Interventional Pain — Injections, Ablations, Neuromodulation
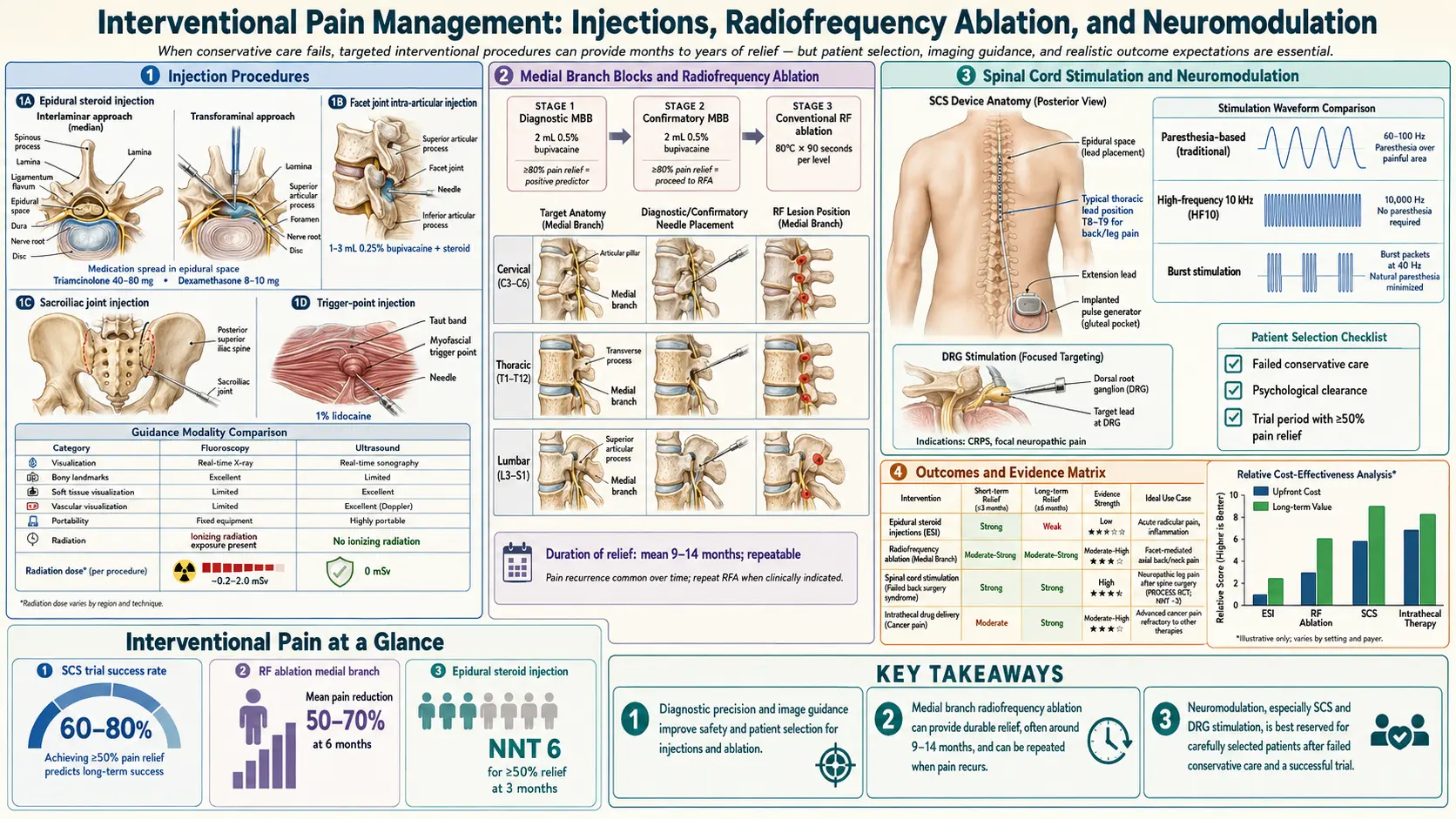
Table of Contents
- What Interventional Pain Medicine Is
- Epidural Steroid Injections
- Facet Joint Injections, Medial Branch Blocks & RFA
- Sacroiliac Joint Injections & RFA
- Genicular Nerve RFA for Knee Osteoarthritis
- Trigger Point Injections
- Sympathetic Blocks — Stellate & Lumbar
- Celiac Plexus Block for Abdominal Pain
- Intrathecal Drug Delivery Pumps
- Spinal Cord Stimulators
- Dorsal Root Ganglion (DRG) Stimulation
- Peripheral Nerve Stimulators
- Vertebroplasty & Kyphoplasty
- Selection Criteria & Failure Modes
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
What Interventional Pain Medicine Is
Interventional pain medicine is the subspecialty that uses image-guided needles, radiofrequency probes, and implanted electrical devices to interrupt pain signals at a specific anatomical target. The umbrella covers everything from a ten-minute steroid injection in an office procedure room to a six-figure implanted spinal cord stimulator in a hospital operating suite. Procedures are performed by fellowship-trained physicians — most commonly anesthesiologists, physiatrists (PM&R), neurologists, or neurosurgeons with pain fellowships.
Two honest truths shape everything below. First, interventional procedures work best when paired with the rest of a pain-care plan — movement, sleep, psychological skills, and evidence-based medication — not as a replacement. Second, the evidence base is uneven. Some procedures (genicular RFA, spinal cord stimulation for failed back surgery syndrome, kyphoplasty in selected fracture patients) have solid randomized data. Others (lumbar epidurals for axial back pain, SI joint blocks for nonspecific pain) have famously modest or mixed results. A good interventional pain physician is candid about which category a recommended procedure falls into.
Image guidance matters. Fluoroscopy (live X-ray), ultrasound, or CT ensures the needle is where the operator believes it is. "Blind" or landmark-based injections — still common for trigger points — have higher miss rates when the target is deep. When someone offers you a spinal injection without fluoro or ultrasound, walk out.
