Opioids for Chronic Non-Cancer Pain
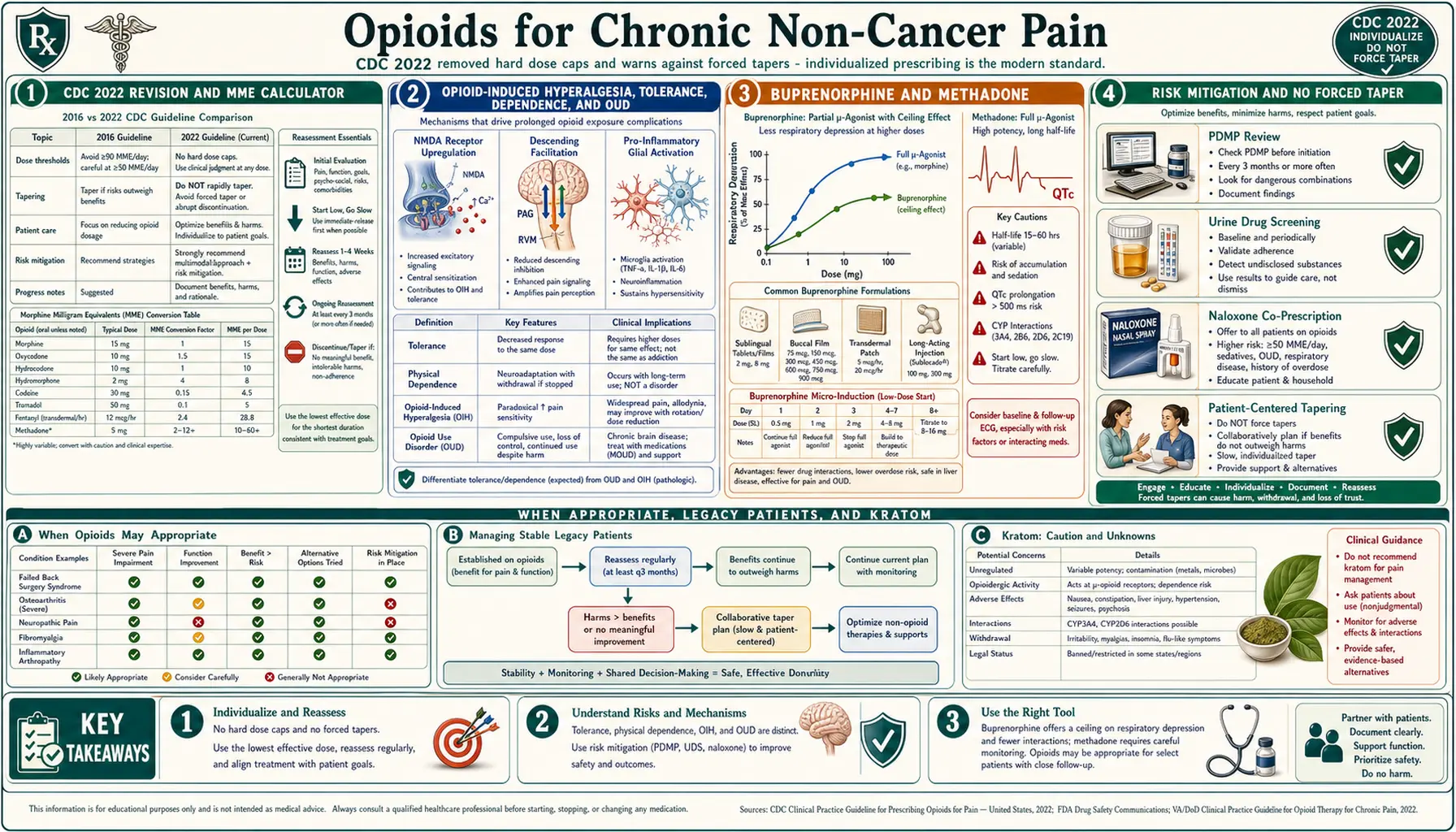
Table of Contents
- An Honest Starting Point
- The CDC 2022 Revision — What Changed
- MME: What It Means and How to Calculate It
- Opioid-Induced Hyperalgesia
- Tolerance vs Physical Dependence vs OUD
- Buprenorphine for Chronic Pain
- Methadone — Powerful and Complicated
- Long-Acting vs Short-Acting Opioids
- Risk Mitigation: PDMP, UDS, Naloxone, REMS
- Tapering — The VA Approach and the No-Forced-Taper Rule
- When Opioids ARE Appropriate
- Legacy Patient Management
- Kratom: What We Know
- Practical Advice for Patients
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
An Honest Starting Point
Opioids for chronic non-cancer pain is one of the most politicized topics in medicine. In the space of a decade, the pendulum swung from aggressive prescribing (the "pain as fifth vital sign" era) to a panicked retreat in which stable, functioning patients were abruptly cut off from medications they had taken safely for years. Both extremes have harmed people. The goal of this page is neither to cheerlead for opioids nor to demonize them, but to describe how thoughtful pain physicians and the current guidelines think about them in 2026.
