Pregnancy and IBD: Fertility, Medications, and Outcomes
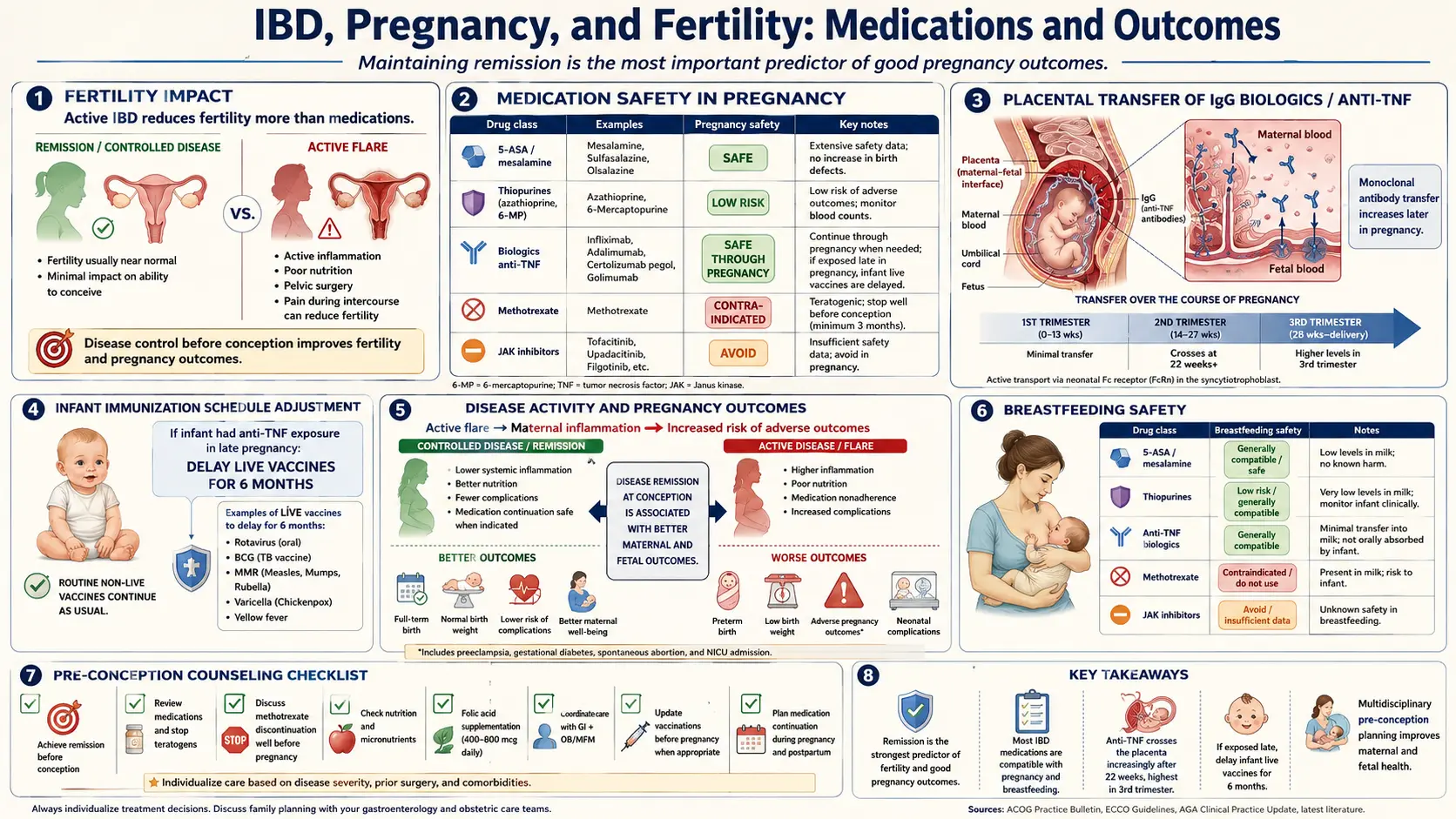
Table of Contents
- The Big Picture: Active Disease Is the Real Risk
- Preconception Counseling — Why 3 to 6 Months Matters
- Fertility and IBD
- Medications That Are Safe to Continue
- Medications to Stop Before Conception
- Newer Drugs: JAK Inhibitors and S1P Modulators
- Monitoring a Flare During Pregnancy
- Delivery Mode: Vaginal vs. C-Section
- Postpartum Flare Risk
- Breastfeeding on IBD Medications
- The Baby: Live Vaccines After In-Utero Biologic Exposure
- The PIANO Registry — What It Actually Found
- Practical Planning Checklist
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
The Big Picture: Active Disease Is the Real Risk
If you take one thing from this article, take this: the biggest threat to a healthy pregnancy with IBD is not your medication — it is uncontrolled inflammation. Women who conceive during an active flare have measurably worse outcomes: higher rates of preterm birth, low birth weight, pregnancy loss, and neonatal intensive care admissions. Women who conceive in deep remission on maintenance therapy have outcomes that closely mirror the general population.
