Complex Regional Pain Syndrome (CRPS / RSD)
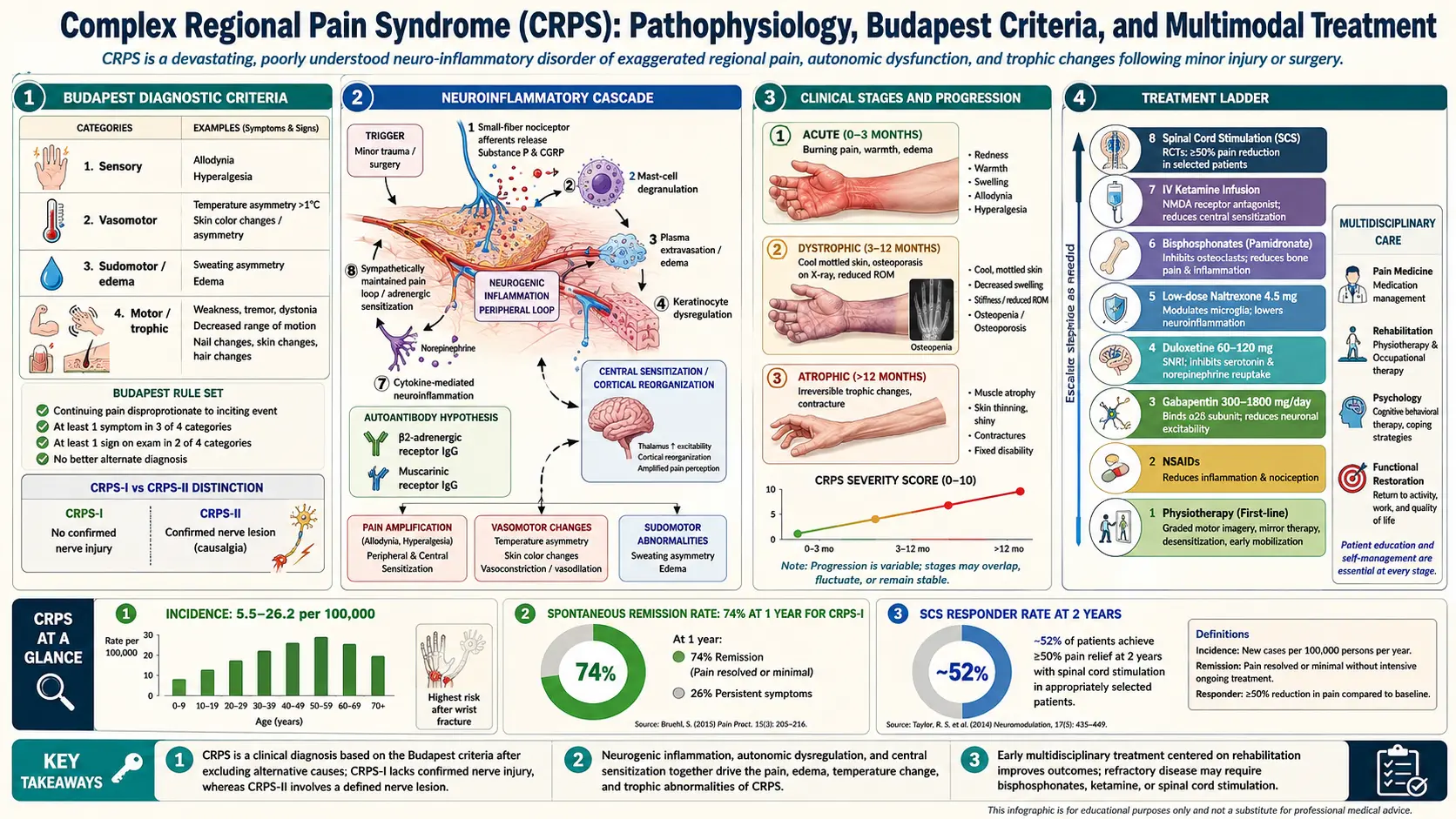
Table of Contents
- What CRPS Is
- Type I vs Type II
- Typical Triggers
- Budapest Diagnostic Criteria
- Pathophysiology — What Goes Wrong
- Stages and Natural History
- Early Mobilization — First-Line Treatment
- Graded Motor Imagery and Mirror Therapy
- Medications That Help
- Bisphosphonates (Pamidronate, Neridronate)
- Ketamine Infusions
- Sympathetic Nerve Blocks
- Spinal Cord and Dorsal Root Ganglion Stimulation
- Refractory CRPS — When Nothing Works
- Vitamin C Prophylaxis for Distal Radius Fracture
- Living With CRPS
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
What CRPS Is
Complex Regional Pain Syndrome (CRPS) — still called Reflex Sympathetic Dystrophy (RSD) in older charts and among long-time patients — is a chronic pain disorder that usually starts in a single limb after a relatively minor injury. The defining feature is a mismatch between the injury and the response: a wrist fracture that should heal in eight weeks instead triggers burning pain, swelling, color and temperature changes, sweating, hair and nail changes, and eventually weakness and stiffness that persist for months or years.