If you are a patient reading this, a few things to hold in mind: opioids are a real tool with real benefits for a real subset of chronic-pain conditions. They are also a real risk. The evidence base for long-term opioid therapy (more than 3 months) in chronic non-cancer pain is weaker than most people assume. But the evidence against them, at doses that worked for you for years, is not strong enough to justify tearing up a working regimen. Individualization — informed by modern guidelines, honest risk assessment, and shared decision-making — is the entire game.
The CDC 2022 Revision — What Changed
The original 2016 CDC Guideline for Prescribing Opioids for Chronic Pain was a well-intentioned document that was widely misapplied. Insurers, pharmacies, state medical boards, and even some physicians treated its suggested 90 MME/day threshold as a hard ceiling and its cautions as mandates. The result: forced tapers, abandoned legacy patients, suicides, and a measurable jump in street-fentanyl overdose deaths as cut-off patients turned to the illicit market.
In November 2022 the CDC issued a substantially revised Clinical Practice Guideline for Prescribing Opioids for Pain. The top-line changes:
- No hard dose caps. The 90 MME/day "ceiling" was removed. The guideline now instructs clinicians to individualize dosing based on benefit and harm, not a number.
- No hard duration limits for acute pain. The 2016 "three-day supply" suggestion for acute pain was removed.
- Explicit warning against abrupt taper or discontinuation. The guideline now states that clinicians should not abruptly taper or discontinue opioids in patients who are stable on long-term therapy unless there is a clear safety indication, and that forced tapers are associated with serious patient harm including overdose and suicide.
- Broader scope. The revision covers acute, subacute, and chronic pain (the 2016 version was chronic-pain-only), and explicitly exempts cancer, sickle-cell, palliative, and end-of-life care.
- Shared decision-making. The guideline emphasizes that opioids, non-opioids, and non-drug therapies all have trade-offs and that the patient's values and function matter.
Critically, the CDC guideline is guidance, not law. State laws, DEA regulations, insurer policies, and pharmacy corporate rules operate independently and often lag the science. Patients and clinicians may still encounter providers who quote the 2016 caps as if nothing changed.
MME: What It Means and How to Calculate It
MME stands for Morphine Milligram Equivalents. It is a rough attempt to compare different opioids on a single scale by converting each drug's daily dose into "how much oral morphine would that equal?" The conversion factors (as used by the CDC and most state PDMPs):
- Morphine (oral) — 1× (baseline)
- Hydrocodone — 1×
- Oxycodone — 1.5×
- Hydromorphone — 4×
- Oxymorphone — 3×
- Tapentadol — 0.4× (the CDC excludes tapentadol from MME tallies because of its mixed mechanism)
- Fentanyl transdermal patch — roughly (mcg/hr × 2.4) = MME/day
- Methadone — dose-dependent and non-linear: 4× at 1–20 mg/day, 8× at 21–40 mg/day, 10× at 41–60 mg/day, and 12× above that.
