Mitochondrial Dysfunction & Energy Metabolism in ME/CFS
If you have ME/CFS, you already know something is wrong with how your body makes and uses energy. A shower can wipe you out for a day. A short walk can cost you a week. A good morning can collapse into a bedbound afternoon for reasons that make no sense to anyone who has never lived it. This page explains what researchers have actually measured inside the cells, muscles, and bloodstream of ME/CFS patients — why your energy system is not just "tired" but genuinely broken — and what, if anything, supplements and pacing can do about it.
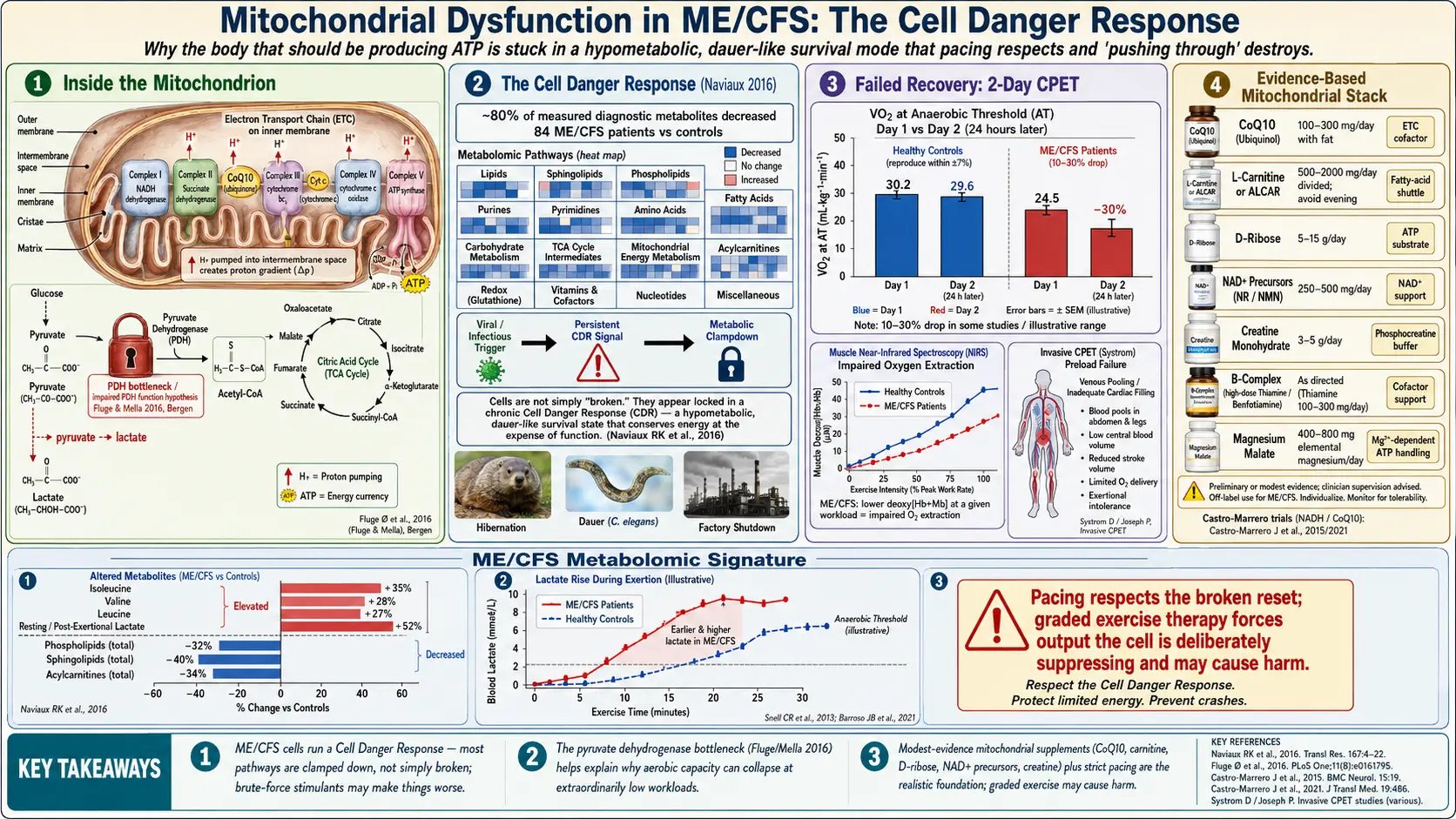
The short version: your mitochondria (the tiny energy factories inside almost every cell) are not producing, recovering, or switching fuel sources the way healthy mitochondria do. That is not a metaphor. It has been measured in breath tests, blood tests, muscle biopsies, and invasive cardiopulmonary exercise tests. Understanding the biology is the foundation for why pacing works and "push through it" does not.
