HIV/AIDS (Human Immunodeficiency Virus / Acquired Immunodeficiency Syndrome)
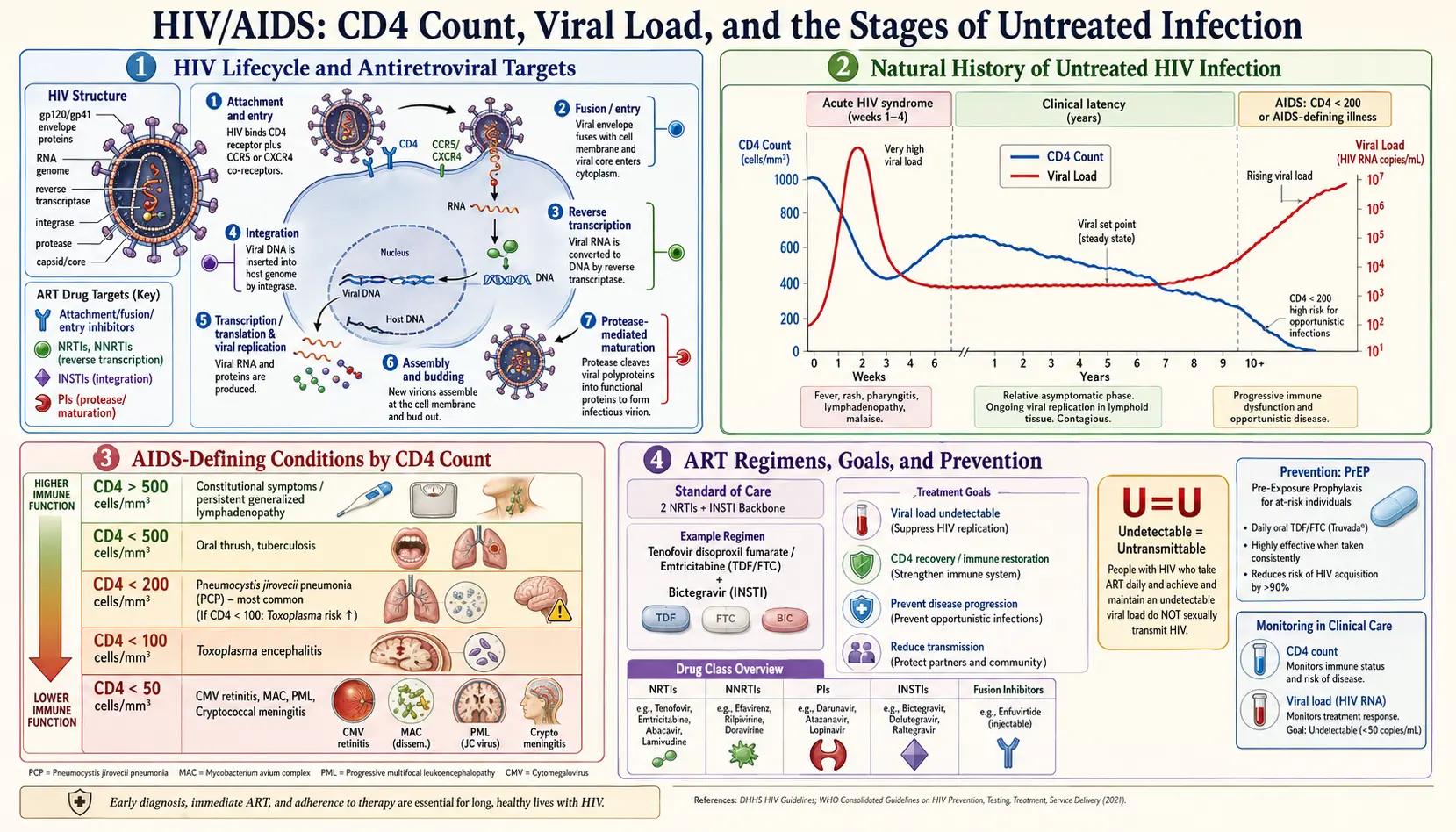
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research and Advances
- References
- Featured Videos
1. Overview
Human Immunodeficiency Virus (HIV) is a lentivirus belonging to the Retroviridae family that infects and progressively depletes CD4+ T-lymphocytes, macrophages, and dendritic cells — the critical mediators of cell-mediated immunity. Acquired Immunodeficiency Syndrome (AIDS) represents the final, advanced stage of HIV infection, defined by a CD4+ count below 200 cells/mm³ or the presence of one or more AIDS-defining illnesses in an HIV-infected individual.