This inverts the instinct almost every newly pregnant patient has. The reflex is: "I am pregnant, I should stop my drugs." For IBD, that reflex is wrong. Stopping biologics, immunomodulators, or mesalamine during pregnancy is one of the most reliable ways to trigger a flare — and a flare in pregnancy is genuinely dangerous. The question is not whether to take medication. It is which medications are compatible with pregnancy, which must be swapped out, and when to make those changes.
Major gastroenterology and obstetric societies — the American Gastroenterological Association (AGA), the European Crohn's and Colitis Organisation (ECCO), and the Toronto Consensus — all agree on this framing. The guidance below reflects that consensus, supplemented with the PIANO pregnancy registry data that has reshaped the field over the past decade.
Preconception Counseling — Why 3 to 6 Months Matters
The ideal time to plan an IBD pregnancy is before you are pregnant. The target most specialists use: three to six months of sustained, objective remission on a stable medication regimen before attempting conception.
"Objective" matters. Feeling well is not enough. Before conception, you want:
- Fecal calprotectin under 150–250 μg/g (ideally below 150).
- CRP in the normal range for your lab.
- Endoscopic or imaging evidence of mucosal healing if the last scope was more than a year ago — colonoscopy for UC or colonic Crohn's, MR enterography for small-bowel Crohn's.
- Hemoglobin above 11 g/dL and iron stores replete (ferritin above 30 ng/mL, ideally above 100). See the fatigue and anemia article.
- Vitamin D above 30 ng/mL, B12 normal, folate adequate.
Three to six months is not arbitrary. It is roughly the window over which a flare that will happen on a given regimen tends to declare itself. If you have been on ustekinumab for nine months with normal calprotectin, you are far less likely to flare during pregnancy than if you just switched therapies two weeks ago.
Start folic acid 400–800 μg daily at least one month before conception. If you are on or have recently taken sulfasalazine, use 2 mg daily, because sulfasalazine interferes with folate absorption. Women who have undergone small-bowel resection or have active ileal Crohn's may need higher folate and a sublingual or injectable B12.
Fertility and IBD
Baseline fertility in IBD is not generally reduced when disease is quiescent and the pelvis is surgically untouched. The important exceptions:
Ileal pouch-anal anastomosis (IPAA / J-pouch) in women
This is the single biggest IBD-related fertility issue. Women who have had a total colectomy with J-pouch construction for ulcerative colitis experience a roughly three-fold reduction in spontaneous fertility compared with medically managed UC. The mechanism is pelvic adhesions encasing the fallopian tubes and ovaries after open surgery. Studies put infertility rates around 26–38% post-IPAA vs. 10–15% in medically managed controls.
Two practical implications:
- Laparoscopic J-pouch surgery appears to reduce but not eliminate this fertility penalty. If surgery is being planned and childbearing is a priority, find a high-volume center that does laparoscopic IPAA.
- IVF works well in post-IPAA patients. Outcomes approach those of the general IVF population, because the problem is mechanical (blocked tubes), not a fundamental ovarian or uterine issue. If you are considering a J-pouch and not done having children, discuss fertility preservation (oocyte or embryo freezing) before the surgery.
Active disease and ovulation
Active Crohn's or UC can suppress ovulation through the usual mechanisms of chronic inflammation and weight loss. Cycles often normalize within months of achieving remission. Persistent menstrual irregularity after two to three months of deep remission warrants standard fertility workup rather than assuming the IBD is the cause.