Table of Contents
- The Core Finding: Cells Fail to Recover After Exertion
- The 2-Day CPET — The Single Most Damning Test
- Naviaux and the Cell Danger Response
- ATP Production and the Pyruvate Dehydrogenase Bottleneck
- Amino Acids, Lipids, and Lactate
- Muscle Oxygen Extraction and Preload Failure
- Mitochondrial Supplements — What Actually Has Evidence
- The Pacing Imperative
- Differentiating from Primary Mitochondrial Disease
- Why "Push Through It" Makes It Worse
- Key Research Papers
- PubMed Topic Searches
- Connections
- Featured Videos
The Core Finding: Cells Fail to Recover After Exertion
In a healthy person, mitochondria produce ATP (the molecule cells spend as energy) by burning glucose, fat, or amino acids with oxygen. You exert yourself, the system works hard, and within hours to a day the mitochondria are reset and ready to perform again. Fitness improves with use.
In ME/CFS this reset is broken. After exertion — physical, cognitive, emotional, or orthostatic — aerobic capacity drops rather than recovering. The cells appear to switch into a defensive, low-output state that persists for days. That low-output state is the biological footprint of post-exertional malaise (PEM), the hallmark symptom of the illness.
This is not deconditioning. Deconditioned people get better with graded exercise. ME/CFS patients get worse. It is also not depression — depressed people who exercise usually feel better afterward, not catastrophically worse 24–72 hours later. The timing, reproducibility, and measurability of the response make this a metabolic disorder, not a motivational one.
The 2-Day CPET — The Single Most Damning Test
The Workwell Foundation pioneered a two-day cardiopulmonary exercise test (CPET) that has become the most objective demonstration of ME/CFS energy dysfunction anywhere in the literature. Here is how it works:
- Day 1: The patient does a maximal exercise test on a stationary bike while breath gases, heart rate, and workload are measured. Key readings are VO2 max (peak oxygen uptake) and VO2 at the anaerobic/ventilatory threshold (VO2 AT) — the workload at which the body shifts from burning fat and oxygen to burning sugar without enough oxygen.
- Day 2: Exactly 24 hours later, the patient repeats the identical test.
In healthy people (even sedentary and deconditioned ones), Day 2 numbers are nearly identical to Day 1, or slightly better from a warm-up effect. In ME/CFS patients, VO2 at the anaerobic threshold drops, often by 10–30%. Workload at the same heart rate falls. The threshold where the cells are forced to burn sugar anaerobically arrives much sooner.
No other disease reliably reproduces this pattern. Heart failure, COPD, and sedentary controls all perform the same or better on Day 2. The 2-Day CPET is the closest thing ME/CFS has to a biomarker. See the 2-Day CPET and Objective Testing article for the clinical and disability-claim details — including why you should not do this test casually, because it will cause a severe PEM crash.
Naviaux and the Cell Danger Response
In 2016, Robert Naviaux and colleagues at UC San Diego published a metabolomic study of 84 ME/CFS patients and controls. They found a striking and counterintuitive pattern: ME/CFS patients showed a generalized hypometabolic signature. Roughly 80% of the measured metabolic pathways were down-regulated compared with healthy controls.
Naviaux described this as a dauer-like state — a biological parallel to the hibernation-like survival mode that roundworms (C. elegans) enter when the environment turns hostile. In that mode, the organism slows nearly every metabolic pathway to survive a stressor. Naviaux's hypothesis is that ME/CFS reflects a Cell Danger Response (CDR) that got triggered — often by a post-viral event, infection, or severe stressor — and never turned off.