- Buprenorphine — intentionally not included in MME calculations in the 2022 CDC revision (partial agonism and ceiling effect make the comparison misleading).
A patient on 60 mg oxycodone/day is at 90 MME. A patient on a 50 mcg/hr fentanyl patch is at roughly 120 MME. A patient on methadone 40 mg/day is at roughly 320 MME, which is why methadone is respected.
Benchmarks to keep in mind (from the revised 2022 CDC guideline, framed as points for re-evaluation, not ceilings):
- Before increasing above 50 MME/day: reassess benefits and harms, document why dose escalation is justified.
- Before increasing above 90 MME/day: proceed with particular caution, involve a pain specialist if possible, and document a clear functional rationale.
- There is no longer a hard cap. Some patients genuinely need more.
Opioid-Induced Hyperalgesia
One of the strangest things long-term opioids can do is make pain worse. Opioid-induced hyperalgesia (OIH) is a paradoxical state in which opioid therapy sensitizes the central nervous system so that the patient becomes more sensitive to painful stimuli, not less. The mechanism involves NMDA receptor upregulation, descending facilitation from the rostroventral medulla, and pro-inflammatory glial activation.
Clinical clues suggesting OIH rather than tolerance:
- Pain that spreads beyond the original site, often becoming diffuse and "whole-body."
- Increased pain with dose escalation rather than improvement.
- New hypersensitivity to light touch, temperature, or pressure.
- Paradoxical improvement when the opioid dose is lowered (not raised).
Tolerance, by contrast, means the same dose produces less analgesia over time; increasing the dose usually restores benefit. OIH and tolerance can coexist and are often difficult to separate clinically, which is one reason a trial of careful dose reduction (or rotation to buprenorphine) is sometimes more useful than escalation.
Tolerance vs Physical Dependence vs OUD
Three terms that get tangled together in public conversation and occasionally even in clinical records. Keeping them separate matters because they have very different implications.
Tolerance is a pharmacological adaptation in which the same dose produces less effect over time. It applies to analgesia, euphoria, sedation, and respiratory depression (though not equally — tolerance to respiratory depression lags behind tolerance to analgesia, which is part of why overdose deaths can happen even in habituated users after a break).
Physical dependence is a predictable physiological state in which abrupt discontinuation produces a withdrawal syndrome: sweating, piloerection, muscle aches, yawning, lacrimation, runny nose, GI cramping, diarrhea, anxiety, restless legs. Physical dependence develops in everyone taking opioids daily for more than a few weeks. It is not addiction. It is not a moral failing. It is the expected biology of mu-opioid receptor adaptation.
Opioid Use Disorder (OUD) is the DSM-5 diagnosis for the behavioral disease of addiction. It is defined by compulsive use despite harm, loss of control, cravings, and a consistent pattern of behaviors: unauthorized dose escalation, doctor-shopping, diversion, use of non-prescribed opioids, use despite clear functional decline, and continued use despite major social, occupational, or legal consequences. The DSM-5 criteria are 11 items, and diagnosis requires 2 or more within 12 months; 2–3 is mild, 4–5 moderate, 6+ severe.
A patient who has been stable on the same opioid dose for five years, takes it as prescribed, functions well, and would go into withdrawal if it stopped is physically dependent, not addicted. Confusing the two is exactly the mistake that drove the forced-taper crisis.
Buprenorphine for Chronic Pain
Buprenorphine deserves its own section because, for chronic pain, it is one of the most underused tools in American medicine. It is a partial mu-opioid agonist with a strong binding affinity, slow receptor dissociation, and a ceiling effect on respiratory depression. In practical terms, that combination makes it measurably safer than full agonists while still providing meaningful analgesia.
Chronic-pain buprenorphine formulations approved in the U.S.:
- Belbuca (buccal film) — dosed in micrograms (75, 150, 300, 450, 600, 750, 900 mcg every 12 hours). Designed explicitly for chronic pain, not OUD.