Sulfasalazine and male fertility
This one is genuinely important and often missed. Sulfasalazine reversibly impairs sperm count, motility, and morphology in up to 60% of men who take it. The effect is due to the sulfapyridine moiety, not the 5-ASA. It fully reverses within about two to three months of switching to pure mesalamine (asacol, lialda, pentasa, apriso) or stopping the drug. A man on sulfasalazine trying to conceive should be switched to mesalamine well before attempts begin.
Mesalamine, azathioprine, 6-mercaptopurine, and the anti-TNF biologics do not appear to impair male fertility at clinically meaningful levels. Methotrexate does — and is teratogenic — so it must be stopped (see below).
Medications That Are Safe to Continue
The core message: most IBD drugs are compatible with pregnancy, and stopping them usually causes more harm than continuing.
Mesalamine (5-ASA)
Safe throughout pregnancy. Decades of data, no increase in malformations or adverse outcomes. Continue at the same dose. The only wrinkle is Asacol HD (the higher-dose branded version), whose coating historically contained dibutyl phthalate — reformulated in 2013, but if you have old stock, switch to a different 5-ASA product.
Azathioprine and 6-mercaptopurine
Despite an old FDA category D label, the pragmatic evidence from PIANO and European registries shows these drugs are safe to continue during pregnancy when needed for remission. Do not start them during pregnancy (let the pregnancy-specific risk pool stay as small as possible), but if you are already on a stable dose and in remission, continue.
Anti-TNF biologics
This is the cornerstone of modern pregnancy-compatible IBD care. Infliximab (Remicade), adalimumab (Humira), and golimumab (Simponi) are IgG1 antibodies that cross the placenta actively in the second and third trimesters, reaching levels in the infant often higher than the mother's at birth. PIANO and multiple national cohorts show no increase in congenital malformations, preterm birth, or infection when mothers continue these drugs.
Certolizumab pegol (Cimzia) is the exception — and a useful one. It is a Fab' fragment conjugated to polyethylene glycol with no Fc region, so it does not cross the placenta via the active neonatal Fc receptor transport system. Cord blood levels are negligible. For women starting biologic therapy during reproductive years, certolizumab is the anti-TNF with the lowest infant drug exposure, though infliximab and adalimumab have larger evidence bases for efficacy in Crohn's.
Historically, some clinicians stopped infliximab or adalimumab in the third trimester to reduce placental transfer. Current AGA and ECCO guidance is not to interrupt therapy in actively managed patients, because the risk of flare outweighs the theoretical infant-exposure benefit. See the biologics article for the drug-by-drug detail.
Ustekinumab (Stelara)
An IgG1 anti-IL-12/23 antibody. Crosses the placenta. Accumulating data from the PIANO registry and post-marketing surveillance show no increase in congenital anomalies or adverse pregnancy outcomes. Continue if needed for remission.
Vedolizumab (Entyvio)
A gut-selective anti-integrin (α4β7) IgG1 antibody. Placental transfer occurs, but because the drug acts locally in the gut rather than systemically, infant exposure concerns are minimal. PIANO data again reassuring. Continue as needed.
Risankizumab (Skyrizi) and mirikizumab (Omvoh)
Anti-IL-23 biologics with more limited pregnancy data than ustekinumab but no signal of harm in the available case series. Reasonable to continue if the patient is in deep remission and switching carries meaningful flare risk. Discuss with a maternal-fetal medicine specialist.
Corticosteroids
Prednisone and budesonide are permissible when needed to control a flare but are not first-line maintenance in pregnancy. Prolonged systemic steroid exposure is associated with a small increase in gestational diabetes, hypertension, and — if used in the first trimester — a historically reported (but modestly-sized) risk of oral clefts. If steroids are needed, use the lowest effective dose for the shortest necessary duration. Budesonide (locally active, low systemic exposure) is often preferred for ileocecal Crohn's. See the gestational diabetes article.
Medications to Stop Before Conception
Methotrexate — absolutely contraindicated
Methotrexate is a known teratogen and abortifacient. It causes "methotrexate embryopathy" — craniofacial malformations, skull ossification defects, limb abnormalities — at doses used for IBD. It must be stopped at least three months before attempting conception in both women and men. Men on methotrexate should also stop three months in advance, both to clear the drug and to complete a full sperm-production cycle. Replace with a pregnancy-compatible maintenance drug (often an anti-TNF or azathioprine) before the methotrexate washout so remission is not lost in the gap.