Epidural Steroid Injections
Epidural steroid injections (ESIs) deliver a corticosteroid — usually dexamethasone, methylprednisolone, or triamcinolone — into the epidural space surrounding irritated nerve roots. The goal is to tamp down the inflammation around a compressed or chemically irritated root causing radicular pain (sciatica, cervical radiculopathy). There are three approaches:
- Transforaminal ESI. The needle enters through the intervertebral foramen, placing medication directly at the target nerve root. This is the most precise approach and the preferred technique for unilateral radicular pain. Rare but serious complication: inadvertent intra-arterial injection of particulate steroid, which has caused spinal cord infarction. Use of non-particulate dexamethasone in the cervical and upper thoracic spine has become standard to mitigate this risk.
- Interlaminar ESI. Needle enters between the laminae in the midline. Simpler and safer than transforaminal but delivers medication more diffusely — better for bilateral or multilevel symptoms.
- Caudal ESI. Needle enters through the sacral hiatus at the base of the spine. Large volume, low specificity. Useful in post-surgical spines where epidural scarring distorts anatomy or when transforaminal access is unsafe.
What the evidence actually shows. For acute lumbar radicular pain (sciatica from a disc herniation), ESIs produce modest short-term relief — on average a 10–20 point reduction on a 100-point pain scale for about 6–12 weeks, with no durable effect on function or surgical rates at one year. For axial low back pain without radiculopathy, the evidence is negative — ESIs do not help nonspecific back pain and should not be offered for that indication. Typical cost in the U.S.: $800–$2,500 per injection under insurance, often several thousand more out-of-pocket. Most payers cap them at 3–4 per year. See the low back pain article for where ESIs fit in a broader care plan.
