Aortic Aneurysm
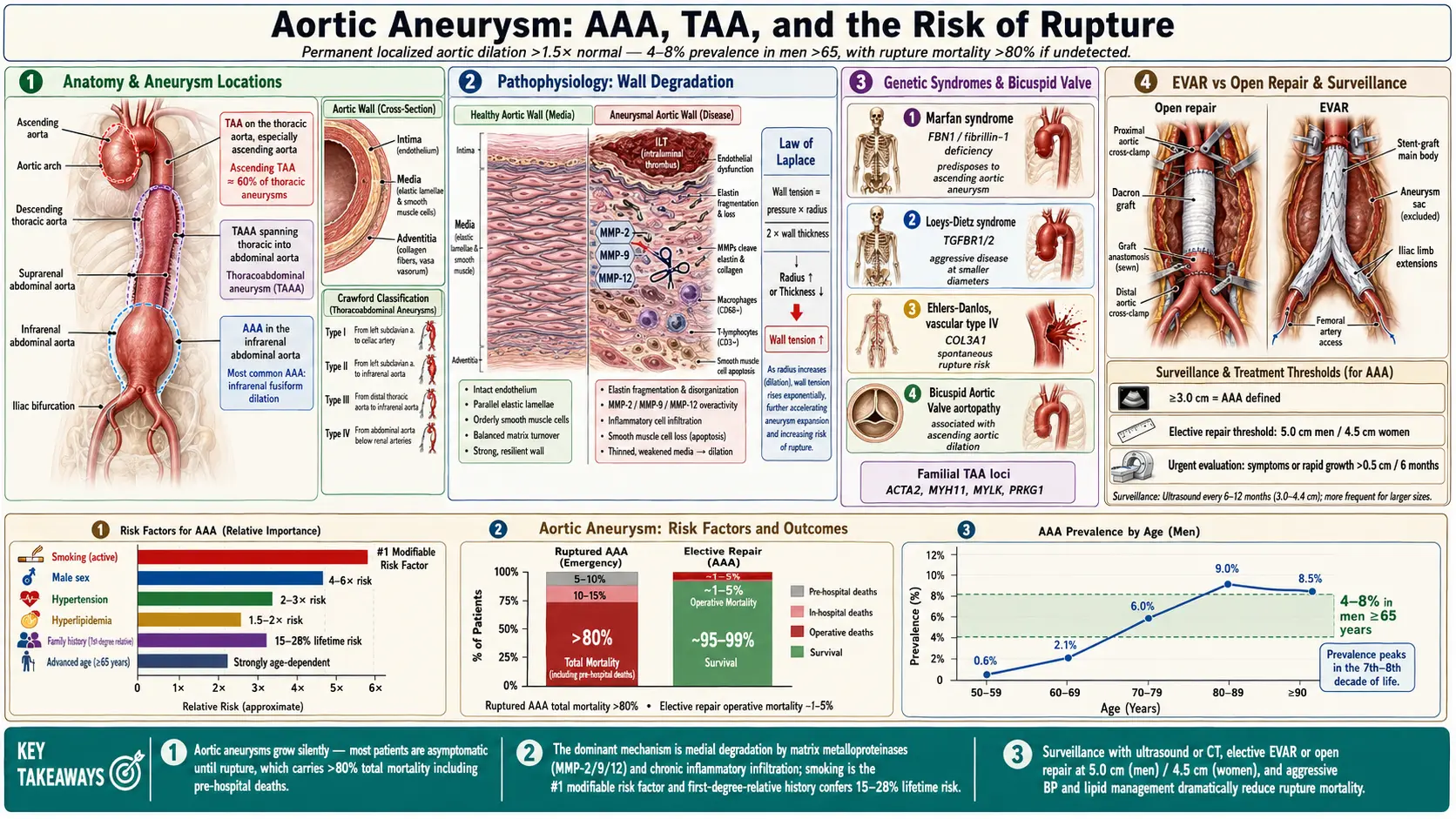
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Classification (Crawford and Other Systems)
- Treatment and Surgical Indications
- EVAR vs. Open Repair
- Surveillance Guidelines
- Complications
- Prognosis
- Prevention
- Recent Research
- Research Papers
- Connections
- Featured Videos
1. Overview
An aortic aneurysm is a pathological, permanent, and localized dilation of the aorta exceeding 1.5 times its normal diameter, or a diameter greater than 3.0 cm in the abdominal aorta and greater than 4.0–5.0 cm in the thoracic aorta. Aneurysms may occur anywhere along the aorta but are most commonly classified anatomically as abdominal aortic aneurysms (AAA) and thoracic aortic aneurysms (TAA), with a subset termed thoracoabdominal aortic aneurysms (TAAA) when they span both anatomic regions.