HIV was first identified in 1983 by researchers at the Institut Pasteur (HIV-1) and subsequently characterized as a retrovirus encoding reverse transcriptase, integrase, and protease — enzymes that have become the primary targets of antiretroviral therapy (ART). Without treatment, median time from HIV acquisition to AIDS is approximately 10 years; with modern ART, life expectancy approaches that of the HIV-negative population.
Two distinct viral types are recognized: HIV-1 (globally predominant, more virulent) and HIV-2 (largely confined to West Africa, slower progression, intrinsically resistant to non-nucleoside reverse transcriptase inhibitors). HIV-1 is further classified into four groups (M, N, O, P) and multiple subtypes (clades A–K), with clade B predominating in North America and Western Europe.
2. Epidemiology
As of 2023, UNAIDS estimates approximately 39.9 million people are living with HIV globally, with sub-Saharan Africa bearing the greatest burden (roughly 25.9 million). Eastern and Southern Africa alone account for more than 54% of all people living with HIV. However, the fastest-growing epidemics are now in Eastern Europe, Central Asia, and parts of Asia-Pacific.
- Incidence: Approximately 1.3 million new infections occurred in 2023, a 59% reduction from the peak of 3.2 million in 1995.
- Mortality: AIDS-related deaths numbered approximately 630,000 in 2023, down from a peak of 2.1 million in 2004.
- Treatment coverage: 29.8 million of those living with HIV (approximately 75%) were accessing ART as of 2023.
- United States: Approximately 1.2 million people live with HIV; roughly 13% are unaware of their infection. Gay, bisexual, and other men who have sex with men (MSM) account for about 67% of new diagnoses.
Demographic disparities are profound: Black/African American individuals represent approximately 13% of the US population but account for 42% of new HIV diagnoses. Transgender women face HIV prevalence rates of 14–22% in high-burden settings. Globally, adolescent girls and young women (aged 15–24) in sub-Saharan Africa account for 20% of new HIV infections despite comprising only 10% of the population.
Transmission routes include sexual contact (vaginal, anal, oral — anal intercourse carries the highest per-act risk at approximately 1.4% receptive, 0.11% insertive), sharing of contaminated injection equipment (approximately 0.67% per-act risk), vertical transmission from mother to child (during pregnancy, labor/delivery, or breastfeeding), and — rarely — receipt of contaminated blood products or needlestick injuries in healthcare workers.
3. Pathophysiology
HIV's life cycle proceeds through several discrete steps, each of which represents a therapeutic target:
- Attachment and co-receptor binding: The viral envelope glycoprotein gp120 binds to CD4 on host T-cells, macrophages, and dendritic cells. Subsequent conformational change enables binding to chemokine co-receptors CCR5 (R5-tropic strains, dominant in early infection) or CXCR4 (X4-tropic, associated with disease progression), or both (dual-tropic).
- Fusion: gp41-mediated membrane fusion enables viral core entry into the cytoplasm.
- Reverse transcription: Viral RNA is reverse-transcribed into double-stranded DNA by HIV reverse transcriptase, an error-prone enzyme that introduces ~1 mutation per replication cycle, generating enormous viral diversity.
- Integration: Viral DNA (provirus) is transported into the nucleus and integrated into host chromosomal DNA by HIV integrase — establishing a permanent reservoir.
- Transcription and translation: Host cellular machinery transcribes viral genes; precursor polyproteins are synthesized.
- Assembly, budding, and maturation: Viral protease cleaves precursor polyproteins into functional structural and enzymatic components; mature, infectious virions bud from the cell membrane.
CD4+ T-cell depletion occurs via direct viral cytopathic effects, immune-mediated killing of infected cells, chronic immune activation, and pyroptosis — a form of inflammatory programmed cell death triggered by abortive HIV infection of resting CD4+ cells. The resulting immunodeficiency is compounded by chronic systemic inflammation driven by microbial translocation across a damaged gut epithelium (HIV enteropathy), generating a persistent pro-inflammatory milieu that accelerates cardiovascular, renal, and neurocognitive disease even in virologically suppressed patients on ART.