CRPS is uncommon but not rare. The best epidemiology, from a Dutch population study, puts the incidence at about 26 cases per 100,000 person-years, with women affected roughly three times more often than men. The upper limb is involved about twice as often as the lower limb. Most cases follow a fracture, sprain, surgery, or crush injury — but roughly 10% have no identifiable trigger at all.
If you are reading this because you or someone you love was just told they "might have CRPS," two things matter most: the earlier it is treated, the better it does, and the treatment that works best is active movement, not rest, not immobilization, and not opioids. The rest of this article explains why.
Type I vs Type II
CRPS is split into two subtypes by whether a nerve injury can be identified:
- CRPS Type I (formerly RSD). No demonstrable nerve injury. The inciting event is tissue damage — a fracture, a sprain, a bruise, a surgery — without a named nerve having been cut or compressed. About 90% of CRPS cases are Type I.
- CRPS Type II (formerly causalgia). A specific peripheral nerve injury can be identified — for example, a severed median nerve during carpal tunnel release, or a traction injury to the brachial plexus. The pain and autonomic changes behave the same way clinically; the only difference is the identifiable nerve lesion.
The distinction matters for insurance coding and for surgical planning (Type II occasionally responds to nerve repair) but not for most day-to-day treatment. Both subtypes are managed the same way: early mobilization, graded sensory and motor rehabilitation, and a stepwise ladder of medications and procedures.
Typical Triggers
CRPS almost always follows something. The most common triggers, in rough order:
- Distal radius (wrist) fracture — the single most studied trigger, and the setting where vitamin C prophylaxis has been tested (see below). Roughly 3–7% of wrist fractures develop CRPS.
- Foot and ankle sprains or fractures, including lateral ankle sprains that seemed trivial at the time.
- Elective surgery on a limb — carpal tunnel release, trigger finger release, ankle arthroscopy, knee replacement, bunion correction.
- Crush injuries and soft-tissue trauma without fracture.
- Prolonged immobilization — casting or splinting for longer than necessary appears to increase risk in susceptible people.
- Stroke, myocardial infarction, or spinal cord injury — "shoulder-hand syndrome" is a CRPS-like presentation after stroke.
- No identifiable trigger in roughly 10% of cases.
The size of the trigger does not predict the size of the response. A sprained ankle can produce severe, treatment-resistant CRPS; a shattered tibia often heals without any CRPS at all. Something about the individual nervous system — genetics, prior central sensitization, inflammation profile, psychological state at the time of injury — tips the balance.
Budapest Diagnostic Criteria
CRPS has no blood test and no imaging finding that confirms it. Diagnosis is clinical, using the Budapest Criteria adopted by the International Association for the Study of Pain (IASP) in 2007. To meet criteria, a patient must have:
- Continuing pain that is disproportionate to any inciting event.