Practically, this means the cells are not "failing" in a classical mitochondrial-disease sense of broken machinery. They are acting as if the machinery is fine but the command from above is "do not produce, we are under attack." The metabolic brakes are clamped on. That distinction matters for treatment: brute-force mitochondrial stimulants are unlikely to fix a downshifted cell, and may make things worse by forcing output the cell is deliberately suppressing.
ATP Production and the Pyruvate Dehydrogenase Bottleneck
Two independent research lines converge on a specific bottleneck inside the mitochondrion.
Myhill, Booth, and McLaren-Howard (2009, 2012, 2017) developed an ATP profile test that measures how efficiently white blood cells produce and recycle ATP. Their published results in ME/CFS cohorts consistently showed reduced ATP output and impaired recycling, with the degree of impairment correlating roughly with symptom severity. The test is controversial — it is not widely replicated outside their group — but the direction of the finding matches the rest of the literature.
Øystein Fluge and Olav Mella (2016) in Bergen, Norway published a seminal paper showing that ME/CFS patients have elevated blood levels of several amino acids that feed into energy metabolism, while simultaneously appearing to have impaired function of the pyruvate dehydrogenase (PDH) complex. PDH is the gatekeeper enzyme that converts pyruvate (from glucose) into acetyl-CoA, the fuel that enters the citric acid cycle and powers oxidative ATP production.
When PDH is throttled, the cell cannot burn sugar aerobically. It must either burn alternative fuels (amino acids, ketones, fats) or dump pyruvate into lactate and shunt energy anaerobically — which is wasteful and produces an acid load. Fluge and Mella proposed that an autoimmune process, or metabolic signaling, was actively inhibiting PDH kinases and related enzymes. Their group has since explored immunomodulatory treatments targeting this axis.
The practical takeaway: the bottleneck is at the entry to the aerobic engine, not the engine itself. That is why ME/CFS patients often hit their anaerobic threshold at extraordinarily low workloads — sometimes below the energy cost of simply standing up.
Amino Acids, Lipids, and Lactate
Metabolomic studies (Naviaux, Fluge/Mella, Germain, Armstrong, and others) have consistently found:
- Altered amino acid profiles — particularly those that can bypass PDH to feed the citric acid cycle directly (isoleucine, valine, leucine, glutamate). Patients appear to be compensating by burning amino acids for energy.
- Reduced phospholipids and sphingolipids — structural fats that make up cell and mitochondrial membranes. This echoes the "dauer" picture of a cell in shutdown.
- Elevated resting or post-exertional lactate — not dramatic enough to trigger a mitochondrial-disease diagnosis, but consistently above control levels, and rising abnormally fast during even mild exertion.
- Reduced acylcarnitines — markers that track fatty-acid transport into the mitochondrion.
None of these findings alone is specific enough to diagnose ME/CFS. But the pattern — a cell running on the wrong fuels, dumping lactate early, and rationing membrane lipids — is reproducible across labs.
Muscle Oxygen Extraction and Preload Failure
Energy is not just about cellular machinery. It is also about delivery. Two vascular findings complicate the pure mitochondrial story:
Impaired oxygen extraction at the muscle level. Studies using near-infrared spectroscopy and arterial/venous blood sampling have shown that ME/CFS patients extract less oxygen from their blood during exercise than controls. The cells are not pulling oxygen out efficiently — possibly because the mitochondria that would consume it are downregulated, and possibly because microvascular blood flow is disordered.
Preload failure on invasive CPET. David Systrom's group at Brigham and Women's Hospital has run invasive cardiopulmonary exercise tests (with pulmonary-artery catheters) on hundreds of ME/CFS and orthostatic-intolerance patients. The dominant finding is preload failure — the heart is not being adequately filled with blood during exercise, often because blood is pooling in the abdomen and legs due to autonomic dysfunction. The heart pumps what it receives, so cardiac output caps out at low workloads even with a structurally normal heart.
