Vitamin D3 and Immune System Regulation
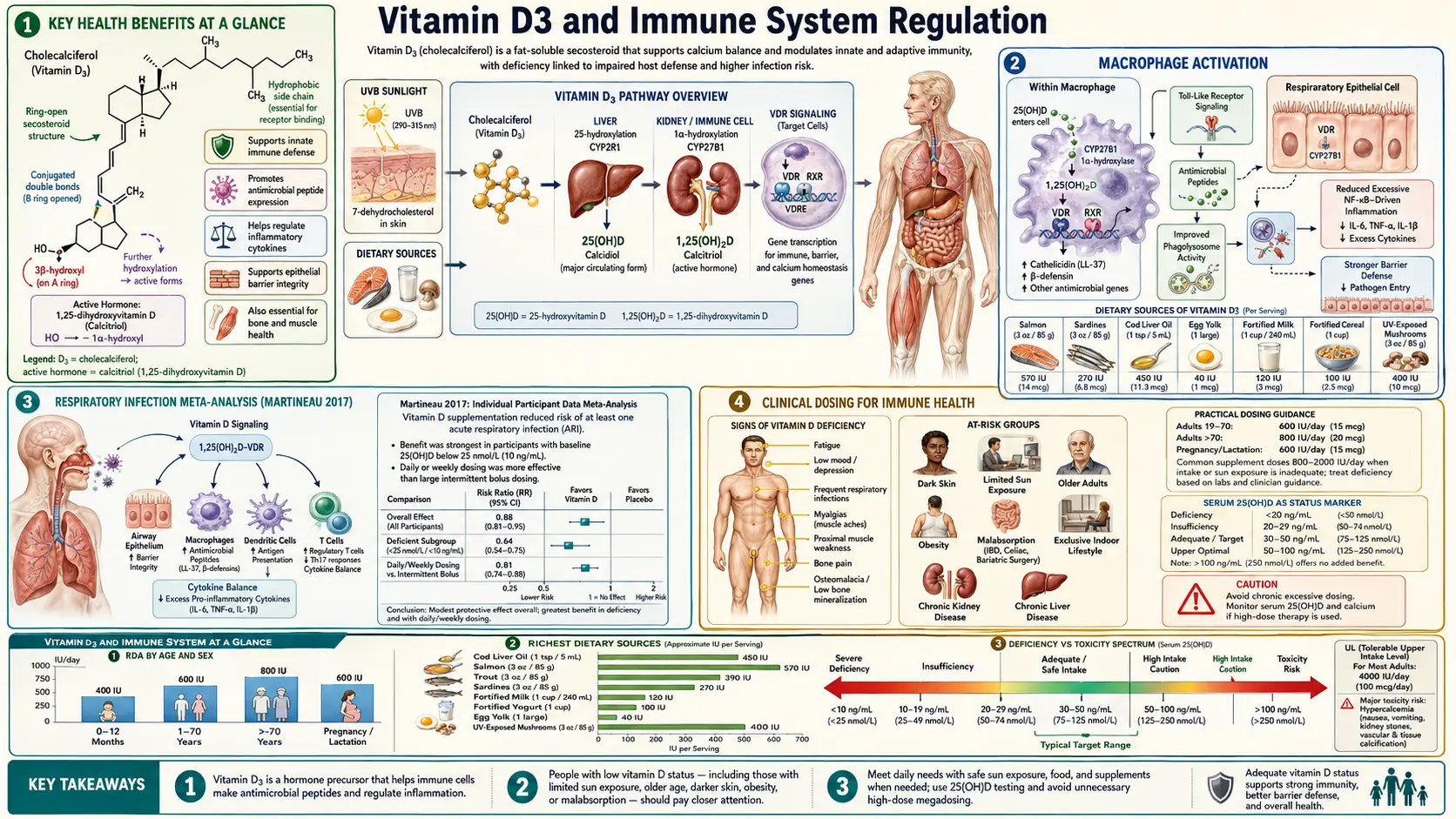
The discovery that virtually all immune cells express the vitamin D receptor (VDR) and that many possess the enzymatic machinery to convert 25(OH)D to active calcitriol locally has fundamentally changed our understanding of vitamin D3. Far beyond its classical role in calcium metabolism, vitamin D3 functions as a powerful immunomodulator, enhancing innate antimicrobial defenses while simultaneously regulating adaptive immune responses to prevent autoimmunity. The 2017 Martineau individual-participant-data meta-analysis in BMJ, pooling 25 randomized trials and 11,321 participants, definitively established that vitamin D supplementation reduces acute respiratory infection risk — with a striking 70% reduction in participants starting below 10 ng/mL. The 2022 VITAL autoimmune substudy further showed that 2,000 IU/day of D3 reduced confirmed autoimmune disease incidence by 22% in older adults.