- At least one symptom reported by the patient in three of four categories:
- Sensory: hyperesthesia (painfully heightened sensation) or allodynia (pain from a non-painful touch, like a bedsheet or a breeze).
- Vasomotor: temperature asymmetry, skin color changes, or color asymmetry between the two limbs.
- Sudomotor / edema: swelling, sweating changes, or sweating asymmetry.
- Motor / trophic: decreased range of motion, weakness, tremor, dystonia, or trophic changes (skin, hair, or nails looking abnormal).
- At least one sign observed by the clinician at the time of evaluation in two or more of the same four categories.
- No other diagnosis better explains the signs and symptoms.
The Budapest Criteria are strict on purpose. Earlier "IASP 1994" criteria were so loose that roughly 40% of people diagnosed with CRPS did not actually have it. The Budapest revision keeps sensitivity above 85% while pushing specificity close to 70%. For research, an even stricter "research Budapest" version requires symptoms in all four categories and signs in two or more.
A few practical notes for patients. First, the signs — especially temperature and color changes — can wax and wane, and a single normal exam at the orthopedist's office does not rule out CRPS. Bring photos taken during a flare. Second, triple-phase bone scintigraphy can support the diagnosis in early CRPS by showing periarticular uptake, but a normal scan does not exclude it. Third, sympathetic nerve blocks (discussed below) are a treatment, not a diagnostic test — response or non-response does not confirm or rule out CRPS.
Pathophysiology — What Goes Wrong
CRPS is the clearest clinical example in medicine of a nervous system that has learned pain and cannot unlearn it. Five overlapping mechanisms drive the syndrome:
Peripheral sensitization and neurogenic inflammation
After the inciting injury, C-fibers in the affected limb become hyperexcitable. They release substance P, calcitonin gene-related peptide (CGRP), and other neuropeptides into the local tissue, causing vasodilation, plasma extravasation, and swelling — the warm, red, puffy early-stage limb. This is called neurogenic inflammation, and it is why NSAIDs and ice usually do little.
Small-fiber nerve loss
Skin biopsies from CRPS limbs show a measurable reduction in small-fiber nerve density compared with the unaffected limb — a partial small-fiber neuropathy confined to the region of pain. This explains the mixed picture of numbness plus hypersensitivity: the large fibers still work, but the small fibers that carry temperature and pain are damaged and misfiring.
Central sensitization
Repeated input from the inflamed limb rewires the spinal cord dorsal horn. NMDA receptors become hyperactive, GABA-mediated inhibition weakens, and non-painful inputs (light touch, normal joint movement) start being interpreted as pain. This is the same central sensitization process that drives fibromyalgia, nociplastic pain, and chronic low back pain — which is why ketamine (an NMDA antagonist) and gabapentinoids (which dampen excitatory signaling) sometimes help.
Immune dysregulation
CRPS patients have elevated circulating pro-inflammatory cytokines (TNF-alpha, IL-1, IL-6) and, in many cases, autoantibodies against autonomic nervous system targets such as the beta-2 adrenergic receptor and the muscarinic-2 receptor. These antibodies can cause vasomotor and sudomotor dysfunction when passively transferred to animals. The implication is that at least a subset of CRPS is a low-grade autoimmune disorder — which is why IVIG and plasmapheresis have been trialed, with modest and inconsistent results.
Cortical reorganization
Functional MRI studies show that the cortical map of the affected limb shrinks in the primary somatosensory cortex, and the brain's representation of the limb becomes distorted. Patients often describe the affected hand or foot as feeling "not mine," swollen when it isn't, or misshapen. Graded motor imagery and mirror therapy are designed specifically to reverse this cortical remapping — and they work better than almost anything else we have.
Stages and Natural History
Older textbooks describe three sequential stages (warm, dystrophic, atrophic). Modern research has shown the staged model is misleading — CRPS does not march predictably through phases, and many patients cycle between "warm" and "cold" presentations without ever entering a truly atrophic stage. What is more useful:
- Warm CRPS: red, hot, swollen, usually within the first 6–12 months. Most responsive to treatment.