Facet Joint Injections, Medial Branch Blocks & RFA
Facet joints are the small paired joints at the back of each vertebral level that guide spinal motion. Like any synovial joint, they can become arthritic and pain-generating. Classic features of facet-mediated pain: axial (spine-centered) pain worse with extension and rotation, not radiating past the knee or elbow, localized paraspinal tenderness over the joint.
Workup proceeds in three careful steps, and skipping steps is a major failure mode:
- Diagnostic medial branch block (MBB). A tiny dose of local anesthetic is placed on the medial branch nerves that supply a suspected facet joint. If the patient records ≥80% pain relief during the anesthetic's active window, the joint is the likely source. Most guidelines require two concordant positive blocks on separate days before proceeding — one block alone has a high false-positive rate.
- Confirmation. Some payers accept a single diagnostic block plus typical clinical features; others (including Medicare in most regions) require two.
- Radiofrequency ablation (RFA). If both diagnostic blocks were strongly positive, a thermal lesion is made on each medial branch nerve using an RF probe heated to 80–90 °C for 90 seconds. The nerve regenerates over 6–18 months, so RFA is repeatable.
Intra-articular facet steroid injections (steroid placed inside the joint itself) have weak evidence and are falling out of favor. The pathway that works is MBB → MBB → RFA.
Outcomes. In appropriately selected patients (two concordant positive MBBs), lumbar facet RFA produces meaningful pain reduction for 6–12 months in about 60% of patients. Cervical facet RFA has similar outcomes for whiplash-associated neck pain. Expect to repeat RFA annually or every other year if it works. Cost: MBB $600–$1,500 per level; RFA $2,000–$5,000 per session depending on number of levels.
Sacroiliac Joint Injections & RFA
The sacroiliac (SI) joints are the large, mostly immobile articulations between the sacrum and the iliac bones. They are a real pain generator in perhaps 15–25% of chronic low back pain cases — particularly after lumbar fusion, during pregnancy, in inflammatory spondyloarthritis, and after falls onto the buttock.
Diagnosis is imperfect. Physical exam provocation tests (FABER, thigh thrust, Gaenslen, distraction, compression) are individually unreliable, but three or more positive tests raise suspicion. The definitive diagnostic step is a fluoroscopically guided intra-articular SI joint anesthetic block: if the patient gets ≥75–80% relief during the anesthetic window, the SI joint is confirmed.
- SI joint steroid injection. Corticosteroid into the joint capsule. Modest and variable duration of relief — weeks to months. Useful for inflammatory SI pain (axial spondyloarthritis flares) but often disappointing for mechanical SI pain.
- Lateral branch RFA. Radiofrequency lesions on the L5 dorsal ramus and the S1–S3 lateral branches that supply the posterior SI ligaments. Better outcomes than injections in mechanical SI pain — roughly half of properly selected patients get 6–12 months of meaningful relief.
- SI joint fusion. A surgical option (minimally invasive triangular titanium implants, the iFuse system) for patients who fail conservative care and have two confirmatory positive blocks. Not an injection procedure but often the next step when RFA fails.
Genicular Nerve RFA for Knee Osteoarthritis
This is one of the clearest success stories in interventional pain. Patients with severe knee osteoarthritis who are not surgical candidates — because they are too frail, too young, too heavy, refuse arthroplasty, or have already maxed out conservative care — can get durable relief from radiofrequency ablation of the superior medial, superior lateral, and inferior medial genicular nerves that innervate the knee capsule.
The procedure is short (30–45 minutes), outpatient, uses fluoroscopic or ultrasound guidance, and is typically preceded by a diagnostic genicular nerve block to confirm responders. Randomized trials show roughly 50% pain reduction at 6 months in more than half of treated patients, with effects often lasting 9–12 months before the nerves regenerate. Cooled RFA (which creates a larger lesion) has slightly better durability than conventional thermal RFA in several trials. The procedure is covered by most insurers when conservative care has failed. See the arthritis overview for where genicular RFA fits in osteoarthritis management.