- Butrans (transdermal patch) — 5, 7.5, 10, 15, 20 mcg/hr, worn for 7 days. Convenient for patients who cannot reliably take oral medications.
- Sublingual buprenorphine (generic tablets and films) — FDA-approved for OUD but commonly used off-label for chronic pain at divided doses (e.g., 2 mg three or four times daily). Much cheaper than Belbuca.
Why buprenorphine is attractive for chronic pain:
- Ceiling on respiratory depression. Full mu-agonists have no ceiling — more drug, more suppression of breathing, eventually death. Buprenorphine's effect on respiration plateaus at higher doses, which is why the overdose profile is fundamentally different.
- Lower euphoria. Partial agonism at the mu receptor produces less of the reinforcing high that drives problematic use.
- Kappa antagonism. Buprenorphine blocks the kappa-opioid receptor, which is involved in dysphoria and stress-related negative affect. Some patients report improved mood.
- Safer in renal disease. Buprenorphine is metabolized hepatically and its metabolites are largely inactive; no dose adjustment is required in renal impairment, unlike morphine (accumulates toxic M6G), oxycodone, or hydromorphone.
- Less hyperalgesia. Clinical observation and some mechanistic studies suggest buprenorphine produces less OIH than full agonists, possibly because of its partial agonism and anti-inflammatory glial effects.
- Less endocrine suppression. Long-term full agonists suppress the hypothalamic-pituitary-gonadal axis, causing low testosterone, low libido, fatigue, and infertility. Buprenorphine appears to have a milder effect.
The catch: rotation onto buprenorphine from a full agonist can precipitate withdrawal because buprenorphine kicks other opioids off the receptor but only partially activates it. Modern protocols — micro-induction (low-dose cross-titration over 7–14 days without stopping the full agonist) — have made this transition far more comfortable than the old "wait until you are in withdrawal" method.
Methadone — Powerful and Complicated
Methadone is an effective analgesic, inexpensive, long-acting, and in some ways ideal for difficult pain — particularly neuropathic pain, because it has NMDA-receptor antagonism in addition to mu-agonism. It is also among the most dangerous opioids to prescribe, and any clinician starting a patient on methadone for chronic pain should have specific experience with it.
The core problems:
- Unpredictable half-life. Methadone's half-life ranges from 8 to 150 hours between individuals. Analgesia lasts only 6–8 hours, but the drug accumulates in tissue for days. Early dose titration is the window when most overdose deaths occur.
- QTc prolongation. Methadone blocks the hERG potassium channel, prolonging the QT interval on the ECG and creating a risk of torsades de pointes. Baseline ECG before starting, repeat at 30 days, then annually. Avoid combinations with other QT-prolonging drugs (ondansetron, certain antibiotics, many antipsychotics).
- Drug interactions. Methadone is metabolized via CYP3A4, 2B6, and 2D6. Inducers (rifampin, many anticonvulsants, St. John's wort) can precipitate withdrawal; inhibitors (fluconazole, many antiretrovirals) can cause toxicity.
- Non-linear MME conversion. The conversion ratio climbs with dose (4× at low doses, up to 12× at high doses), so a straightforward dose conversion from another opioid can kill a patient.
When done right by an experienced prescriber, methadone can be transformative for neuropathic pain refractory to other options. When done wrong, it is a leading cause of opioid overdose death per prescription written.
Long-Acting vs Short-Acting Opioids
The 2016 CDC guideline suggested short-acting opioids for initiation of chronic therapy. The 2022 revision softened this language — the choice now depends on the clinical situation.
Short-acting (immediate-release oxycodone, hydrocodone, morphine) peak in 30–60 minutes and wear off in 3–4 hours. Pros: easier dose titration, good for episodic or breakthrough pain, lower peak-trough total opioid load than a poorly chosen long-acting. Cons: more frequent dosing, roller-coaster pharmacokinetics that can reinforce clock-watching behavior, and the "rescue" mindset.