- Cold CRPS: cool, bluish or mottled, often drier. Associated with longer duration, more central sensitization, and worse prognosis.
Prognosis varies widely. In population-based cohorts, roughly 70% of patients have substantial recovery within the first year with appropriate treatment. The remaining 20–30% develop persistent or progressive disease. The single strongest predictor of a bad outcome is delayed diagnosis and delayed mobilization. Weeks lost to casting a painful limb or to opioid-only management add up to months of rehabilitation later.
Early Mobilization — First-Line Treatment
This is the single most important sentence in this article: the treatment for CRPS is to move the limb, gently, early, and daily, even when it hurts. Immobilization, guarding, and "resting until the pain settles" make CRPS worse. Movement — graded, paced, guided by a physical or occupational therapist familiar with CRPS — is what retrains the nervous system.
A good early-stage program looks like this:
- Desensitization: systematic exposure to textures (silk, cotton, terrycloth, corduroy) for a few minutes several times a day, starting with whatever the skin tolerates and progressing over weeks.
- Active range-of-motion within the pain-tolerable range, every day, multiple times a day. Passive stretching by a therapist is kept minimal early on — patient-driven movement teaches the brain that the limb is safe to use.
- Weight-bearing (for leg CRPS) in a graded way: standing, then stepping, then walking, with a walker or crutch if needed. Staying non–weight-bearing past the orthopedic healing point is harmful.
- Edema management with elevation, compression garments, and contrast baths (alternating warm and cool water).
- Pacing — doing a little, consistently, rather than pushing through a 30-minute session and crashing for three days.
If the pain is severe enough that no movement is possible at all, the first job of medical treatment (nerve blocks, ketamine, medications) is to open a window so physical therapy can happen. The drugs and procedures do not cure CRPS. Movement, repeated many thousands of times, is what cures CRPS — the drugs just make the movement possible.
Graded Motor Imagery and Mirror Therapy
Graded Motor Imagery (GMI) is a three-stage cognitive rehabilitation program developed specifically for CRPS and phantom limb pain. Randomized controlled trials by Moseley and colleagues show clinically meaningful reductions in pain and disability. The three stages:
- Left/right discrimination. The patient looks at photographs of hands or feet and decides, as quickly as possible, whether each image shows a left or a right. In CRPS, this ability is measurably slower on the affected side. Practicing reboots the brain's internal model of the limb.
- Motor imagery. The patient imagines performing movements with the affected limb — without actually moving it. Imagined movement activates the same motor cortex regions as real movement, without provoking peripheral pain.
- Mirror therapy. The patient places a mirror vertically along the body's midline, reflecting the unaffected limb so that it appears to be in the position of the affected limb. Watching the reflection move convinces the brain that the painful limb is moving normally — and cortical pain signaling often drops within minutes.
GMI costs almost nothing, has no side effects, and can be done at home. The Recognise app (NOI Group) is the standard tool for the left/right discrimination stage. A cheap flat mirror works for stage three. Patients typically spend 10–15 minutes at each session, several times a day, progressing through the stages over weeks to months. If a therapist in your area has been trained in GMI, that is worth the visit; if not, the protocol can be taught from the published literature.
Medications That Help
No drug is specifically FDA-approved for CRPS. The evidence base is built on small trials, case series, and extrapolation from other neuropathic pain syndromes. The most reasonable first-line medical options:
- Gabapentin (typically titrated to 1800–3600 mg/day in divided doses) or pregabalin (150–600 mg/day). Dampens central sensitization. See the gabapentinoid article.
- Amitriptyline (10–75 mg at bedtime) or duloxetine (30–60 mg/day). Tricyclics and SNRIs have the best neuropathic-pain evidence overall.
- Low-dose naltrexone (1.5–4.5 mg at bedtime) for the glial-modulation and nociplastic component. Case series only, but cheap and benign.