Thalidomide and tofacitinib — contraindicated
Thalidomide's teratogenicity is the textbook case. It should never be used in anyone who could become pregnant without a strict pregnancy-prevention program, and it must be stopped before conception.
Tofacitinib (Xeljanz), upadacitinib (Rinvoq), and other JAK inhibitors are category-equivalent contraindicated in pregnancy based on animal teratogenicity data. They should be stopped and replaced with a pregnancy-compatible alternative before conception. There is no safe in-pregnancy JAK strategy; switch off. More on these drugs in the next section.
Ciprofloxacin and metronidazole — avoid when possible
Both are sometimes used in perianal Crohn's or pouchitis. Neither is a clear teratogen, but first-trimester metronidazole and any-trimester ciprofloxacin have theoretical concerns (cartilage effects in animal studies for fluoroquinolones). Use only when clinically necessary and no safer alternative exists.
Newer Drugs: JAK Inhibitors and S1P Modulators
The past five years have added two oral drug classes to IBD care — and both are not pregnancy-compatible. Patients using these must plan ahead.
JAK inhibitors: tofacitinib and upadacitinib
Tofacitinib (UC) and upadacitinib (UC and Crohn's) are small-molecule Janus kinase inhibitors. Animal reproductive toxicology shows teratogenicity, embryo-fetal death, and reduced fetal weight at clinically relevant doses. Human data are limited but offer no reassurance. The manufacturer recommends stopping these drugs and using effective contraception during therapy and for at least four weeks after the last dose (tofacitinib) or longer (upadacitinib). Practically, plan to transition to a pregnancy-compatible drug at least three months before attempts.
S1P modulators: ozanimod and etrasimod
Ozanimod (Zeposia) and etrasimod (Velsipity) are sphingosine-1-phosphate receptor modulators approved for UC. Animal studies show embryo-fetal toxicity. Both drugs have long half-lives and active metabolites, so washout is lengthy: women stopping ozanimod should use effective contraception for at least three months after the last dose before attempting conception; etrasimod washout is shorter but still requires planning. See the JAK and S1P article for mechanism and switching strategies.
The switching choreography
If you are on a non-pregnancy-compatible drug and planning pregnancy, the usual sequence is:
- Confirm you are in objective remission on current therapy.
- Overlap-start the pregnancy-compatible replacement (typically an anti-TNF, ustekinumab, or vedolizumab) at least one dosing interval before stopping the JAK/S1P drug.
- Stop the JAK/S1P drug.
- Wait the washout period (minimum 4 weeks for tofacitinib, 3 months for ozanimod) before attempting conception.
- Reconfirm remission with calprotectin and CRP before trying.
Monitoring a Flare During Pregnancy
The monitoring toolkit shifts during pregnancy because of imaging and endoscopy constraints:
- Fecal calprotectin remains reliable throughout pregnancy and is the most useful non-invasive tool.
- CRP rises modestly in normal pregnancy, so interpret values in context. Significant elevations still indicate inflammation.
- MRI without gadolinium is the imaging modality of choice for suspected active small-bowel Crohn's. Avoid gadolinium contrast throughout pregnancy because of fetal retention concerns.
- CT scans should be avoided except in life-threatening emergencies (suspected perforation, obstruction with non-diagnostic MRI). Ionizing radiation to the fetus is the concern.
- Flexible sigmoidoscopy without sedation or with minimal sedation is considered safe in any trimester if needed to distinguish flare from infection.
- Full colonoscopy is best deferred to the second trimester if required.
- Stool cultures and C. difficile PCR should be sent with any apparent flare — infection mimics flare and is treated differently.
Iron deficiency is common and worsens with pregnancy. Check ferritin, transferrin saturation, and CBC each trimester. IV iron (ferric carboxymaltose or ferric derisomaltose) is safe in the second and third trimesters and is often more effective than oral iron in IBD.
Delivery Mode: Vaginal vs. C-Section
The default answer in IBD is vaginal delivery unless there is a specific obstetric or IBD indication for C-section. The IBD-specific indications that shift the decision toward C-section:
- Active perianal Crohn's disease — fistulas, abscesses, or active inflammation in the perineum. Vaginal delivery through actively inflamed or fistulized tissue worsens disease and impairs wound healing. A planned C-section is standard in this group.