The condition carries significant morbidity and mortality, primarily due to the risk of rupture, dissection, or thromboembolic complications. Because most aneurysms are asymptomatic until a catastrophic event occurs, screening, surveillance, and timely surgical or endovascular intervention are cornerstone strategies in management. This article provides a comprehensive clinical review suitable for cardiovascular surgeons, vascular specialists, cardiologists, and internists involved in the care of patients with known or suspected aortic disease.
2. Epidemiology
Aortic aneurysms represent a significant public health burden worldwide. The estimated prevalence of AAA in men over 65 years of age ranges from 4% to 8% in Western populations, with a markedly lower prevalence of 0.5–1.5% in women of the same age group. The incidence of TAA is approximately 10.4 per 100,000 person-years, with ascending aortic aneurysms accounting for roughly 60% of thoracic cases.
Key epidemiological features include:
- Age: Peak incidence is in the seventh and eighth decades of life.
- Sex: Males are 4–6 times more likely to develop AAA than females, though females have worse outcomes after rupture.
- Race: Higher prevalence in white men; lower in Black, Hispanic, and Asian populations.
- Mortality: Ruptured AAA carries a total mortality (including pre-hospital deaths) exceeding 80%. Aortic aneurysm and dissection account for approximately 9,000–15,000 deaths annually in the United States.
- Incidental discovery: With increasing use of cross-sectional imaging, a growing proportion of aneurysms are detected incidentally.
The prevalence of AAA has declined in several high-income countries over the past two decades, attributed in part to reductions in smoking rates and improvements in cardiovascular risk factor management. Despite this trend, the aging global population sustains a high absolute burden of disease.
3. Pathophysiology
The pathogenesis of aortic aneurysm is multifactorial, involving inflammatory, proteolytic, hemodynamic, and genetic mechanisms that collectively degrade the structural integrity of the aortic wall.
Extracellular Matrix Degradation
Central to aneurysm formation is the breakdown of elastin and collagen within the tunica media and adventitia. Matrix metalloproteinases (MMPs), particularly MMP-2, MMP-9, MMP-12, and ADAM family members, are upregulated within the aneurysm wall and are elaborated by infiltrating macrophages, neutrophils, and smooth muscle cells. This proteolytic cascade leads to progressive loss of medial lamellar structure and biomechanical failure.
Inflammatory Infiltration
Histopathological analysis of AAA tissue reveals a dense transmural inflammatory infiltrate composed predominantly of macrophages, T lymphocytes, and B lymphocytes. This chronic inflammation drives ongoing proteolysis, neovascularization, and smooth muscle cell apoptosis. Oxidative stress and reactive oxygen species further amplify tissue injury. In contrast, non-inflammatory aneurysms may be seen in connective tissue disorders where the primary defect is intrinsic to the structural proteins themselves.
Hemodynamic Factors
Aortic wall stress is governed by the Law of Laplace: wall tension = (pressure × radius) / (2 × wall thickness). As the aneurysm enlarges, wall tension increases disproportionately, predisposing to accelerated expansion and eventual rupture. Infrarenal aortic flow characteristics — including increased pulsatility, oscillating shear stress, and intraluminal thrombus formation — contribute to localized ischemia and biological activity that accelerate degeneration.
Genetic and Connective Tissue Disorders
Heritable conditions affecting the aorta include:
- Marfan syndrome — FBN1 gene mutation causing fibrillin-1 deficiency, leading to cystic medial necrosis and predominantly ascending aortic dilation.
- Loeys-Dietz syndrome — TGFBR1/TGFBR2 mutations; aggressive aortic and arterial aneurysms at smaller diameters.
- Ehlers-Danlos syndrome type IV (vascular) — COL3A1 mutation; risk of spontaneous arterial rupture.
- Bicuspid aortic valve (BAV) aortopathy — Associated with ascending aortic dilation via flow-mediated and intrinsic wall abnormalities.
- Familial TAA — Mutations in ACTA2, MYH11, MYLK, PRKG1 and other smooth muscle cytoskeletal genes.
Intraluminal Thrombus
Most large AAAs harbor an intraluminal thrombus (ILT), which, paradoxically, does not appear to protect the wall from elevated stress. The ILT is metabolically active, harboring proteases and hypoxia-inducing conditions that further degrade the underlying aortic wall.