- Topical lidocaine 5% patches or capsaicin 8% patches for focal allodynia.
- Prednisone taper in the first weeks (for example, 30 mg/day tapering over 2–4 weeks) can reduce the early neurogenic-inflammatory component in warm CRPS.
Opioids are not a good chronic treatment for CRPS. They do not reverse central sensitization, they blunt motivation for the rehabilitation that does work, and they can actually worsen the syndrome through opioid-induced hyperalgesia. Short courses around surgery or during severe flares are reasonable; months-long daily opioid use is not. See the opioid article for the full argument.
Bisphosphonates (Pamidronate, Neridronate)
Bisphosphonates — drugs normally used for osteoporosis — have the strongest randomized evidence of any medication for early CRPS. The landmark trial, published by Varenna and colleagues in 2013, gave neridronate 100 mg IV on days 1, 4, 7, and 10 to patients with CRPS of the foot or hand of less than four months' duration. Active treatment produced a roughly 50% reduction in pain and a sustained effect at one year, compared to near-zero change with placebo.
Neridronate is not available in the United States but is used routinely for CRPS in Italy and parts of Europe. The closest U.S. equivalent is intravenous pamidronate (typical regimens: 60 mg IV over 2–4 hours, with a second dose often given at 4–8 weeks) or oral alendronate (40 mg/day for 8 weeks in one small trial). Side effects include an acute-phase reaction (fever, flu-like symptoms, bone pain) in the first 48 hours after infusion, occasional eye inflammation, and, rarely with extended use, osteonecrosis of the jaw and atypical femoral fractures.
Practical points. Bisphosphonates work best when started within the first year of symptoms — they mainly affect the warm, bone-edema-predominant phase. Pre-treatment dental exam is standard. Calcium and vitamin D should be optimized. Insurance coverage for bisphosphonates in CRPS is off-label in the U.S.; documentation of a Budapest-criteria diagnosis and of failed first-line therapy helps with prior authorization.
Ketamine Infusions
Ketamine is an NMDA receptor antagonist that, at sub-anesthetic doses, can interrupt central sensitization. Two delivery models exist:
- Outpatient low-dose infusions: 0.25–0.5 mg/kg/hour for 4 hours a day, repeated for 5–10 consecutive weekdays. This is the most common U.S. protocol. Roughly half of CRPS patients report meaningful pain reduction; the effect typically lasts weeks to a few months before boosters are needed.
- Inpatient high-dose / coma-dose protocols (Germany, Mexico): continuous infusion at anesthetic doses for several days. Dramatic responses have been reported, but serious risks — hepatotoxicity, dissociative and psychiatric effects, infection — make this a last-resort option. U.S. insurers do not cover it.
Side effects at the low-dose outpatient level are usually manageable: dissociation and "K-hole" sensations during infusion, nausea, transient blood-pressure elevation. A trained anesthesiologist or pain physician monitors vital signs and offers a benzodiazepine if dysphoria becomes intolerable. Cost in the U.S. typically runs $500–$1,500 per infusion and is poorly covered by insurance — ask the clinic for a superbill and prepare an appeal. Response is most likely in patients with strong central-sensitization features (widespread allodynia, hyperalgesia, bilateral spread).
Sympathetic Nerve Blocks
The sympathetic nervous system is entangled with CRPS — the older name "reflex sympathetic dystrophy" reflects the hypothesis that sympathetic efferents drive the pain. The hypothesis turned out to be partly right: some patients have sympathetically maintained pain, where blocking the sympathetic chain dramatically relieves symptoms; others have sympathetically independent pain, where blocks do nothing. There is no reliable way to predict in advance which type a patient has — you find out by trying one.
- Stellate ganglion block (upper limb CRPS): local anesthetic injected around the stellate ganglion at C6–C7. Done under fluoroscopy or ultrasound. If the first block produces clear pain relief and the characteristic Horner's sign (drooping eyelid, small pupil), a series of 3–6 blocks over several weeks is commonly pursued.