Long-acting (oxycodone ER, morphine ER, fentanyl patch, methadone, Belbuca, Butrans) provide steadier serum levels and once- or twice-daily dosing. Pros: smoother analgesia, less peak-trough swing, less behavioral reinforcement around the clock. Cons: higher baseline opioid exposure, slower dose adjustment, and in the case of extended-release tablets a small abuse-liability from tampering (which is why many have abuse-deterrent formulations).
A common modern approach for stable chronic pain is a long-acting backbone with minimal or no short-acting rescue, rather than stacking both continuously.
Risk Mitigation: PDMP, UDS, Naloxone, REMS
These are the four practical tools that allow ongoing opioid therapy to be defensible and, more importantly, safer.
PDMP (Prescription Drug Monitoring Program). Every U.S. state operates an electronic database of controlled-substance prescriptions. Clinicians are expected (and in most states required) to check it before each opioid prescription. This is not surveillance of the patient for its own sake — it flags dangerous combinations (benzodiazepines, gabapentinoids, sleeping pills) and detects unintentional duplication from multiple prescribers after emergency visits.
UDS (Urine Drug Screening). A periodic urine test confirms that the patient is taking the prescribed opioid and is not taking unprescribed or illicit substances. Done right, it is a clinical-care tool, not a punishment mechanism. Done wrong (patients discharged for a single unexpected result without a conversation), it is one of the leading sources of therapeutic-relationship rupture in pain medicine. Current best practice: risk-stratified testing (lower frequency for stable, low-risk patients), interpretation that accounts for metabolites and cross-reactivity, and a conversation before action.
Naloxone co-prescribing. Any patient on chronic opioids — and especially anyone on ≥50 MME/day, on concurrent benzodiazepines, with sleep apnea, with a history of overdose, or with substance-use history — should be co-prescribed naloxone (Narcan nasal spray, now available over-the-counter in the U.S.) and family members trained to use it. This is standard of care in 2026. It has no downside and saves lives.
REMS (Risk Evaluation and Mitigation Strategy). The FDA's REMS program for extended-release and long-acting opioids and for transmucosal fentanyl products requires prescriber education and certain patient counseling. For fentanyl buccal tablets and similar breakthrough-cancer-pain products, enrollment is mandatory.
Tapering — The VA Approach and the No-Forced-Taper Rule
The single most important shift of the last five years is the recognition that forced tapers cause harm. Peer-reviewed data from the Veterans Health Administration, large insurance databases, and multiple states have documented that patients cut off against their will experience increased rates of overdose (often on illicit fentanyl), suicide, emergency-department visits, and dramatic worsening of pain and function.
The Veterans Health Administration, which tapered tens of thousands of patients during the 2013–2019 period, revised its own approach sharply. Current VA / Department of Defense guidance (and the CDC 2022 revision) emphasizes:
- No forced tapers for stable patients on long-term opioid therapy who are benefiting and not showing harm.
- Patient-centered rate. When a taper is indicated, reductions of 5–10% every 4 weeks are standard; slower (2–5% every month or longer) for patients on long-term high-dose therapy.
- Pause and resume. If withdrawal symptoms, loss of function, or mood decline appear, pause the taper or return to the last tolerated dose. A taper that cannot be tolerated is not a taper that should be completed.
- Adjunct support. Clonidine or lofexidine for autonomic symptoms, hydroxyzine for sleep, non-opioid pain strategies scaled up as opioid is scaled down, behavioral health access.
- Consider buprenorphine rotation rather than a traditional taper. Many patients who cannot taper off full agonists successfully transition to buprenorphine and find improved pain, sleep, mood, and safety at a stable buprenorphine dose.
Indications where a taper is clinically justified: loss of benefit, intolerable side effects, evidence of OUD developing, serious safety events (overdose, sedation), and patient preference. "The CDC guideline says 90" is not an indication.
When Opioids ARE Appropriate
Despite the past decade's hard lessons, there is a legitimate place for opioids in chronic non-cancer pain. The clearest indications:
- True nociceptive pain refractory to multimodal therapy. Severe osteoarthritis in a patient who cannot have surgery, advanced spinal stenosis with imaging concordance, sickle cell disease, pancreatitis with documented structural disease, severe rheumatoid or psoriatic arthritis not responding to biologics, failed-back syndrome after real structural surgery.