Viral reservoirs — long-lived resting CD4+ T-cells, macrophages, and potentially astrocytes — harbor integrated provirus and are impervious to ART, representing the principal barrier to cure. The gut-associated lymphoid tissue (GALT) is seeded within days of transmission and represents the largest anatomical compartment of HIV replication.
4. Etiology and Risk Factors
HIV infection is caused exclusively by the HIV-1 or HIV-2 retrovirus. Key risk factors for acquisition include:
- Sexual behavior: Unprotected receptive anal intercourse (highest per-act risk), multiple sexual partners, concurrent partnerships, sexually transmitted infections (particularly genital ulcer disease), and lack of male circumcision (for penile HIV acquisition).
- Injection drug use: Sharing needles, syringes, or other drug preparation equipment.
- Mother-to-child transmission (MTCT): Maternal viral load, duration of membrane rupture, breastfeeding, and maternal CD4 count are key determinants.
- Occupational exposure: Percutaneous needlestick injuries in healthcare workers (average risk 0.23% per incident).
- Biological susceptibility factors: Absence of CCR5-Δ32 homozygosity (a naturally protective mutation), high genital inflammation, microbiome dysbiosis, concurrent STIs.
- Socioeconomic and structural determinants: Poverty, gender inequality, criminalization of same-sex behavior, limited healthcare access, and stigma each independently amplify population-level risk.
5. Clinical Presentation
CDC Staging System (2014 Revised Classification)
HIV infection is classified into three stages based on CD4+ T-lymphocyte count and clinical criteria:
- Stage 1: CD4 count ≥500 cells/mm³, no AIDS-defining condition.
- Stage 2: CD4 count 200–499 cells/mm³, no AIDS-defining condition.
- Stage 3 (AIDS): CD4 count <200 cells/mm³ OR presence of an AIDS-defining illness regardless of CD4 count.
Clinical Phases
Acute HIV infection (Fiebig stages I–VI): Occurs 2–4 weeks post-exposure. Approximately 40–90% of newly infected individuals develop acute retroviral syndrome — a mononucleosis-like illness featuring fever, lymphadenopathy, pharyngitis, rash (maculopapular, trunk-predominant), myalgia, arthralgia, headache, and occasionally aseptic meningitis or encephalitis. Viral load peaks at 10⁶–10⁷ copies/mL; CD4 count transiently drops. Plasma HIV RNA (NAAT) is detectable before HIV antibodies, creating a seronegative window period of approximately 10–33 days.
Chronic HIV infection (clinical latency): Persistent generalized lymphadenopathy may occur. CD4 count declines at a median rate of 50–100 cells/mm³ per year in the absence of treatment. Patients are generally asymptomatic but infectious. HIV-associated conditions (e.g., oral candidiasis, hairy leukoplakia, herpes zoster, idiopathic thrombocytopenic purpura, seborrheic dermatitis) may emerge as CD4 count falls below 500 cells/mm³.
AIDS: CD4 <200 cells/mm³ or AIDS-defining illnesses including Pneumocystis jirovecii pneumonia (PCP), cerebral toxoplasmosis, cryptococcal meningitis, CMV retinitis, Mycobacterium avium complex (MAC), Kaposi sarcoma, primary CNS lymphoma, HIV wasting syndrome, and HIV encephalopathy/dementia.
6. Diagnosis
The CDC and USPSTF recommend opt-out HIV screening for all individuals aged 13–64 years in healthcare settings, and more frequent testing (at least annually) for persons at higher risk.
Recommended Diagnostic Algorithm (CDC 2014)
- Fourth-generation combination HIV-1/2 antigen/antibody immunoassay (Ag/Ab combo test): Detects both HIV p24 antigen and antibodies to HIV-1 and HIV-2. Sensitivity >99.9%; detects infection as early as 18 days post-exposure. This is the recommended initial test.
- HIV-1/HIV-2 antibody differentiation immunoassay: Performed on all reactive Ag/Ab combo specimens to distinguish HIV-1 from HIV-2 antibodies.
- HIV-1 nucleic acid test (NAT/NAAT): Performed if Ag/Ab combo is reactive but differentiation assay is negative or indeterminate — confirms acute HIV-1 infection and establishes plasma viral load baseline.
Monitoring Parameters
- CD4+ T-cell count: Prognostic marker; guides prophylaxis thresholds (e.g., PCP prophylaxis at CD4 <200, MAC prophylaxis at CD4 <50). Target: >500 cells/mm³ on ART.