- Ileal pouch (IPAA / J-pouch) — a relative indication. Pouch function is generally well-preserved with vaginal delivery in experienced hands, but some surgeons and patients prefer C-section to avoid any risk to sphincter integrity. Reasonable to discuss with the colorectal surgeon who built the pouch.
- Permanent ileostomy with strong patient preference — not a hard indication but often considered.
Ulcerative colitis without perianal involvement and luminal-only Crohn's are not indications for C-section on IBD grounds alone.
Postpartum Flare Risk
Roughly one-third of women with IBD experience a flare within six months postpartum, with the highest risk in the first three months. The drivers are sleep deprivation, relative immunologic shift out of the pregnancy state, medication interruption or dose delay around delivery, and sometimes self-initiated drug discontinuation for breastfeeding.
Protect this window by:
- Continuing biologics and immunomodulators through delivery and postpartum. Do not pause infliximab, adalimumab, ustekinumab, or vedolizumab for "safety" reasons around delivery — the flare risk is far greater than the drug risk.
- Scheduling the next biologic dose to fall within two weeks after delivery rather than letting it drift.
- Checking calprotectin at six weeks and three months postpartum if symptomatic.
- Replacing iron and B12 aggressively. Postpartum anemia on top of IBD anemia is brutal.
- Sleep triage. Not a joke — sleep loss is an independent flare trigger. Any support that protects four-hour sleep blocks is medical, not optional.
Breastfeeding on IBD Medications
Most IBD medications are compatible with breastfeeding. Milk transfer of the large monoclonal antibodies is minimal, and even the tiny amount that transfers is largely degraded in the infant gut.
- Mesalamine: compatible. Watch infant for rare diarrhea.
- Azathioprine / 6-MP: compatible; metabolite levels in milk are very low.
- Infliximab, adalimumab, certolizumab, golimumab, ustekinumab, vedolizumab, risankizumab: compatible. Antibodies are degraded by the infant's digestive tract.
- Corticosteroids: compatible. For high-dose prednisone (over 20 mg/day), waiting 4 hours after the dose before nursing further reduces infant exposure.
- Methotrexate: contraindicated.
- Tofacitinib, upadacitinib, ozanimod, etrasimod: not recommended; limited data and small-molecule drugs transfer to milk.
- Ciprofloxacin, metronidazole: short courses are generally tolerated; some sources prefer alternatives.
The Baby: Live Vaccines After In-Utero Biologic Exposure
Infants born to mothers on placenta-crossing biologics (infliximab, adalimumab, golimumab, ustekinumab, vedolizumab, risankizumab) carry measurable drug levels at birth. The drug gradually clears over six to twelve months. During that window, the infant is mildly immunosuppressed, and live vaccines should be delayed.
Practically:
- Rotavirus vaccine is the main near-term issue. It is a live oral vaccine given in the U.S. at 2, 4, and sometimes 6 months. Current guidance from the AGA and most pediatric IBD experts is to avoid rotavirus vaccination in infants exposed in utero to placenta-crossing biologics. Recent observational data suggest rotavirus vaccination may actually be safe in this population, and individual pediatricians may make a case-by-case decision, but the default remains skip.
- BCG vaccine (given in countries with high TB burden, not routinely in the U.S.) should be delayed until at least 6 months after birth, ideally 12 months, in infants with significant biologic exposure. Historical case reports of disseminated BCG after in-utero infliximab exposure reinforce this.
- MMR and varicella vaccines are given at 12 months or later — by that point most placentally transferred biologic has cleared. Proceed on the normal schedule.
- Inactivated vaccines (DTaP, Hib, pneumococcal, polio, hepatitis B, influenza) are fine on the normal schedule. In-utero biologic exposure does not impair their response in any clinically meaningful way.
Certolizumab is an exception. Because it does not cross the placenta, infants of certolizumab-exposed mothers can receive rotavirus and BCG on the normal schedule. Tell your pediatrician the specific drug — "biologic" is not enough information.
The PIANO Registry — What It Actually Found
The PIANO (Pregnancy in Inflammatory Bowel Disease and Neonatal Outcomes) registry is the largest prospective cohort of pregnancies in IBD, enrolling over 1,700 women across multiple U.S. centers. Its findings rewrote the pregnancy playbook:
- No increase in congenital malformations, infant infections, or developmental delay at one year in children exposed to anti-TNFs, thiopurines, or combination therapy compared to unexposed IBD pregnancies.