- Specific neuropathic pain syndromes where first-line agents (SNRIs, gabapentinoids, topicals, TCAs) have failed. See non-opioid medications for the stepwise approach.
- Palliative-type situations not covered by formal hospice: severe heart failure, end-stage COPD, ALS, advanced renal or liver disease.
- Cancer, sickle cell, palliative, and end-of-life care — explicitly excluded from opioid-restrictive guidelines in the first place.
Opioids are a poor fit for nociplastic pain (fibromyalgia, many central-sensitization syndromes) where the problem is amplified signaling rather than tissue damage. In these conditions, opioids often provide little long-term benefit and risk making the system more sensitive over time. See central sensitization and fibromyalgia.
For peripheral neuropathy, first-line therapies remain the gabapentinoids, SNRIs (duloxetine), TCAs, and topicals; opioids are reserved for cases where those have genuinely failed. See peripheral neuropathy.
Legacy Patient Management
A "legacy patient" in opioid-medicine slang is someone who was initiated on chronic opioids (often at high doses) years or decades ago, under a different standard of care, and has been stable and functional ever since. The 2016 panic produced widespread forced tapering of these patients, with catastrophic results documented in multiple studies.
The current consensus — endorsed by the CDC 2022 revision, the AMA, the American Academy of Pain Medicine, and the FDA — is that legacy patients who are stable, functional, and not showing signs of harm should generally be maintained on their current regimen. Dose reductions should be considered only when clearly justified by a change in the patient's clinical picture, not by external pressure.
If you are a legacy patient whose prescriber is leaving practice, retiring, or refusing to continue prescribing:
- Ask for a written referral to a clinician who will continue your regimen. This is ethical standard of care.
- Request that any taper be patient-centered, slow, and pausable.
- Consider rotation to buprenorphine as a safer long-term maintenance option.
- Know that pharmacy refusal to fill is a separate problem from prescriber decisions; some patients must call several pharmacies to find one that will stock and dispense a legitimate prescription.
- The Human Rights Watch 2018 report "Not Allowed to Be Compassionate" documents the systemic failure of the forced-taper era and has been cited in subsequent guideline revisions.
Kratom: What We Know
Kratom (Mitragyna speciosa) is a tree native to Southeast Asia whose leaves contain mitragynine and 7-hydroxymitragynine, compounds with partial mu-opioid-receptor activity and some adrenergic effects. It is sold in the U.S. as a dietary supplement, without FDA approval, in powder, capsule, and extract forms.
Where the evidence stands in 2026:
- Pharmacology is real. Mitragynine is a partial mu-agonist with kappa and delta effects. At low doses it is stimulating; at higher doses it produces opioid-like analgesia and sedation.
- Clinical evidence is thin. No high-quality randomized trials for chronic pain. Survey data and case series suggest some users find benefit, particularly as a self-directed alternative when opioids have been withdrawn.
- Dependence and withdrawal occur with regular use and mimic mild-to-moderate opioid withdrawal.
- Adulteration is a real risk. "Kratom" products have been found adulterated with 7-hydroxymitragynine concentrates far exceeding natural levels, and in some cases with synthetic opioids. Quality control is unregulated.
- Overdose deaths have occurred, almost always in polysubstance contexts (benzodiazepines, alcohol, fentanyl).
- Regulatory status is unsettled. The DEA has twice proposed scheduling kratom and twice withdrawn the proposal. Some U.S. states (Indiana, Vermont, Arkansas, Wisconsin, Rhode Island, Alabama) have banned or restricted it; others regulate it as a consumer product. Federal status remains unscheduled as of this writing.
Kratom is not a substitute for medical care, not a tested chronic-pain therapy, and should be disclosed to any prescriber — both because it will show up (or be missed) on certain drug screens and because it interacts with other opioids and sedatives.