Trigger Point Injections
Trigger points are palpable, tender nodules within a taut band of skeletal muscle that reproduce the patient's pain when pressed. They are classic in myofascial pain, tension-type headache, trapezius and quadratus lumborum pain, and the cervical and thoracic paraspinals after whiplash. The injection can use local anesthetic alone (lidocaine or bupivacaine), local anesthetic with corticosteroid, or be performed as dry needling without any medication — the mechanical disruption of the taut band appears to matter more than whatever is in the syringe.
Evidence is modest and mostly short-term. Trigger point injections are cheap, low-risk, and occasionally dramatically effective, but they are not a durable fix in isolation. The right use is as an adjunct to targeted physical therapy and postural correction — the injection breaks a pain cycle long enough for movement-based treatment to take hold. If trigger points recur in the same location repeatedly, the underlying mechanical or postural driver needs attention, not more needles.
Sympathetic Blocks — Stellate & Lumbar
The sympathetic nervous system — a parallel chain of ganglia running alongside the spine — drives the "sympathetically maintained" component of some neuropathic pain syndromes, most classically Complex Regional Pain Syndrome (CRPS). Anesthetizing the sympathetic chain can produce striking, sometimes dramatic relief.
- Stellate ganglion block. The stellate (cervicothoracic) ganglion sits at the base of the neck and supplies sympathetic fibers to the head, neck, and upper extremity. Image-guided injection of local anesthetic can relieve upper-extremity CRPS, post-herpetic neuralgia of the face or arm, and refractory hot flashes. A successful block produces a transient Horner syndrome (drooping eyelid, small pupil, warm flushed face) on the injected side — that is the expected sign, not a complication. Stellate blocks are also used off-label for PTSD with a growing but still debated evidence base.
- Lumbar sympathetic block. Targets the lumbar sympathetic chain at L2–L4 anterolateral to the vertebral bodies. Used for lower-extremity CRPS, refractory peripheral vascular pain, and some cases of lower limb painful peripheral neuropathy. A successful block produces warming and pinking of the treated leg from restored blood flow.
A series of 3–6 blocks over weeks is typical when the first block produces meaningful relief. Blocks are diagnostic as well as therapeutic — a non-responder tells you the pain is not sympathetically maintained, redirecting care toward somatic or central mechanisms. See the CRPS article for how sympathetic blocks fit into the broader treatment ladder.
Celiac Plexus Block for Abdominal Pain
The celiac plexus is a dense sympathetic network in the upper retroperitoneum that carries nociceptive signals from the pancreas, liver, gallbladder, stomach, and proximal small bowel. Blocking it with local anesthetic (diagnostic or therapeutic) or destroying it with alcohol or phenol (neurolysis) is a mainstay for pain from pancreatic cancer and chronic pancreatitis. In pancreatic cancer pain, randomized trials show that celiac plexus neurolysis reduces opioid requirements and improves quality of life. For benign chronic pancreatitis, results are more variable but still meaningful in selected patients. The block is performed under fluoroscopic, CT, or endoscopic-ultrasound (EUS) guidance; the EUS approach is increasingly standard because it avoids a posterior needle path near the aorta. Transient orthostatic hypotension and diarrhea are common post-procedure and expected — they reflect successful sympathetic interruption.
Intrathecal Drug Delivery Pumps
An intrathecal pump is an implanted, programmable reservoir that infuses tiny amounts of medication directly into the cerebrospinal fluid via a catheter threaded into the spinal subarachnoid space. Because the drug bypasses the blood-brain barrier, the intrathecal dose is a small fraction of the equivalent oral dose — an oral-to-intrathecal morphine conversion is roughly 300:1. The practical effect is powerful analgesia at doses far below systemic toxicity thresholds.