- HIV plasma viral load (RNA PCR): Therapeutic response marker. Target: undetectable (<20–50 copies/mL) within 24 weeks of ART initiation. Measured at baseline, 2–4 weeks after ART initiation, every 4–8 weeks until undetectable, then every 3–6 months.
- Resistance testing: Genotypic resistance testing (HIV-1 reverse transcriptase, protease, integrase sequencing) at baseline before ART initiation, and with virologic failure.
- HIV-1 tropism assay (Trofile/genotypic prediction): Required before use of CCR5 antagonists (maraviroc).
- Additional baseline labs: Complete blood count, comprehensive metabolic panel, urinalysis, HBV serology (HBsAg, anti-HBs, anti-HBc), HCV antibody, syphilis serology, gonorrhea/chlamydia NAATs, tuberculin skin test or IGRA, chest radiograph, toxoplasma IgG, CMV IgG, varicella IgG, fasting lipid panel, glucose, HbA1c, urinary protein-to-creatinine ratio.
7. Treatment
Current guidelines (DHHS, EACS, WHO) recommend ART for all HIV-infected individuals, regardless of CD4 count, to prevent AIDS progression, reduce transmission risk (Treatment as Prevention, TasP), and decrease non-AIDS morbidity driven by chronic inflammation.
Antiretroviral Drug Classes
- Nucleoside/nucleotide reverse transcriptase inhibitors (NRTIs): Tenofovir alafenamide (TAF), tenofovir disoproxil fumarate (TDF), emtricitabine (FTC), abacavir (ABC), lamivudine (3TC), zidovudine (ZDV).
- Non-nucleoside reverse transcriptase inhibitors (NNRTIs): Rilpivirine, efavirenz, doravirine, etravirine, nevirapine.
- Integrase strand transfer inhibitors (INSTIs): Bictegravir, dolutegravir, raltegravir, elvitegravir/cobicistat, cabotegravir.
- Protease inhibitors (PIs): Darunavir/ritonavir, atazanavir/ritonavir, lopinavir/ritonavir.
- CCR5 antagonists: Maraviroc (for R5-tropic virus only).
- Fusion inhibitors: Enfuvirtide (injectable, reserved for salvage).
- Post-attachment inhibitors: Ibalizumab (for multidrug-resistant HIV).
- Capsid inhibitors: Lenacapavir (long-acting injectable, approved 2022 for heavily treatment-experienced patients).
Preferred Initial Regimens (DHHS 2024)
- Bictegravir/TAF/FTC (Biktarvy) — single-tablet, once daily; high barrier to resistance; preferred for most treatment-naive adults.
- Dolutegravir + TAF/FTC (or TDF/FTC) — highly effective; preferred in pregnancy (dolutegravir + TDF/FTC or ABC/3TC).
- Dolutegravir/rilpivirine (Juluca) — two-drug maintenance regimen; requires viral suppression, CD4 >200, no prior NNRTI resistance, no HBV coinfection.
- Cabotegravir + rilpivirine IM (Cabenuva) — long-acting injectable given monthly or every 2 months; approved for virologically suppressed adults.
Opportunistic Infection Prophylaxis
- PCP: TMP-SMX DS once daily (primary prophylaxis if CD4 <200 cells/mm³ or history of oropharyngeal candidiasis).
- Toxoplasmosis: TMP-SMX DS once daily (if CD4 <100 and toxoplasma IgG positive).
- MAC: Azithromycin 1200 mg weekly (if CD4 <50 and not on suppressive ART).
- Cryptococcus: Fluconazole prophylaxis in settings with high prevalence and CD4 <100.
- Tuberculosis (LTBI): Isoniazid 300 mg daily × 9 months (or rifapentine-based 3-month regimen) for all HIV+ with positive IGRA/TST.
8. Complications
AIDS-defining conditions (selected): Pneumocystis jirovecii pneumonia, cerebral toxoplasmosis, cryptococcal meningitis, CMV retinitis/colitis, MAC disseminated infection, Kaposi sarcoma (HHV-8), primary CNS lymphoma, progressive multifocal leukoencephalopathy (JC virus), cryptosporidiosis, HIV wasting syndrome, HIV-associated dementia.