- Lumbar sympathetic block (lower limb CRPS): local anesthetic injected around the L2–L4 sympathetic chain. Same principle, same decision algorithm.
Blocks are low-risk, outpatient, and diagnostic as well as therapeutic — a positive response signals a treatable sympathetic component. Blocks are not a long-term monotherapy. They work best as a window that lets the patient participate in aggressive physical therapy for the days or weeks after each injection. If the first two well-placed blocks do nothing, further blocks are unlikely to help, and the pain is sympathetically independent. See the interventional pain article for the broader landscape.
Spinal Cord and Dorsal Root Ganglion Stimulation
For CRPS that persists despite conservative and medical treatment, neuromodulation is one of the better-evidenced options in chronic pain medicine.
Spinal cord stimulation (SCS) places electrodes in the epidural space over the dorsal columns, connected to an implanted pulse generator. The device delivers electrical pulses that modulate pain signaling. Older "tonic" SCS created a tingling (paresthesia) over the painful area; newer "high-frequency" (10 kHz) and "burst" modes provide relief without paresthesia. The landmark RCT by Kemler et al. in NEJM 2000 demonstrated durable pain relief for CRPS patients who had failed conservative care.
Dorsal root ganglion (DRG) stimulation targets the sensory cell bodies in the DRG rather than the dorsal columns. The ACCURATE trial showed DRG stimulation superior to conventional SCS for CRPS of the lower limb, particularly for focal pain in the foot. For upper-limb CRPS, either modality is reasonable.
Both are done in two stages: a 5–7 day trial with percutaneous leads and an external generator, followed by permanent implantation only if the trial produces at least 50% pain relief. Complications include lead migration, infection (roughly 2–5%), and, rarely, neurological injury. Insurance coverage in the U.S. is generally good once conservative care has been documented to fail. Average total cost (trial plus implant) runs $40,000–$80,000 and is typically covered by commercial plans and Medicare.
Refractory CRPS — When Nothing Works
Roughly one in ten CRPS patients progresses to severe, treatment-resistant disease with dystonia, contracture, or spread to other limbs. Options at this point are limited, experimental, and best pursued at a dedicated CRPS center:
- Intrathecal drug delivery (baclofen for dystonia; ziconotide or opioid for pain).
- IVIG (intravenous immunoglobulin) — small trials suggest benefit in a subset with autoantibody-mediated disease.
- Plasmapheresis — case series only.
- Scrambler therapy (Calmare) — a surface electrical stimulator that provides non-pain signals to the affected dermatomes. Mixed evidence; low risk.
- Intensive multidisciplinary inpatient rehabilitation — 3–4 weeks of combined physical therapy, occupational therapy, psychology, and medication management. This is the single most effective option for severe long-duration CRPS, and the hardest to get insurance to cover.
- Amputation — historically a dead end (pain recurs in the stump or phantom limb in the majority). Considered only for non-functional, infected, or contracted limbs and with realistic expectations.
Psychological care is not optional at this stage — not because CRPS is psychological, but because living with severe refractory pain erodes mood, sleep, and relationships, and treating those makes the pain more tolerable. Cognitive behavioral therapy, acceptance and commitment therapy, and pain reprocessing therapy all have roles.
Vitamin C Prophylaxis for Distal Radius Fracture
This is the one clean piece of prevention evidence in CRPS. In 1999, Zollinger and colleagues published a randomized controlled trial in The Lancet showing that patients given 500 mg of vitamin C daily for 50 days after a wrist fracture had roughly a fourfold reduction in CRPS incidence compared with placebo (about 2.4% versus 10.1%). A larger follow-up trial in 2007 confirmed the benefit at 500 mg/day and showed no additional benefit from 1,500 mg.
On the strength of these trials, several orthopedic societies — including the American Academy of Orthopaedic Surgeons — recommend 500 mg of vitamin C daily for 50 days after distal radius fracture. It is cheap (a bottle of 500 mg tablets costs a few dollars), well tolerated, and has almost no downside other than occasional loose stools or the theoretical concern about kidney stones in stone-formers.