4. Etiology and Risk Factors
The etiology of aortic aneurysm spans degenerative, inflammatory, infectious, traumatic, and genetic categories.
Degenerative (Most Common)
The vast majority of AAAs and many TAAs result from atherosclerosis-related medial degeneration, though contemporary understanding recognizes that the process is distinct from typical luminal atherosclerosis. Shared risk factors include:
- Tobacco smoking — Single most important modifiable risk factor; risk increases with pack-year history and persists for years after cessation.
- Male sex — Hormonal influences including testosterone promote aneurysm development.
- Advanced age — Cumulative connective tissue degradation and hemodynamic stress.
- Hypertension — Elevated wall stress accelerates dilation.
- Hyperlipidemia and atherosclerosis — Correlates strongly with infrarenal AAA.
- Family history — First-degree relatives of AAA patients have a 15–28% lifetime risk; genetic loci on chromosomes 19q13 and others have been implicated.
- Caucasian ethnicity
Inflammatory Aneurysms
Accounting for approximately 5–10% of AAAs, inflammatory aortic aneurysms are characterized by perianeurysmal fibrosis, dense adventitial inflammation, and frequent adherence to surrounding structures (duodenum, left renal vein, inferior vena cava). They may present with constitutional symptoms, elevated ESR/CRP, and a distinctive CT appearance with periaortic soft tissue rind.
Infectious (Mycotic) Aneurysms
Mycotic aneurysms result from bacterial seeding of the aortic wall, most commonly by Salmonella species, Staphylococcus aureus, and Streptococcus species. They are typically saccular, rapidly expanding, and carry high rupture risk. Risk factors include bacteremia, endocarditis, immunosuppression, and IV drug use.
Post-Dissection Aneurysm
Chronic aortic dissection (particularly type B) may result in progressive aneurysmal dilation of the false lumen. These post-dissection aneurysms have distinct wall properties and require careful surveillance.
Connective Tissue and Genetic Causes
As outlined in the pathophysiology section, Marfan syndrome, Loeys-Dietz syndrome, BAV aortopathy, and familial TAA syndromes represent important genetic etiologies, often presenting at younger ages and requiring individualized surgical thresholds.
Traumatic Aneurysm
Blunt thoracic trauma, most commonly from deceleration injuries, can cause acute aortic injury at the aortic isthmus (just distal to the left subclavian artery). If not immediately fatal, these may evolve into pseudoaneurysms. Endovascular repair is now preferred for most cases.
5. Clinical Presentation
The clinical presentation of aortic aneurysm spans a wide spectrum from entirely asymptomatic (the most common scenario) to life-threatening emergency.
Asymptomatic Aneurysm
The majority of aortic aneurysms are discovered incidentally on imaging performed for unrelated indications (e.g., abdominal ultrasound, CT scan for nephrolithiasis or pulmonary disease). Routine screening programs also identify a significant proportion of cases.
Symptomatic (Unruptured) Aneurysm
Symptomatic but intact aneurysms may present with:
- Abdominal or back pain: Constant or intermittent dull aching, often in the periumbilical region or lumbar area; new-onset pain in a known aneurysm warrants urgent evaluation.
- Pulsatile abdominal mass: Palpable in thin patients with large AAAs; sensitivity is poor overall.
- Dysphagia lusoria or hoarseness (TAA): Compression of the esophagus or recurrent laryngeal nerve by thoracic aneurysms.
- Stridor or respiratory compromise: Large ascending or arch aneurysms may compress the trachea or mainstem bronchi.
- Superior vena cava syndrome: Compression by large ascending or arch aneurysm.
- Lower extremity ischemia: Distal embolization from mural thrombus.
- Aortoenteric fistula: Rare, presenting with upper GI bleeding and/or sepsis.
- Vertebral erosion: Chronic large aneurysms may erode into adjacent vertebral bodies, causing back pain.
Ruptured Aortic Aneurysm
Rupture constitutes a surgical emergency with extremely high mortality. The classic triad consists of:
- Sudden-onset severe abdominal, back, or flank pain — often described as tearing or ripping
- Hypotension or hemodynamic instability
- Pulsatile abdominal mass
However, this complete triad is present in only 25–50% of cases. Retroperitoneal ruptures may be temporarily tamponaded, allowing transient hemodynamic stability. Frank intraperitoneal rupture leads to rapid hemodynamic collapse and death without immediate intervention. Rupture into adjacent structures (aortocaval fistula, aortoenteric fistula) may produce distinctive presentations including continuous abdominal bruit, high-output cardiac failure, or GI hemorrhage.