- Active disease — not drug exposure — was the clearest driver of preterm birth and low birth weight.
- Developmental milestones at four years were no different in biologic-exposed vs. unexposed children.
- The only caveat: a signal of slightly higher infant infection rates in the first year among those exposed to combination therapy (biologic + thiopurine) compared with monotherapy, though absolute rates remained low.
This is why modern guidelines lean heavily toward continuing biologics through pregnancy. The old intuition — stop the drug, protect the baby — turns out to be the opposite of what the data show.
Practical Planning Checklist
For a patient six to twelve months out from trying to conceive:
- Confirm remission objectively — calprotectin under 150 μg/g, CRP normal, updated imaging or scope if due.
- Switch off incompatible drugs — methotrexate (3 months before), tofacitinib/upadacitinib (at least 4 weeks before, with replacement started first), ozanimod/etrasimod (3+ months before).
- Switch from sulfasalazine to mesalamine if male partner is on it — 2 to 3 months before attempts.
- Start folic acid (400–800 μg, or 2 mg if on sulfasalazine or post-resection).
- Optimize iron, B12, vitamin D.
- Update vaccinations before pregnancy — especially MMR, varicella, and any live vaccines you may need, since you cannot receive them during pregnancy or while on biologics.
- Establish care with a maternal-fetal medicine specialist and an obstetrician experienced with IBD pregnancies.
- Plan delivery around perianal disease status; document the plan in the chart early.
- Anticipate the postpartum drug schedule — no biologic gaps.
- Brief your pediatrician on in-utero drug exposure and the live-vaccine schedule before the baby is born.
Many of these steps parallel the preparation for other autoimmune pregnancies — compare the pregnancy and Hashimoto's article for a similar playbook in autoimmune thyroid disease.
Key Research Papers
- Mahadevan U, et al. Inflammatory Bowel Disease in Pregnancy Clinical Care Pathway: A Report From the American Gastroenterological Association IBD Parenthood Project Working Group. Gastroenterology. 2019.
- Mahadevan U, et al. Pregnancy Outcomes in Women With Inflammatory Bowel Disease: A Large Multicenter Study (PIANO). Gastroenterology. 2021.
- Torres J, et al. European Crohn's and Colitis Organisation Topical Review on Reproduction and Pregnancy in Inflammatory Bowel Disease. J Crohns Colitis. 2023.
- Nguyen GC, et al. The Toronto Consensus Statements for the Management of Inflammatory Bowel Disease in Pregnancy. Gastroenterology. 2016.
- Mahadevan U, et al. Placental transfer of anti-tumor necrosis factor agents in pregnant patients with inflammatory bowel disease. Clin Gastroenterol Hepatol. 2013.
- Waljee A, et al. Threefold increased risk of infertility: a meta-analysis of infertility after ileal pouch anal anastomosis in ulcerative colitis. Gut. 2006.
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on pregnancy, fertility, and medication safety in IBD:
- IBD pregnancy outcomes
- PIANO registry data
- Anti-TNF placental transfer
- Certolizumab and the placenta
- Ustekinumab and vedolizumab in pregnancy
- JAK inhibitors in pregnancy
- S1P modulators in pregnancy
- Ileal pouch and female fertility
- Sulfasalazine and male fertility
- Rotavirus vaccine after in-utero biologic exposure
- Perianal Crohn's and mode of delivery
- Postpartum flare in IBD
Connections
- Inflammatory Bowel Disease Overview
- Crohn's vs Ulcerative Colitis: Key Differences
- Diagnostic Workup: Calprotectin, Colonoscopy, MRE
- Biologics: TNF, IL-23, and Integrin Inhibitors
- JAK Inhibitors and S1P Modulators
- Diet for IBD: SCD and Mediterranean
- IBD Surgery Decisions
- Extraintestinal Manifestations: Joints, Skin, Eyes
- IBD Fatigue, Anemia, and Nutrient Deficiencies
- Crohn's Disease
- Ulcerative Colitis
- Gestational Diabetes
- Pregnancy and Hashimoto's Thyroiditis
- Infertility
- Iron
- Anemia
- Lupus and Pregnancy