The evidence for vitamin C prophylaxis in other fractures (ankle, hip, foot) is less consistent, but the intervention is cheap enough that many orthopedists prescribe it after any significant extremity fracture. If you are having surgery on a limb and have any CRPS history — or a family member with CRPS — it is reasonable to ask about vitamin C, regional anesthesia instead of general, and shorter rather than longer immobilization.
Living With CRPS
A few things patients consistently report helping, beyond the formal medical pathway:
- Sleep first. Poor sleep amplifies central sensitization more reliably than almost any other variable. See sleep, exercise and lifestyle.
- Clothing and bedding. Soft, seamless fabrics. A bed cradle to keep blankets off a hypersensitive foot. A silk glove sleeve for the hand.
- Heat and cold strategically. Many patients find warmth helps cold CRPS and cool compresses help warm CRPS — test each in a small way before committing.
- A pacing diary. Log activity and pain for two weeks to find the level of movement that does not trigger a two-day flare. That is your baseline. Increase by 10% every one to two weeks.
- Community. The Reflex Sympathetic Dystrophy Syndrome Association (RSDSA) and online CRPS patient communities are one of the better resources for finding CRPS-literate physicians and physical therapists in your area.
- Disability and insurance planning. CRPS is recognized by the Social Security Administration (SSR 03-2p). If the syndrome is limiting work, start documentation early — including photographs of color and swelling changes, physical therapy notes showing objective progress or plateaus, and a rheumatology or pain-medicine provider willing to write a detailed functional statement.
Key Research Papers
- Harden RN, et al. Proposed new diagnostic criteria for complex regional pain syndrome. Pain Med. 2007. (The Budapest Criteria.)
- Varenna M, et al. Treatment of complex regional pain syndrome type I with neridronate: a randomized, double-blind, placebo-controlled study. Rheumatology. 2013.
- Zollinger PE, et al. Effect of vitamin C on frequency of reflex sympathetic dystrophy in wrist fractures: a randomised trial. Lancet. 1999.
- Zollinger PE, et al. Can vitamin C prevent complex regional pain syndrome in patients with wrist fractures? A randomized, controlled, multicenter dose-response study. J Bone Joint Surg Am. 2007.
- Kemler MA, et al. Spinal cord stimulation in patients with chronic reflex sympathetic dystrophy. N Engl J Med. 2000.
- Deer TR, et al. Dorsal root ganglion stimulation yielded higher treatment success rate for complex regional pain syndrome and causalgia: the ACCURATE randomized comparative trial. Pain. 2017.
- Moseley GL. Graded motor imagery for pathologic pain: a randomized controlled trial. Neurology. 2006.
- Sigtermans MJ, et al. Ketamine produces effective and long-term pain relief in patients with complex regional pain syndrome type 1. Pain. 2009.
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on CRPS diagnosis, pathophysiology, and management:
- Budapest criteria and CRPS diagnosis
- CRPS pathophysiology and mechanisms
- Graded motor imagery and mirror therapy for CRPS
- Bisphosphonates for CRPS (neridronate, pamidronate)
- Ketamine infusion for CRPS
- Sympathetic blocks (stellate, lumbar) for CRPS
- Spinal cord and dorsal root ganglion stimulation for CRPS
- Vitamin C prophylaxis for CRPS after wrist fracture
Connections
- Interventional Pain
- Fibromyalgia
- Central Sensitization and Nociplastic Pain
- Non-Opioid Medications
- Pain Reprocessing Therapy
- Sleep and Lifestyle for Chronic Pain
- Opioids for Chronic Pain
- Chronic Low Back Pain
- Vitamin C
- Chronic Pain Overview
- Pain Mechanisms
- Peripheral Neuropathy
- Arthritis
- Depression
- Chronic Fatigue Syndrome
- Low-Dose Naltrexone for Fibromyalgia
- Low Dose Naltrexone