Thoracic Aortic Aneurysm Presentations
Chest pain, back pain, or dyspnea may occur. Ascending aneurysms may cause aortic regurgitation due to annular dilation. Arch aneurysms may involve the great vessels. Descending thoracic aneurysms may erode into the esophagus (aortoesophageal fistula) presenting with massive hematemesis, or into the bronchial tree (aortobronchial fistula) with hemoptysis.
6. Diagnosis
Accurate diagnosis requires identification of the aneurysm, precise anatomic characterization, and assessment of associated pathology. Imaging is the cornerstone of diagnosis.
Ultrasound
Abdominal duplex ultrasonography is the primary screening modality for AAA. It is noninvasive, inexpensive, radiation-free, and has sensitivity exceeding 95% and specificity approaching 100% for infrarenal AAA. Limitations include poor visualization of suprarenal or iliac extension and inability to adequately image TAA. Ultrasound is appropriate for initial diagnosis and serial surveillance of stable infrarenal AAA.
Computed Tomography Angiography (CTA)
CTA with contrast is the definitive preoperative planning modality. It provides precise measurements of aneurysm diameter, length, and morphology; delineates the relationship to renal arteries, visceral vessels, and iliac bifurcation; identifies associated pathology (thrombus, calcification, occlusive disease); and is essential for EVAR planning. For thoracic and thoracoabdominal aneurysms, CTA with electrocardiographic gating is preferred to minimize cardiac motion artifact for the ascending aorta.
Key measurements reported in CTA include:
- Maximum outer-wall diameter (perpendicular to the aortic axis, not axial)
- Proximal and distal neck dimensions and angulation
- Length of proximal neck (must be ≥15 mm for standard EVAR)
- Iliac artery diameters and access vessel quality
- Presence of accessory renal arteries or anomalous anatomy
Magnetic Resonance Angiography (MRA)
MRA with gadolinium provides excellent soft-tissue characterization and avoids ionizing radiation. It is particularly useful for patients with contrast allergy (gadolinium-based agents may be used when iodinated contrast is contraindicated) and for assessing connective tissue disorders. Limitations include longer acquisition time, contraindication in patients with certain metallic implants, and potential for gadolinium-associated nephrogenic systemic fibrosis in advanced renal failure.
Aortography
Conventional catheter-based aortography has been largely supplanted by CTA/MRA for diagnostic purposes but remains used adjunctively during endovascular interventions to assess branch vessel involvement, confirm device positioning, and identify endoleaks after EVAR.
Echocardiography
Transthoracic echocardiography (TTE) provides assessment of the aortic root and proximal ascending aorta and is useful for evaluating associated bicuspid aortic valve, aortic regurgitation, and left ventricular function. Transesophageal echocardiography (TEE) offers superior visualization of the descending thoracic aorta and is useful intraoperatively.
Laboratory Evaluation
No serum biomarker has sufficient sensitivity or specificity for clinical diagnosis. Elevated serum MMP-9 and D-dimer may correlate with aneurysm activity and rupture risk in research settings. Standard preoperative labs include CBC, comprehensive metabolic panel, coagulation studies, type and screen, and creatinine/eGFR for contrast-related planning.
7. Classification (Crawford and Other Systems)
Several classification systems are used to characterize aortic aneurysms anatomically, guiding surgical planning, risk stratification, and prognostication.
Crawford Classification of Thoracoabdominal Aortic Aneurysms
Proposed by E. Stanley Crawford, this classification divides TAAAs based on extent of aortic involvement and relationship to visceral and renal vessels, directly informing operative approach and predicting spinal cord ischemia risk:
- Type I: Extends from the proximal descending thoracic aorta (distal to the left subclavian artery) to the abdominal aorta above the renal arteries. Involves most of the descending thoracic aorta but spares the renal segment.
- Type II: Extends from the proximal descending thoracic aorta to the infrarenal aorta, encompassing the entire thoracoabdominal segment. This type carries the highest risk of spinal cord ischemia (15–20%) due to the extensive intercostal and lumbar artery coverage required.
- Type III: Originates in the distal descending thoracic aorta (below T6) and extends into the abdominal aorta, including the visceral segment. Moderate spinal cord ischemia risk.
- Type IV: Entirely within the abdominal aorta from the diaphragm to the iliac bifurcation, encompassing the visceral and renal vessels. Lowest spinal cord ischemia risk.
- Type V (Safi modification): Involves the distal descending thoracic aorta from T6 to just above the celiac axis, without including the infrarenal segment. Intermediate risk profile.
