for pain (CBT) — targets the thought-emotion-behavior patterns that amplify pain. See also Pain Reprocessing Therapy for a related approach.
- Time in nature. Studies consistently show that outdoor time (90+ minutes a week in green or blue space) reduces rumination and stress markers.
None of these replace medical care. All of them shift the background tone of your nervous system in a direction that makes every other treatment work better.
Social Connection, Loneliness, and Meaning
This last section may be the most uncomfortable, because it is the least controllable. But it is also among the most consistently supported in the pain literature.
Loneliness is a pain amplifier. Large cohort studies (including the English Longitudinal Study of Ageing and the U.S. Health and Retirement Study) have shown that people who report chronic loneliness have measurably higher rates of chronic pain, slower recovery from acute pain, and higher use of pain medications — independent of depression. Functional MRI work by Naomi Eisenberger at UCLA showed that social rejection activates overlapping brain regions with physical pain. The brain processes "I am alone and unsupported" through the same circuitry that processes tissue injury.
The flip side: social connection and purpose measurably buffer pain. Patients with strong support networks report lower pain at equivalent disease severity. Support groups for specific conditions (fibromyalgia, EDS, CRPS) improve coping and reduce catastrophizing. Having a reason to get out of bed — work, caregiving, a hobby, a community role — predicts better function than pain severity alone.
Practical moves:
- Reach out to one person a week you have drifted from. A text, a short call, a coffee. Small doses add up.
- Find your people. Online communities for your specific condition (Reddit subs, Facebook groups moderated by clinicians, patient nonprofits) reduce the isolation of being misunderstood.
- Purposeful work or volunteering — even small amounts — is strongly protective. It gives the nervous system a reason to show up.
- Guard against full retreat. Chronic pain often shrinks social life. That shrinkage then amplifies pain. Push back where you can.
Putting It Together — A Realistic Week
None of this works if you try to do it all on Monday. A realistic starting plan for someone overwhelmed:
- Month 1. Pick one habit. Most patients should start with sleep — set a consistent wake time, cap caffeine at noon, get 10 minutes of morning daylight.
- Month 2. Add daily walking. Start at 5–10 minutes and build 10 percent a week.
- Month 3. Add twice-weekly strength work (bodyweight or bands). Consider a PT referral.
- Month 4. Shift one meal a day toward Mediterranean pattern — more vegetables, olive oil, beans; fewer ultra-processed foods.
- Month 5. Add a stress tool — MBSR, 10 minutes of paced breathing, yoga class, CBT therapist.
- Month 6. Address smoking, alcohol, or social isolation if any apply.
Six months. One thing at a time. This is the pattern that produces durable change. Trying to overhaul everything at once is the same boom-bust trap that drives pain flares — just at the lifestyle level.
Key Research Papers
- Smith MT, et al. The effects of sleep deprivation on pain inhibition and spontaneous pain in women. Sleep. 2007.
- Messier SP, et al. Effects of intensive diet and exercise on knee joint loads, inflammation, and clinical outcomes among overweight and obese adults with knee osteoarthritis: the IDEA randomized clinical trial. JAMA. 2013.
- Qaseem A, et al. Management of chronic insomnia disorder in adults: a clinical practice guideline from the American College of Physicians. Ann Intern Med. 2016.
- Geneen LJ, et al. Physical activity and exercise for chronic pain in adults: an overview of Cochrane reviews. Cochrane Database Syst Rev. 2017.
- Estruch R, et al. Primary prevention of cardiovascular disease with a Mediterranean diet (PREDIMED). N Engl J Med. 2018.
- Bliddal H, et al. Once-weekly semaglutide in persons with obesity and knee osteoarthritis (STEP-9). N Engl J Med. 2024.
- Finan PH, Goodin BR, Smith MT. The association of sleep and pain: an update and a path forward. J Pain. 2013.
- Eisenberger NI, Lieberman MD, Williams KD. Does rejection hurt? An fMRI study of social exclusion. Science. 2003.
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on sleep, exercise, and lifestyle approaches to chronic pain:
- Sleep deprivation and pain threshold
- CBT-I for comorbid chronic pain and insomnia
- Aerobic exercise dose-response in chronic pain
- Resistance training in fibromyalgia and osteoarthritis
- Activity pacing for chronic pain
- Mediterranean diet, CRP, and pain
- Weight loss and knee OA joint load
- Smoking as a chronic pain risk factor
- Loneliness, social isolation, and chronic pain
- Mindfulness and MBSR for chronic pain
Connections
- Chronic Fatigue Syndrome
- Exercise Pacing and Graded Movement
- Central Sensitization and Nociplastic Pain
- Pain Reprocessing Therapy and PNE
- Chronic Low Back Pain — Evidence-Based Care
- Chronic Pain Overview
- Fibromyalgia
- Insomnia
- Non-Opioid Medications — Gabapentinoids, SNRIs, LDN
- Interventional Pain — Injections, Ablations, Neuromodulation
- Nociceptive vs Neuropathic vs Nociplastic Pain
- Opioids for Chronic Non-Cancer Pain
- Complex Regional Pain Syndrome
- Sleep and Non-Restorative Sleep
- Arthritis
- Depression
- Obesity
- Sleep Hygiene