Non-AIDS complications (driven by chronic immune activation even on suppressive ART): Cardiovascular disease (2–3× increased risk of MI; HIV-associated accelerated atherosclerosis), chronic kidney disease (HIV-associated nephropathy, FSGS, ART nephrotoxicity), liver disease (HCV/HBV coinfection, NAFLD, cirrhosis), neurocognitive impairment (HIV-associated neurocognitive disorders, HAND), osteopenia/osteoporosis, metabolic syndrome, non-AIDS malignancies (anal cancer, lung cancer, Hodgkin lymphoma, hepatocellular carcinoma — all increased), and immune reconstitution inflammatory syndrome (IRIS) following ART initiation.
Drug toxicities: TDF — renal tubular dysfunction, Fanconi syndrome, reduced bone mineral density; ABC — hypersensitivity reaction (HLA-B*57:01 screening mandatory before use); NNRTIs — hepatotoxicity, rash, CNS symptoms; PIs — dyslipidemia, lipodystrophy, insulin resistance, QTc prolongation; dolutegravir — insomnia, weight gain; cabotegravir — injection site reactions.
9. Prognosis
Prognosis has been transformed by effective ART. A 20-year-old HIV-positive individual initiating ART with a CD4 count >350 cells/mm³ now has a projected life expectancy of approximately 70–73 years — approaching that of the general population. Key prognostic determinants include:
- CD4 nadir: Lower nadir is associated with increased risk of non-AIDS complications even after viral suppression.
- Viral suppression: Undetectable viral load is the strongest predictor of favorable outcome.
- Adherence: >95% adherence is associated with durable viral suppression; modern single-tablet regimens have substantially simplified adherence.
- Comorbidities: Concurrent HCV, HBV, tuberculosis, or substance use disorder worsens prognosis.
- Timing of ART initiation: Earlier initiation (START trial, 2015) reduces serious illness and death even at CD4 >500.
Untreated HIV has a median survival from AIDS diagnosis of approximately 1–3 years. In resource-limited settings where ART access remains constrained, mortality remains substantial. The concept of Undetectable = Untransmittable (U=U) is now established: persons with plasma HIV RNA consistently <200 copies/mL have effectively zero risk of sexual transmission.
10. Prevention
Biomedical Prevention
- Pre-exposure prophylaxis (PrEP): Daily oral TDF/FTC (Truvada) or TAF/FTC (Descovy, for AMAB only) reduces HIV acquisition by >99% with consistent use. Long-acting injectable cabotegravir (CAB-LA) every 8 weeks has demonstrated superiority over oral TDF/FTC in the HPTN 083 and 084 trials (86–89% reduction vs. oral PrEP).
- Post-exposure prophylaxis (PEP): Must be initiated within 72 hours of potential exposure; 28-day course of preferred regimen (TDF/FTC + dolutegravir or raltegravir). Highly effective if started promptly.
- Treatment as Prevention (TasP): ART achieving viral suppression eliminates sexual transmission (PARTNER studies, HPTN 052).
- Voluntary medical male circumcision (VMMC): Reduces female-to-male HIV transmission by approximately 60%.
- Prevention of MTCT (PMTCT): Universal ART in pregnant women reduces MTCT to <2% (from ~30% without intervention).
- Harm reduction: Needle/syringe programs, medication-assisted treatment (methadone, buprenorphine) for opioid use disorder, naloxone distribution.
Behavioral and Structural Prevention
- Consistent and correct condom use, HIV testing and counseling, partner notification, and treatment of STIs.
- Decriminalization of HIV non-disclosure and same-sex behavior; addressing gender-based violence and structural poverty.
11. Recent Research and Advances
- Long-acting injectables: Lenacapavir (subcutaneous, every 6 months) combined with injectable broadly neutralizing antibodies is under evaluation in the PURPOSE 1 and 2 PrEP trials, with PURPOSE 1 demonstrating 100% efficacy in women (2024 interim analysis).
- Functional cure strategies: "Kick-and-kill" (latency reversing agents + immune-mediated clearance), gene therapy (CCR5 knockout via CRISPR), and shock-and-kill approaches are in clinical trials. The "Berlin patient" (allogeneic CCR5-Δ32 bone marrow transplant), "London patient," and several subsequent cases demonstrate proof-of-concept for HIV eradication.