Stanford and DeBakey Classifications (Aortic Dissection)
While primarily used for dissection, these classifications are relevant to post-dissection aneurysm:
- Stanford Type A: Involves the ascending aorta; requires emergency surgery.
- Stanford Type B: Confined to the descending aorta; managed medically or with TEVAR unless complicated.
- DeBakey Type I: Ascending + descending aorta.
- DeBakey Type II: Ascending aorta only.
- DeBakey Type III: Descending aorta only (IIIa limited, IIIb extending to abdomen).
Anatomic Location Classification
- Ascending aortic aneurysm: Aortic root to innominate artery origin.
- Aortic arch aneurysm: Involves the arch vessels (innominate, left common carotid, left subclavian).
- Descending thoracic aortic aneurysm (DTAA): From left subclavian artery to diaphragm.
- Abdominal aortic aneurysm (AAA): Infradiaphragmatic; suprarenal, juxtarenal, or infrarenal.
- Iliac artery aneurysm: Often concurrent with AAA; common iliac most affected.
Morphology
- Fusiform: Symmetric circumferential dilation involving the full aortic circumference; most common type.
- Saccular: Asymmetric outpouching; often mycotic, post-traumatic, or ulcer-penetrating; higher rupture risk per size.
8. Treatment and Surgical Indications
Management encompasses medical therapy for all patients, surveillance for those below repair thresholds, and intervention (endovascular or open surgical) when risk of rupture or complications outweighs procedural risk.
Medical Management
All patients with aortic aneurysm should receive aggressive medical management targeting modifiable risk factors:
- Smoking cessation: The most impactful intervention; smoking increases expansion rate approximately 0.4 mm/year and doubles rupture risk.
- Blood pressure control: Target systolic BP <130 mmHg. Beta-blockers (particularly in Marfan syndrome) reduce aortic wall stress; ACE inhibitors and ARBs may have additional benefits in reducing aneurysm progression via TGF-β pathway modulation (especially in connective tissue disorders).
- Statin therapy: Reduces cardiovascular mortality; some evidence suggests slowing AAA expansion and reducing operative risk, though evidence is not definitive for aneurysm growth.
- Antiplatelet therapy: For concurrent coronary artery disease; not proven to affect aneurysm progression.
- Doxycycline: MMP inhibitor properties; early promising results not confirmed in randomized trials (DOXY-1 trial).
Indications for Repair: Abdominal Aortic Aneurysm
Current guidelines from the Society for Vascular Surgery (SVS), American College of Cardiology/American Heart Association (ACC/AHA), and European Society for Vascular Surgery (ESVS) recommend repair based on diameter thresholds and other criteria:
- Diameter ≥5.5 cm in men: Repair indicated for standard-risk patients.
- Diameter ≥5.0 cm in women: Lower threshold recommended due to higher rupture risk at smaller diameters and larger size relative to body surface area.
- Rapid expansion ≥0.5 cm in 6 months or ≥1.0 cm/year: Indicates accelerated growth and prompts repair regardless of absolute diameter.
- Symptomatic aneurysm: New-onset pain attributable to aneurysm; urgent repair required.
- Rupture: Emergency repair.
- Saccular morphology or mycotic etiology: Lower threshold for intervention.
- Aortoenteric or aortocaval fistula: Emergency repair.
Indications for Repair: Thoracic Aortic Aneurysm
- Ascending aorta ≥5.5 cm: Repair recommended for standard-risk patients.
- Ascending aorta ≥5.0 cm: Repair considered when low surgical risk or when concomitant cardiac surgery (e.g., CABG, aortic valve replacement) is planned.
- Ascending aorta ≥4.5 cm with Marfan or Loeys-Dietz syndrome: Earlier intervention warranted due to risk of dissection at smaller diameters.
- Bicuspid aortic valve aortopathy ≥5.5 cm (or ≥5.0 cm with risk factors): Repair recommended.
- Descending thoracic aorta ≥5.5–6.0 cm: Repair considered; TEVAR preferred if anatomy suitable.
- Rate of growth >0.5 cm/year: Prompts earlier intervention at any diameter.
- Symptoms, dissection, or other complications: Prompt repair.
Indications for Repair: Thoracoabdominal Aortic Aneurysm
- Diameter ≥6.0 cm in standard-risk patients (open or branched/fenestrated endovascular repair)
- Diameter ≥5.5 cm in Marfan or other connective tissue disorder
- Symptomatic aneurysm regardless of size
9. EVAR vs. Open Repair
The choice between endovascular aortic repair (EVAR) and open surgical repair is determined by anatomical suitability, patient physiologic status, institutional expertise, and long-term durability considerations.