Table of Contents
- Key Health Benefits at a Glance
- Vitamin D Receptor (VDR) in Immune Cells
- Cathelicidin (LL-37) Induction
- Defensin Production
- Macrophage Activation
- Th1/Th2/Th17/Treg Balance
- Autoimmune Disease Prevention
- VITAL Study: Autoimmune Disease Results
- Respiratory Infection Meta-Analysis (Martineau 2017)
- Tuberculosis Connection
- Cancer Immunosurveillance
- Latitude Gradient and Seasonal Immunity
- Clinical Dosing for Immune Health
- Research Papers and References
- Connections
- Featured Videos
Key Health Benefits at a Glance
The following is a high-level summary of the evidence-backed immune benefits of adequate vitamin D3 status. Each is explored in more depth below, and the supporting studies are linked in the Research Papers section.
- Cuts respiratory infections – Martineau et al. (2017, BMJ) pooled 25 RCTs of 11,321 participants and found a 12% reduction in acute respiratory infections overall, rising to 70% in those starting below 10 ng/mL.
- Reduces autoimmune disease incidence – The VITAL autoimmune substudy showed a 22% reduction in incident autoimmune disease (RA, psoriasis, polymyalgia rheumatica, autoimmune thyroid, IBD) over 5 years with 2,000 IU/day.
- Induces the antimicrobial peptide cathelicidin – The CAMP gene carries a vitamin D response element; adequate 25(OH)D is required for macrophages to manufacture LL-37, the broad-spectrum peptide that kills bacteria, fungi, and enveloped viruses.
- Promotes regulatory T cells – Calcitriol expands FoxP3+ Tregs and tolerogenic dendritic cells, suppressing the Th17 responses implicated in MS, RA, psoriasis, and IBD.
- Protects pancreatic beta cells – Finnish birth cohort data link 2,000 IU/day in infancy with an 80% reduction in type 1 diabetes risk over 30 years.
- Lowers MS risk along a latitude gradient – U.S. military data show a 62% lower MS incidence in service members with 25(OH)D above 40 ng/mL compared to below 25 ng/mL.
- Enhances macrophage autophagy – Vitamin D-sufficient macrophages can clear intracellular M. tuberculosis via cathelicidin-driven autophagy, linking the pre-antibiotic heliotherapy era to modern molecular immunology.
- Balances inflammation – Calcitriol suppresses TNF-alpha, IL-6, and IL-1-beta while upregulating IL-10, promoting pathogen clearance without excessive tissue damage.
Vitamin D Receptor (VDR) in Immune Cells
- Widespread Expression – The VDR is expressed in monocytes, macrophages, dendritic cells, neutrophils, natural killer cells, and both T and B lymphocytes. This ubiquitous presence indicates that vitamin D signaling is integral to immune function across both the innate and adaptive arms.
- 1-Alpha-Hydroxylase (CYP27B1) – Immune cells, particularly macrophages and dendritic cells, express CYP27B1, the enzyme that converts circulating 25(OH)D to active 1,25(OH)2D (calcitriol). Unlike renal CYP27B1, which is regulated by PTH and calcium, immune cell CYP27B1 is regulated by immune signals such as toll-like receptor (TLR) activation and interferon-gamma. This allows immune cells to produce calcitriol locally in response to infection, independently of systemic calcium homeostasis.
- Autocrine and Paracrine Signaling – The locally produced calcitriol acts in an autocrine manner (on the producing cell) and paracrine manner (on neighboring immune cells) to modulate immune responses. This localized production depends entirely on the availability of the substrate 25(OH)D, directly linking circulating vitamin D status to immune competence.