The two mainstay medications are morphine (the only FDA-approved intrathecal opioid) and ziconotide (Prialt), a synthetic snail-venom peptide that blocks N-type calcium channels and is non-opioid. Ziconotide is approved for severe chronic pain refractory to conventional therapy; it does not cause tolerance or respiratory depression but has a narrow therapeutic window and can cause psychiatric side effects including hallucinations, confusion, and rarely suicidal ideation. Off-label intrathecal agents include bupivacaine, clonidine, and hydromorphone.
Pumps are reserved for patients with severe, refractory cancer pain or carefully selected non-cancer pain (failed back surgery syndrome, severe spasticity) who have failed or cannot tolerate oral therapy. Cost is substantial — device and implantation run $30,000–$60,000, with ongoing refill costs. A trial (single intrathecal dose or short-term external catheter) precedes permanent implantation. Failure modes include catheter kinking, pump malfunction, granuloma at the catheter tip (particularly with high-dose morphine), and infection.
Spinal Cord Stimulators
Spinal cord stimulation (SCS) implants thin wire electrodes in the epidural space over the dorsal columns of the spinal cord. A pulse generator (battery pack) implanted in the buttock or flank delivers electrical pulses that modulate pain signaling. The original rationale was Melzack and Wall's gate-control theory — stimulating large fibers "closes the gate" on small pain fibers — though the real mechanisms are more complex and involve changes in spinal interneurons, descending modulation, and possibly cortical reorganization.
SCS is a two-stage process. First comes a percutaneous trial: temporary leads are placed through a needle in the epidural space and connected to an external generator for 5–10 days. If the patient experiences ≥50% pain reduction and improved function during the trial, they proceed to permanent implantation with a surgically anchored lead and subcutaneous pulse generator. The trial is essential — it weeds out non-responders before the expensive, surgical implant.
Several waveform platforms exist and matter for outcomes:
- Tonic stimulation (traditional). Produces a paresthesia (tingling) in the painful area. Most-studied, longest history. Some patients find the paresthesia itself bothersome.
- Burst stimulation (DeRidder). Paresthesia-free. Delivers clusters of pulses mimicking cortical burst firing. Often tried when tonic stimulation loses efficacy.
- High-frequency 10 kHz (HF-10, Nevro Senza). Paresthesia-free, randomized trial (SENZA-RCT) showed superiority to tonic stimulation for back and leg pain at 24 months. The flagship platform for failed back surgery syndrome.
- Closed-loop (Saluda Evoke). Measures the electrical response from the spinal cord (ECAPs) in real time and adjusts stimulation dynamically with posture changes. Aims to keep dosing in the therapeutic window regardless of body position.
Best indications: failed back surgery syndrome (persistent radicular leg pain after lumbar spine surgery), painful diabetic neuropathy refractory to medications, CRPS, refractory angina, and ischemic limb pain. Randomized trials show about half to two-thirds of appropriately selected patients have >50% pain reduction at one year, with benefit persisting in many out to 5 years. See the peripheral neuropathy article for SCS in diabetic neuropathy.
Costs and failure modes. Device plus implantation runs $40,000–$80,000 in the U.S., typically covered by insurance when conservative care has failed and the trial is positive. Common failures: lead migration requiring revision, loss of efficacy over time (the "accommodation" problem), infection (1–4%), and explantation rates that reach 20% at five years in some series. SCS is a tool, not a cure.
Dorsal Root Ganglion (DRG) Stimulation
The dorsal root ganglion is a cluster of sensory-neuron cell bodies sitting just outside the spinal cord at each level, inside the neural foramen. DRG stimulation (Abbott Proclaim DRG, formerly St. Jude Axium) places leads directly on the DRG rather than over the dorsal columns. Because each DRG maps to a specific dermatome, DRG stim is uniquely effective for focal neuropathic pain in small, hard-to-cover territories: the foot, groin, post-surgical scar pain, and especially CRPS of the lower extremity.