- Broadly neutralizing antibodies (bNAbs): VRC01, 3BNC117, 10-1074, and combinations are in trials for treatment and prevention. bNAbs targeting the CD4 binding site, V3-glycan, and MPER epitopes show promise.
- Reservoir quantification: Intact proviral DNA assay (IPDA) has replaced total HIV DNA as the preferred measure of replication-competent reservoir size.
- Dolutegravir in pregnancy: WHO now recommends dolutegravir-based ART throughout pregnancy following reassuring data from the Tsepamo study clarifying the initial neural tube defect signal.
- Weight gain on INSTI-based ART: Ongoing investigation of mechanisms (off-target MC4R antagonism by dolutegravir/bictegravir) and mitigation strategies.
12. References
- UNAIDS. Global HIV & AIDS Statistics — Fact Sheet 2024. unaids.org
- Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the Use of Antiretroviral Agents in Adults and Adolescents with HIV. DHHS 2024. https://clinicalinfo.hiv.gov/en/guidelines/adult-and-adolescent-arv
- Cohen MS, Chen YQ, McCauley M, et al. Prevention of HIV-1 infection with early antiretroviral therapy (HPTN 052). N Engl J Med. 2011;365:493–505.
- The INSIGHT START Study Group. Initiation of antiretroviral therapy in early asymptomatic HIV infection. N Engl J Med. 2015;373:795–807.
- Rodger AJ, Cambiano V, Bruun T, et al. Risk of HIV transmission through condomless sex in serodifferent gay couples (PARTNER): final results of a multicentre, prospective, observational study. Lancet. 2019;393:2428–2438.
- Landovitz RJ, Donnell D, Clement ME, et al. Cabotegravir for HIV prevention in cisgender men and transgender women (HPTN 083). N Engl J Med. 2021;385:595–608.
- Bekker LG, et al. Twice-yearly lenacapavir for HIV prevention in women (PURPOSE 1). N Engl J Med. 2024 (interim results).
- Günthard HF, Calvez V, Paredes R, et al. European AIDS Clinical Society (EACS) Guidelines Version 12.0. HIV Med. 2023;24:S1–S201.
- Finzi D, Hermankova M, Pierson T, et al. Identification of a reservoir for HIV-1 in patients on highly active antiretroviral therapy. Science. 1997;278:1295–1300.
- Riddler SA, Haubrich R, DiRienzo AG, et al. Class-sparing regimens for initial treatment of HIV-1 infection (ACTG 5142). N Engl J Med. 2008;358:2095–2106.
- Cihlar T, Fordyce M. Current status and prospects of HIV treatment. Curr Opin Virol. 2016;18:50–56.
- Deeks SG, Lewin SR, Havlir DV. The end of AIDS: HIV infection as a chronic disease. Lancet. 2013;382:1525–1533.
- Saez-Cirion A, Pancino G. HIV controllers: a heterogeneous group of HIV-infected individuals with spontaneous control of viral replication. Cell Mol Immunol. 2013;10:15–26.
- Chu C, Selwyn PA. Complications of HIV infection: a systems-based approach. Am Fam Physician. 2011;83:395–406. https://www.aafp.org/pubs/afp/issues/2011/0215/p395.html
- CDC. Revised recommendations for HIV testing of adults, adolescents, and pregnant women in health-care settings. MMWR Recomm Rep. 2006;55(RR-14):1–17. https://www.cdc.gov/mmwr/preview/mmwrhtml/rr5514a1.htm
Research Papers
The following PubMed topic searches retrieve current peer-reviewed literature on HIV/AIDS. Each link opens a live PubMed query so you always see the most recent publications.
- HIV review
- Antiretroviral therapy HIV outcomes
- HIV pre-exposure prophylaxis PrEP
- HIV tenofovir emtricitabine
- HIV viral load undetectable untransmittable
- HIV mother to child transmission prevention
- HIV long acting injectable cabotegravir
- HIV cure Berlin patient stem cell
- HIV opportunistic infection prophylaxis
- HIV tuberculosis coinfection
- HIV integrase inhibitor dolutegravir
- HIV vaccine development trial
Connections
- Tuberculosis
- Hepatitis C
- Hepatitis B
- Immune Boosting
- Zinc
- NAC
- Vitamin D3
- Echinacea
- Meningitis
- Lymphoma
- Cancer
- Pneumonia
- Complete Blood Count
- Comprehensive Metabolic Panel
- Kidney Disease
- Cardiovascular Disease