This overlaps heavily with POTS and small-fiber neuropathy. Many patients have both a cellular (mitochondrial) and a vascular (preload) limit to their energy envelope. Treating one without the other rarely works.
Mitochondrial Supplements — What Actually Has Evidence
Online ME/CFS communities circulate long supplement stacks. Most of the evidence is modest — small trials, open-label studies, anecdote — but the biological rationale is plausible and the risk is generally low. A realistic honest summary:
- CoQ10 / ubiquinol, 100–300 mg/day. Electron-transport-chain cofactor. Small randomized trials (Castro-Marrero and colleagues) showed modest improvements in fatigue and autonomic scores, particularly combined with NADH. Ubiquinol (the reduced form) is better absorbed than standard ubiquinone. Take with fat.
- L-carnitine or acetyl-L-carnitine (ALCAR), 500–2000 mg/day. Carries fatty acids into the mitochondrion. Small studies have shown benefit; acetyl-L-carnitine crosses into the brain and may help cognitive symptoms. Divided doses (morning and midday); avoid evening doses which can disturb sleep.
- D-ribose, 5–15 g/day (divided). A sugar that feeds directly into ATP synthesis. Teitelbaum's open-label data suggested benefit in a large ME/CFS and fibromyalgia cohort. Modest evidence, safe, sweet-tasting; some patients report it disrupts blood sugar.
- NAD+ precursors — nicotinamide riboside (NR) or nicotinamide mononucleotide (NMN), 250–500 mg/day. NAD+ is essential for the citric acid cycle. The evidence in ME/CFS specifically is limited but mechanistically reasonable. Expensive.
- Creatine monohydrate, 3–5 g/day. Buffers ATP in muscle and brain. Cheap, extensively studied in other contexts, now being trialed in long-term fatigue syndromes for cognitive benefit. Safe long-term in typical doses.
- B-complex (especially thiamine/B1, riboflavin/B2, niacinamide/B3). These are cofactors throughout energy metabolism. High-dose thiamine (e.g., benfotiamine 300 mg) has a small but intriguing evidence base. Inexpensive.
- Magnesium malate, 400–800 mg elemental magnesium/day (divided). Magnesium is a cofactor for ATP; malate feeds the citric acid cycle. Particularly helpful when fibromyalgia pain overlaps. Cut dose if it causes loose stools.
Honest expectations: most patients who respond report a roughly 10–25% improvement in baseline function on a well-chosen stack. Nobody is cured by supplements. Add them one at a time over 2–4 weeks each, so you can tell what is helping, what is neutral, and what makes you worse. Drop anything that produces no benefit after 8 weeks.
The Pacing Imperative
Given the biology above, pacing is not optional lifestyle advice — it is the single intervention with the strongest evidence base and the clearest mechanism.
The logic is simple. Your anaerobic threshold is abnormally low. Once you cross it, the cell switches to inefficient anaerobic metabolism, pumps out lactate, triggers the cell danger response, and drives a PEM crash that can last days to weeks. Each crash may cause an accumulating downward step in baseline function. Avoiding crashes is therefore a neuroprotective and mitochondrial-protective strategy, not an indulgence.
Practical pacing means staying below your anaerobic threshold as much as possible. Tools include heart-rate monitoring (many clinicians use a simple ceiling around 60% of [220 − age] as a starting point), activity logs, rest before and after any demand, and aggressive use of recumbent postures. The goal is to stay inside your energy envelope and slowly expand it over months — not to push it.
Differentiating from Primary Mitochondrial Disease
A common question: is ME/CFS actually an undiagnosed primary mitochondrial disease (a classical genetic disorder like MELAS or mitochondrial myopathy)? Almost always, no.
Primary mitochondrial diseases usually show:
- Onset in childhood or adolescence (though adult-onset forms exist).
- Multi-organ involvement with hard structural findings — deafness, stroke-like episodes, optic nerve damage, cardiomyopathy, diabetes, ptosis, external ophthalmoplegia.
- Persistently and markedly elevated lactate (often 3× upper limit of normal) with high lactate/pyruvate ratio.