The ACCURATE trial compared DRG stimulation to traditional SCS for CRPS and peripheral causalgia below the waist. DRG produced superior pain relief at 3 and 12 months. For the right indication — focal neuropathic pain with a clean dermatomal pattern — DRG stim is often the first-line neuromodulation choice. It is less well-suited to broad axial pain or multi-dermatomal distributions.
Peripheral Nerve Stimulators
Peripheral nerve stimulation (PNS) places a small electrode directly along a named peripheral nerve — for example, the suprascapular nerve for shoulder pain, the genicular nerves for knee pain, the occipital nerves for occipital neuralgia, or a truncal nerve for post-thoracotomy pain. Two paradigms exist:
- Temporary 60-day PNS (SPRINT). A percutaneous lead is placed under ultrasound guidance and removed after 60 days of stimulation. Surprising durable benefit in many patients — thought to reflect central neuroplastic changes. Attractive because nothing stays implanted long-term.
- Permanent PNS. Implanted systems (StimRouter, Nalu) for patients who respond to a trial but need ongoing stimulation.
PNS is a rapidly growing space, and device platforms change every couple of years. For focal, anatomically definable peripheral nerve pain — particularly post-surgical or post-traumatic — PNS is often a better first neuromodulation choice than SCS.
Vertebroplasty & Kyphoplasty
Osteoporotic vertebral compression fractures cause severe, localized, position-sensitive back pain that worsens with weight-bearing. Most heal with 6–12 weeks of conservative care (pain control, bracing, careful mobilization). A minority do not — pain persists, the vertebra continues to collapse, or the patient cannot tolerate bedrest. In those cases:
- Vertebroplasty. Percutaneous injection of polymethylmethacrylate (PMMA) bone cement into the fractured vertebral body under fluoroscopy. Stabilizes the fracture and often produces immediate pain relief.
- Kyphoplasty. A balloon tamp is inflated inside the vertebra first, creating a cavity and partially restoring vertebral height; cement is then injected into the cavity. Lower cement-leak rates than vertebroplasty and some height restoration.
The evidence controversy. Two 2009 sham-controlled trials (INVEST, Buchbinder) showed no benefit over sham, and vertebroplasty nearly collapsed. Subsequent better-selected trials (VERTOS IV, VAPOUR) in acute, highly painful fractures showed significant benefit. The current consensus: vertebroplasty and kyphoplasty help acute (under 6 weeks) fractures with severe pain in patients who fail conservative care. They do not help old, healed fractures, and they do not prevent adjacent-level fractures — in fact, adjacent-level fracture risk may increase because the cemented vertebra is now stiffer than its neighbors. Long-term fracture prevention requires treating the underlying osteoporosis aggressively. See the osteoporosis article and fracture prevention article.
Selection Criteria & Failure Modes
Who benefits from interventional pain procedures? The short version: patients with a clearly identified anatomical or physiological pain generator, failed first-line conservative care, and realistic expectations. Patients do worse when:
- The pain is predominantly nociplastic/centralized. Widespread, fluctuating pain with high tender-point counts, poor sleep, and prominent fatigue typically does not have a single local generator to inject or ablate. These patients need the approach outlined in the central sensitization article and pain reprocessing therapy article.
- Untreated depression, anxiety, or catastrophizing dominates the clinical picture. Psychological screening before neuromodulation trials substantially predicts outcome. Treat the mood disorder first.
- Active opioid use disorder or uncontrolled substance use. Most programs defer device implantation until the substance issue is stabilized. See the opioids article.
- Unrealistic expectations. "Fix me so I can stop taking everything" is a red flag. Interventional procedures reduce pain — they rarely eliminate it, and they rarely replace an entire medication plan.
- Smoking, uncontrolled diabetes, or active infection. Surgical-site infection risk goes up substantially with neuromodulation implants.
Common failure modes to know about as a patient:
- Epidural steroid tachyphylaxis — each successive injection often works less well and less long than the last.