Practical Advice for Patients
- Keep an honest pain-and-function log. A week's worth of daily entries (pain 0–10, what you were able to do, medications taken) is the single most useful thing you can bring to an appointment.
- Bring a list of what has been tried. First-line non-opioids, physical therapy, injections, interventional procedures, behavioral health — documenting this protects you from being told "you haven't tried enough" when you have.
- Ask for naloxone, even if you think you don't need it. It is over-the-counter in the U.S. now and is a seatbelt, not an admission of risk.
- Avoid concurrent benzodiazepines where possible. The opioid-benzodiazepine combination is one of the strongest predictors of overdose and is the single biggest avoidable pharmacology risk in chronic pain.
- Treat sleep apnea if you have it. Untreated OSA plus opioids dramatically raises respiratory-depression risk.
- If your prescriber is tapering you against your will, ask for a written rationale, ask whether the taper can be slowed or paused, ask about buprenorphine rotation, and consider a second opinion from a board-certified pain physician.
- Read the CDC 2022 guideline yourself. It is shorter than people think and contradicts a great deal of what patients are told.
Key Research Papers
- Dowell D, Ragan KR, Jones CM, Baldwin GT, Chou R. CDC Clinical Practice Guideline for Prescribing Opioids for Pain — United States, 2022. MMWR Recomm Rep. 2022;71(3):1–95.
- Agnoli A, Xing G, Tancredi DJ, et al. Association of Dose Tapering With Overdose or Mental Health Crisis Among Patients Prescribed Long-term Opioids. JAMA. 2021;326(5):411–419.
- Krebs EE, Gravely A, Nugent S, et al. Effect of Opioid vs Nonopioid Medications on Pain-Related Function in Patients With Chronic Back Pain or Hip or Knee Osteoarthritis Pain: The SPACE Randomized Clinical Trial. JAMA. 2018;319(9):872–882.
- Darnall BD, Juurlink D, Kerns RD, et al. International Stakeholder Community of Pain Experts and Leaders Call for an Urgent Action on Forced Opioid Tapering. Pain Med. 2019;20(3):429–433.
- Pergolizzi JV, Raffa RB. Safety and efficacy of the unique opioid buprenorphine for the treatment of chronic pain. J Pain Res. 2019;12:3299–3317.
- Lee M, Silverman SM, Hansen H, Patel VB, Manchikanti L. A comprehensive review of opioid-induced hyperalgesia. Pain Physician. 2011;14(2):145–161.
- Oliva EM, Bowe T, Manhapra A, et al. Associations between stopping prescriptions for opioids, length of opioid treatment, and overdose or suicide deaths in US veterans. BMJ. 2020;368:m283.
- Chou R, Hartung D, Turner J, et al. Opioid Treatments for Chronic Pain. Comparative Effectiveness Review No. 229. AHRQ, 2020.
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on opioid prescribing, buprenorphine for pain, and safe tapering:
- CDC 2022 opioid prescribing guideline
- Buprenorphine for chronic non-cancer pain
- Opioid-induced hyperalgesia
- Forced opioid tapering and patient harm
- Methadone, QTc prolongation, and chronic pain
- Naloxone co-prescribing in chronic pain
- Opioid use disorder vs physical dependence
- Kratom and mitragynine pharmacology
Connections
- Peripheral Neuropathy
- Fibromyalgia
- Central Sensitization and Nociplastic Pain
- Non-Opioid Medications — Gabapentinoids, SNRIs, LDN
- Chronic Pain Overview
- Interventional Pain — Injections, Ablations, Neuromodulation
- Pain Reprocessing Therapy and PNE
- Chronic Low Back Pain — Evidence-Based Care
- Nociceptive vs Neuropathic vs Nociplastic Pain
- Sleep, Exercise, and Lifestyle for Chronic Pain
- Complex Regional Pain Syndrome
- Depression
- Chronic Fatigue Syndrome
- Arthritis
- Central Sensitization Explained
- Low-Dose Naltrexone for Fibromyalgia
- Sickle Cell Disease
- Migraine