- Diagnostic findings on muscle biopsy (ragged red fibers, COX-negative fibers, reduced respiratory-chain enzyme activity) or on mtDNA sequencing (specific heteroplasmic mutations).
- A family history that follows maternal inheritance.
ME/CFS does not typically show these features. A muscle biopsy and mitochondrial genetic testing are usually not warranted in the standard workup. They are reasonable to pursue only if there is a clinical signal suggesting primary mitochondrial disease — persistent very high lactate, neurological deficits, cardiomyopathy, hearing loss, or a strong maternal family history.
The better framing: ME/CFS is a functional mitochondrial and metabolic disorder — the machinery is intact but the regulation is off — rather than a structural one.
Why "Push Through It" Makes It Worse
For decades, patients were told that ME/CFS was essentially deconditioning with a psychological overlay, and that graded exercise therapy (GET) would fix it. The 2011 PACE trial promoted this view. It has since been thoroughly critiqued on methodological grounds, and in 2021 the UK's NICE guideline was rewritten to explicitly remove GET as a recommended therapy for ME/CFS. The U.S. CDC, the Institute of Medicine, and the Mayo Clinic have made similar moves.
The biology explains why. When you push past your anaerobic threshold:
- You force the cell into inefficient anaerobic metabolism that it cannot sustain.
- Lactate and metabolic byproducts rise.
- The cell danger response is reinforced rather than resolved.
- PEM begins, often 12–72 hours later, and persists for days.
- Repeated crashes appear to lower the baseline over time. Some patients progress from mild to severe or very severe ME after a series of pushed-through episodes.
That is not theoretical. It is the common arc described by patient registries and long-term observational cohorts. The most important single thing you can do for your mitochondria in ME/CFS is not a supplement. It is to respect the envelope, avoid crashes, and give the cell the environment it is signalling it needs.
Key Research Papers
- Naviaux RK, et al. Metabolic features of chronic fatigue syndrome. PNAS. 2016. — The hypometabolic/dauer signature paper.
- Fluge Ø, Mella O, et al. Metabolic profiling indicates impaired pyruvate dehydrogenase function in myalgic encephalopathy/chronic fatigue syndrome. JCI Insight. 2016. — The PDH-bottleneck paper.
- Myhill S, Booth NE, McLaren-Howard J. Chronic fatigue syndrome and mitochondrial dysfunction. Int J Clin Exp Med. 2009. — The ATP profile cohort.
- Davenport TE, Stevens SR, Stevens J, Snell CR, Van Ness JM. Properly measuring post-exertional malaise using CPET. Medicina. 2020. — 2-Day CPET methodology and findings.
- Oldham WM, Lewis GD, Opotowsky AR, Waxman AB, Systrom DM. Unexplained Exertional Dyspnea Caused by Low Ventricular Filling Pressures: Results from Clinical Invasive Cardiopulmonary Exercise Testing. Pulm Circ. 2016;6(1):55–62. — Preload failure in unexplained exertional intolerance.
- Castro-Marrero J, et al. Does oral coenzyme Q10 plus NADH supplementation improve fatigue and biochemical parameters in chronic fatigue syndrome? Nutr Metab. 2013.
PubMed Topic Searches
For further reading, these PubMed searches return current peer-reviewed work on ME/CFS energy metabolism:
- ME/CFS and mitochondrial dysfunction
- ME/CFS and two-day CPET
- ME/CFS and pyruvate dehydrogenase
- ME/CFS metabolomics
- Cell danger response (Naviaux)
- ME/CFS lactate and anaerobic threshold
- Invasive CPET and preload failure
- CoQ10 in ME/CFS
- D-ribose and fatigue
- Graded exercise therapy harms in ME/CFS
Connections
- ME/CFS Overview
- Post-Viral Triggers: EBV and Others
- Post-Exertional Malaise
- 2-Day CPET and Objective Testing
- Pacing and the Energy Envelope
- Severe and Very Severe ME
- Orthostatic Intolerance in ME/CFS
- ME/CFS Diagnostic Criteria
- LDN, Abilify and Experimental Therapies
- POTS
- MCAS
- Fibromyalgia
- Fatigue
- Magnesium
- Creatine
- Lyme Disease