Endovascular Aortic Repair (EVAR / TEVAR)
EVAR for infrarenal AAA involves percutaneous or femoral cutdown delivery of a modular stent-graft system that excludes the aneurysm sac from arterial pressure. TEVAR (thoracic EVAR) applies the same principle to descending thoracic aneurysms.
Anatomic requirements for standard EVAR:
- Proximal neck length ≥15 mm (ideally ≥20 mm)
- Neck angulation <60°
- Neck diameter 18–32 mm
- Adequate iliac access (≥7–8 mm diameter, limited tortuosity)
- Absence of circumferential thrombus or calcification in the neck
Advantages of EVAR:
- Lower 30-day mortality vs. open repair (approximately 1.4% vs. 4.2% in EVAR-1 trial)
- Reduced blood loss, ICU stay, hospital length of stay
- Suitable for high-risk surgical patients (cardiopulmonary comorbidities)
- Less physiologic stress; quicker recovery
Disadvantages and limitations of EVAR:
- Endoleak: Persistent blood flow into the aneurysm sac. Types I (proximal/distal attachment) and III (fabric tear/component separation) require urgent intervention. Type II (collateral back-filling, typically from lumbar or IMA) is managed conservatively unless sac enlarges. Type IV (graft porosity) is rare with modern devices.
- Reintervention rate: Significantly higher than open repair; approximately 20–30% at 10 years.
- Graft migration and late rupture: Particularly in hostile necks or with suboptimal device sizing.
- Sac expansion without endoleak (endotension): Poorly understood phenomenon requiring continued surveillance.
- Lifelong surveillance: CTA at 1 and 12 months, then annually; significant radiation exposure over time.
- Survival equivalence lost at 2–3 years post-procedure (EVAR-1, DREAM, OVER trials).
Advanced Endovascular Techniques
- Fenestrated EVAR (FEVAR): Custom-fabricated or off-the-shelf devices with fenestrations aligning to renal/visceral arteries; extends EVAR to juxtarenal and suprarenal AAAs.
- Branched EVAR (BEVAR): For TAAAs; uses branched components to perfuse celiac, SMA, and renal arteries.
- Parallel graft techniques (chimney/snorkel/periscope): Used in urgent cases or when fenestrated devices are unavailable.
Open Surgical Repair
Open AAA repair involves aortic clamping, aneurysm sac opening, mural thrombus removal, and placement of a prosthetic Dacron graft (tube or bifurcated). The transperitoneal midline approach or retroperitoneal approach (preferred for suprarenal, hostile abdomen, or horseshoe kidney) is used.
Advantages of open repair:
- Definitive repair with excellent long-term durability
- No need for lifelong imaging surveillance post-repair
- Suitable for anatomically challenging cases (short necks, highly angulated, iliac occlusive disease)
- Proven long-term survival benefit over EVAR beyond 2–3 years in anatomically eligible patients
Disadvantages of open repair:
- Higher 30-day mortality than EVAR (approximately 3–5% in elective cases at experienced centers)
- Significant physiologic stress; unsuitable for high-risk patients
- Longer recovery (4–6 weeks)
- Risks include myocardial infarction, renal failure, bowel ischemia, wound complications, sexual dysfunction (retrograde ejaculation)
- Aortic cross-clamp time and level (infrarenal vs. suprarenal) determine organ ischemia risk
Open Repair of TAAA
Open TAAA repair (Crawford-type) is among the most physiologically demanding vascular operations. Spinal cord protection strategies are critical and include:
- Cerebrospinal fluid (CSF) drainage to maintain spinal cord perfusion pressure
- Left heart bypass or partial cardiopulmonary bypass to maintain distal perfusion during aortic cross-clamp
- Selective visceral and renal perfusion
- Moderate hypothermia
- Reimplantation of critical intercostal arteries (T8–L1 segment)
- Electrophysiologic monitoring (SSEP, MEP)
10. Surveillance Guidelines
Evidence-based surveillance protocols guide frequency of imaging for known aneurysms below repair thresholds, post-repair follow-up, and screening of high-risk populations.
Screening
The U.S. Preventive Services Task Force (USPSTF) and SVS recommend one-time ultrasound screening for AAA in:
- Men aged 65–75 who have ever smoked (≥100 cigarettes lifetime) — Grade B recommendation.
- Men aged 65–75 who have never smoked — Selective screening at clinician discretion, especially with family history.