- Genomic Effects – Once calcitriol binds the VDR, the receptor forms a heterodimer with the retinoid X receptor (RXR) and binds to vitamin D response elements (VDREs) in the promoter regions of target genes. In immune cells, calcitriol-VDR signaling regulates the expression of over 200 genes involved in antimicrobial defense, inflammation, and immune tolerance.
Cathelicidin (LL-37) Induction
- Discovery – The landmark 2006 study by Liu et al. demonstrated that activation of TLR2/1 on human macrophages by mycobacterial lipopeptides upregulates CYP27B1 and VDR, leading to local calcitriol production, which in turn induces the antimicrobial peptide cathelicidin (LL-37). This elegant mechanism directly linked vitamin D status to the innate ability to kill intracellular pathogens.
- Antimicrobial Properties – LL-37 is a 37-amino acid peptide with broad-spectrum antimicrobial activity. It kills bacteria by disrupting their cell membranes through electrostatic interactions with negatively charged lipid bilayers. LL-37 is also active against fungi, enveloped viruses (including influenza A and respiratory syncytial virus), and mycobacteria (including M. tuberculosis).
- Vitamin D Dependence – The cathelicidin gene (CAMP) contains a vitamin D response element in its promoter, making its expression directly dependent on VDR activation. Serum from vitamin D-deficient individuals fails to support cathelicidin induction in macrophages in vitro, while serum from vitamin D-sufficient individuals supports robust LL-37 production. This is one of the clearest demonstrations of how vitamin D deficiency impairs innate immunity.
- Beyond Direct Killing – LL-37 also functions as a chemoattractant, recruiting neutrophils, monocytes, and T cells to sites of infection. It modulates TLR signaling, promotes autophagy (an intracellular pathogen-clearing mechanism), and neutralizes bacterial endotoxin (lipopolysaccharide), reducing the risk of septic shock.
Defensin Production
- Beta-Defensins – Vitamin D induces the expression of beta-defensin 2 (DEFB4) and beta-defensin 4 in epithelial cells of the respiratory tract, skin, and gastrointestinal tract. These antimicrobial peptides provide a chemical barrier at mucosal surfaces, killing pathogens before they can establish infection.
- Barrier Defense – The induction of defensins by vitamin D strengthens the first line of defense at epithelial surfaces, which are the primary entry points for respiratory, gastrointestinal, and urogenital pathogens. This barrier-enhancing function helps explain the epidemiological association between vitamin D deficiency and increased susceptibility to mucosal infections.
Macrophage Activation
- Enhanced Phagocytosis – Calcitriol promotes macrophage differentiation from monocyte precursors and enhances phagocytic activity. Vitamin D-stimulated macrophages show increased expression of pattern recognition receptors and improved capacity to engulf and process pathogens.
- Autophagy Induction – Vitamin D promotes autophagy in macrophages through induction of the autophagy gene BECN1 (beclin-1) and cathelicidin-mediated activation of the autophagy machinery. Autophagy is a critical mechanism for killing intracellular pathogens such as M. tuberculosis that survive within phagosomes by preventing phagolysosomal fusion.
- Anti-Inflammatory Shift – While vitamin D enhances microbial killing, it simultaneously promotes an anti-inflammatory macrophage phenotype by suppressing pro-inflammatory cytokines (TNF-alpha, IL-6, IL-1-beta) and upregulating anti-inflammatory cytokines (IL-10). This balanced response enhances pathogen clearance while limiting collateral tissue damage.
Th1/Th2/Th17/Treg Balance
Vitamin D3 exerts profound effects on the differentiation and function of CD4+ T helper cell subsets, shifting the balance toward immune tolerance.
- Th1 Suppression – Calcitriol inhibits the differentiation and cytokine production of Th1 cells, which produce interferon-gamma and drive cell-mediated immunity. While Th1 responses are essential for intracellular pathogen defense, excessive Th1 activity underlies many organ-specific autoimmune diseases. Vitamin D moderates Th1 responses without eliminating them.