- False-positive single blocks — placebo response and non-specific anesthetic spread can fool a single diagnostic block. Insist on two concordant blocks before RFA.
- Lead migration in SCS — especially in the early weeks, and in active patients. Revision is common.
- Accommodation/tolerance in neuromodulation — stimulator efficacy can wane over 1–3 years; reprogramming or waveform switching sometimes helps.
- Steroid side effects — repeated epidural or joint corticosteroid injections raise blood sugar in diabetics, cause facial flushing, and may contribute to bone loss and adrenal suppression over time. Limit cumulative exposure.
The best interventional pain physicians work inside a team — physical therapy, pain psychology, primary care, surgery — and treat procedures as one lever among several. Before agreeing to any significant intervention, ask: what is the realistic best-case outcome? What fraction of patients like me get that outcome? What are the alternatives? What do you recommend I do with or without this procedure for exercise, sleep, and medication? A good answer to those questions is more important than the specific device or technique.
Key Research Papers
- Chou R, et al. Epidural corticosteroid injections for radiculopathy and spinal stenosis: a systematic review and meta-analysis. Ann Intern Med. 2015.
- Davis T, et al. Prospective, multicenter, randomized, crossover clinical trial comparing the safety and effectiveness of cooled radiofrequency ablation with corticosteroid injection for knee osteoarthritis pain. Reg Anesth Pain Med. 2018.
- Deer TR, et al. ACCURATE study: Dorsal root ganglion stimulation yielded higher treatment success rate for CRPS and causalgia at 3 and 12 months. Pain. 2017.
- Kapural L, et al. Novel 10-kHz high-frequency therapy (HF10 therapy) is superior to traditional low-frequency spinal cord stimulation for the treatment of chronic back and leg pain (SENZA-RCT). Anesthesiology. 2015.
- Kallmes DF, et al. A randomized trial of vertebroplasty for osteoporotic spinal fractures (INVEST). N Engl J Med. 2009.
- Clark W, et al. Safety and efficacy of vertebroplasty for acute painful osteoporotic fractures (VAPOUR): a multicentre, randomised, double-blind, placebo-controlled trial. Lancet. 2016.
- Wong GY, et al. Effect of neurolytic celiac plexus block on pain relief, quality of life, and survival in patients with unresectable pancreatic cancer: a randomized controlled trial. JAMA. 2004.
- Deer TR, et al. The Polyanalgesic Consensus Conference (PACC): Recommendations on intrathecal drug infusion systems best practices and guidelines. Neuromodulation. 2017.
- Manchikanti L, et al. A systematic review and best evidence synthesis of the effectiveness of therapeutic facet joint interventions in managing chronic spinal pain. Pain Physician. 2015.
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on interventional pain procedures and neuromodulation:
- Transforaminal epidural steroid injections for radiculopathy
- Medial branch blocks and facet radiofrequency ablation
- Sacroiliac joint RFA and lateral branch neurotomy
- Genicular nerve RFA for knee osteoarthritis
- Stellate ganglion block and CRPS
- Celiac plexus neurolysis for pancreatic cancer pain
- Intrathecal ziconotide and morphine pump therapy
- Spinal cord stimulation for failed back surgery syndrome
- High-frequency 10 kHz spinal cord stimulation
- Dorsal root ganglion stimulation for CRPS
- Peripheral nerve stimulation for chronic pain
- Vertebroplasty and kyphoplasty for compression fractures
Connections
- Complex Regional Pain Syndrome
- Chronic Low Back Pain
- Peripheral Neuropathy
- Arthritis
- Osteoporosis
- Central Sensitization and Nociplastic Pain
- Pain Reprocessing Therapy
- Opioids for Chronic Pain
- Fracture Prevention and Fall Risk
- Chronic Pain Overview
- Non-Opioid Medications
- Pain Mechanisms
- Sleep and Lifestyle for Chronic Pain
- Depression
- Sciatica
- Fibromyalgia