- First-degree relatives of AAA patients — Ultrasound screening at age 65 (or 55 if family history significant).
- Current USPSTF guidelines do not recommend routine screening in women; SVS recommends considering screening in women aged 65+ with smoking history or family history.
Surveillance of Known AAA (Below Repair Threshold)
Based on initial diameter:
- 2.5–2.9 cm: Ultrasound every 5 years.
- 3.0–3.9 cm: Ultrasound every 3 years.
- 4.0–4.9 cm: Ultrasound every 12 months.
- 5.0–5.4 cm (men) or 4.5–4.9 cm (women): Ultrasound every 6 months; CTA annually for anatomic characterization and repair planning.
- Any rapid expansion (≥0.5 cm/6 months): Prompt evaluation for repair.
Surveillance of Known TAA (Below Repair Threshold)
- Initial imaging: CTA (ECG-gated for ascending) to establish baseline.
- <4.5 cm ascending: MRA or CTA every 5 years.
- 4.5–5.4 cm ascending: CTA/MRA every 6–12 months.
- Descending TAA <4.5 cm: Annual CTA or MRA.
- Descending TAA 4.5–5.5 cm: CTA every 6 months.
- Patients with Marfan, Loeys-Dietz, or BAV aortopathy require more frequent imaging (every 6 months until stability confirmed, then annually).
Post-EVAR Surveillance
- CTA at 1 month and 12 months post-procedure.
- If no endoleak, stable sac: CTA or duplex ultrasound annually.
- Any endoleak, sac growth, or device concern: more frequent imaging and intervention planning.
- Duplex ultrasound is an acceptable alternative to CTA for stable post-EVAR surveillance to reduce cumulative radiation.
Post-Open Repair Surveillance
- CTA or duplex ultrasound at 1 month post-repair.
- If stable, imaging every 3–5 years to assess for anastomotic pseudoaneurysm, graft complications, or new aneurysm formation at adjacent segments.
- Annual clinical examination recommended.
11. Complications
Complications may arise from the aneurysm itself or from its repair.
Aneurysm-Related Complications
- Rupture: The most feared complication; overall mortality exceeds 80% when accounting for pre-hospital deaths. Even with emergency repair, mortality ranges from 40–70%.
- Aortic dissection: More common with TAA, particularly in connective tissue disorders.
- Thromboembolism: Mural thrombus may embolize distally, causing lower extremity ischemia (blue toe syndrome, limb-threatening ischemia) or renal infarction.
- Aortoenteric fistula: Rare but life-threatening; secondary fistula from prior aortic graft more common.
- Aortocaval fistula: High-output cardiac failure, continuous abdominal bruit, venous hypertension.
- Vertebral erosion and neurologic compromise.
Perioperative Complications of Open Repair
- Myocardial infarction (most common cause of 30-day mortality)
- Renal failure (suprarenal cross-clamp, atheroembolism)
- Spinal cord ischemia / paraplegia (particularly with TAAA)
- Bowel ischemia (left colon from IMA sacrifice)
- Pulmonary complications (pneumonia, prolonged ventilation)
- Wound complications, anastomotic bleeding
- Retrograde ejaculation (disruption of autonomic plexus)
- Late graft infection (rare; highly morbid)
Post-EVAR Complications
- Endoleak (Types I–V)
- Graft migration
- Limb occlusion / thrombosis
- Access vessel injury (iliac rupture, dissection)
- Post-implantation syndrome (fever, elevated CRP)
- Renal embolism from stent-graft deployment
- Late sac expansion and rupture
12. Prognosis
Prognosis depends on aneurysm size, rupture status, patient comorbidities, and type of repair performed.
- Small AAA (<4.0 cm): Annual expansion rate approximately 1–3 mm/year; rupture risk <1% per year; 5-year survival comparable to age-matched controls but limited by cardiovascular comorbidities.
- AAA 4.0–5.4 cm: Annual rupture risk 0.5–5%; expansion rate increases with size; ongoing cardiovascular mortality remains the primary cause of death.
- AAA ≥5.5 cm untreated: Annual rupture risk 10–20%; median survival without repair significantly reduced.
- Elective open AAA repair: 30-day mortality 1–5% at experienced centers; 5-year survival approximately 60–70%.
- Elective EVAR: 30-day mortality <2%; equivalent long-term survival to open repair; higher reintervention rate.
- Ruptured AAA (operative): 30-day mortality 40–50%; higher in women and older patients.
- TAAA open repair: Mortality 5–15% depending on extent; paraplegia rate 5–20% for type II.
- Patients with Marfan syndrome: Improved survival with prophylactic root replacement; life expectancy approaches near-normal with appropriate management.
Cardiovascular disease (coronary artery disease, stroke) is the leading cause of long-term mortality in surgically repaired aneurysm patients, underscoring the importance of comprehensive cardiovascular risk reduction.
13. Prevention
Primary prevention focuses on modifying risk factors for aneurysm formation and progression:
- Smoking cessation: The single most effective intervention for both primary prevention and slowing expansion.
- Hypertension management: Tight blood pressure control reduces wall stress and cardiovascular mortality.
- Lipid management: Statin therapy reduces overall cardiovascular events; data on direct aneurysm prevention are limited.
- Screening programs: Population-based one-time ultrasound screening in high-risk groups has been shown to reduce AAA-related mortality by 40–50% (MASS, Viborg, Western Australia trials).
- Genetic counseling and testing: Recommended for patients with connective tissue disorders, familial TAA/AAA, or multiple affected family members; enables prophylactic monitoring and timely repair.
- Avoidance of fluoroquinolones: Emerging evidence (FDA warning, 2018) links fluoroquinolone antibiotics to increased risk of aortic aneurysm and dissection; caution warranted in high-risk patients.
- Physical activity: Moderate exercise is safe and beneficial for cardiovascular health; vigorous isometric exercise (heavy weightlifting) is cautioned against in patients with known large aneurysms due to acute BP elevation.
14. Recent Research
Significant advances have been made across the spectrum of aortic aneurysm research:
- Pharmacologic inhibition of aneurysm growth: Beyond statins and beta-blockers, trials of metformin (MAAAGI trial), doxycycline (DOXY-1), and angiotensin receptor blockers in non-Marfan AAA have shown modest or inconclusive results. The AARDVARK trial investigated atenolol vs. perindopril in BAV aortopathy. Ongoing trials examine IL-6 inhibition and MMP-directed therapies.
- Branched and fenestrated EVAR expansion: Multicenter registry data demonstrate improving outcomes for complex EVAR (FEVAR, BEVAR) for TAAAs, with centers of excellence reporting mortality and paraplegia rates approaching those of open repair with significantly reduced physiologic burden.
- Machine learning and AI for rupture prediction: Computational fluid dynamics and finite element analysis models incorporating aneurysm geometry, wall stress distribution, and thrombus properties are being validated as adjuncts to diameter-based repair thresholds. AI-driven peak wall stress calculation may identify high-risk aneurysms below conventional repair thresholds.
- Biomarkers: Circulating MMP-9, neutrophil-to-lymphocyte ratio, and microRNA profiles are being investigated as non-imaging biomarkers of aneurysm activity and rupture risk.
- Genetic discoveries: Whole-exome and genome-wide association studies continue to identify novel loci associated with sporadic AAA (e.g., DAB2IP, LRP1, SORT1); polygenic risk scores are under development.
- Post-EVAR surveillance optimization: Studies comparing contrast-enhanced ultrasound (CEUS) to CTA for endoleak detection support CEUS as a radiation-free alternative with high sensitivity for type II endoleaks.
- EVAR vs. open repair long-term outcomes: The OVER trial 14-year follow-up confirmed no significant difference in overall mortality between EVAR and open repair in men fit for both procedures; EVAR's early advantage is offset by higher late reinterventions.
- Anti-inflammatory strategies: Low-dose colchicine, IL-1β inhibition (canakinumab in the CANTOS trial subgroup), and neutrophil elastase inhibitors are being explored for slowing AAA progression.
Research Papers
The following PubMed topic searches return current peer-reviewed literature relevant to this condition. Each link opens a live PubMed query.
- Abdominal aortic aneurysm
- Thoracic aortic aneurysm
- Aortic aneurysm screening
- Endovascular aneurysm repair (EVAR)
- Open surgical repair aortic aneurysm
- Aortic aneurysm rupture
- Marfan syndrome aortic aneurysm
- Aortic dissection
- Aortic aneurysm risk factors
- Aortic aneurysm guidelines
- Aortic aneurysm CT imaging
- Aortic aneurysm mortality
Connections
- Hypertension
- Atherosclerosis
- Coronary Artery Disease
- Peripheral Artery Disease
- Cardiovascular Disease
- Stroke
- Valvular Heart Disease
- Lipid Panel
- Full Body MRI
- Deep Vein Thrombosis
- Omega-3 Fatty Acids
- Anti-Inflammatory Diet
- Chest Pain
- Oxidative Stress
- Garlic
- Ehlers-Danlos Syndrome