- Th2 Modulation – The effect of vitamin D on Th2 cells (which produce IL-4, IL-5, IL-13 and drive humoral immunity and allergic responses) is more nuanced. Some studies suggest vitamin D mildly promotes Th2 responses, potentially contributing to allergic sensitization in early life when given in excess, though this remains controversial.
- Th17 Suppression – Calcitriol strongly suppresses the differentiation of Th17 cells, which produce IL-17 and IL-22 and are major drivers of inflammatory and autoimmune pathology. Th17 cells are implicated in multiple sclerosis, rheumatoid arthritis, psoriasis, and inflammatory bowel disease. The suppression of Th17 differentiation is one of the most therapeutically relevant immune effects of vitamin D.
- Regulatory T Cell (Treg) Promotion – Vitamin D promotes the differentiation and function of FoxP3+ regulatory T cells, which suppress excessive immune responses and maintain self-tolerance. Tregs are critical for preventing autoimmunity, and their enhancement by vitamin D is considered a key mechanism underlying its protective effect against autoimmune diseases.
- Dendritic Cell Tolerogenesis – Vitamin D promotes a tolerogenic dendritic cell phenotype characterized by reduced expression of MHC class II molecules and co-stimulatory molecules (CD80, CD86), decreased IL-12 production, and increased IL-10 secretion. These tolerogenic dendritic cells preferentially induce Treg differentiation rather than effector T cell activation.
Autoimmune Disease Prevention
Epidemiological, genetic, and clinical evidence strongly links vitamin D status to autoimmune disease risk.
- Multiple Sclerosis (MS) – MS prevalence follows a striking latitude gradient, increasing with distance from the equator. Higher 25(OH)D levels are associated with reduced MS risk in prospective studies. Among U.S. military personnel, those with 25(OH)D levels above 40 ng/mL had a 62 percent lower risk of developing MS compared to those with levels below 25 ng/mL. Vitamin D supplementation may also reduce relapse rates in established MS, though evidence from randomized trials remains limited.
- Type 1 Diabetes (T1D) – A Finnish birth cohort study found that infants receiving 2,000 IU of vitamin D daily during the first year of life had an 80 percent reduced risk of developing T1D over the next 30 years. Meta-analyses confirm a consistent inverse association between vitamin D supplementation in infancy and T1D risk. Mechanistically, vitamin D protects pancreatic beta cells from autoimmune destruction by suppressing Th1/Th17 responses and promoting Tregs.
- Rheumatoid Arthritis (RA) – Low vitamin D levels are associated with increased RA risk and disease activity in observational studies. The Iowa Women's Health Study found that higher vitamin D intake was associated with a reduced risk of RA. Vitamin D supplementation may reduce disease activity in some RA patients, though evidence is inconsistent.
- Inflammatory Bowel Disease (IBD) – Both Crohn's disease and ulcerative colitis show latitude-dependent prevalence patterns and are associated with low vitamin D levels. Vitamin D deficiency is both a consequence (due to malabsorption and reduced sun exposure) and a potential contributor to IBD pathogenesis through effects on intestinal barrier function and mucosal immunity.
VITAL Study: Autoimmune Disease Results
- Study Design – The VITAL study was a large randomized, double-blind, placebo-controlled trial of 25,871 U.S. adults (men aged 50 and older, women aged 55 and older) receiving 2,000 IU of vitamin D3 daily, 1 gram of omega-3 fatty acids daily, both, or placebo, with a median follow-up of 5.3 years.
- Primary Autoimmune Finding – The vitamin D arm showed a 22 percent reduction in confirmed autoimmune disease incidence (hazard ratio 0.78, 95% confidence interval 0.61 to 0.99, p = 0.045). This represented the first large randomized trial evidence that vitamin D supplementation can prevent autoimmune diseases.
- Diseases Included – The autoimmune endpoint included rheumatoid arthritis, polymyalgia rheumatica, autoimmune thyroid disease, psoriasis, and inflammatory bowel disease, among others. The effect appeared to strengthen over time, with greater risk reduction in years 3 through 5 compared to the first two years.
- Clinical Significance – This finding is considered landmark because it demonstrates that a simple, safe, and inexpensive intervention (2,000 IU vitamin D3 daily) can meaningfully reduce the burden of autoimmune disease in older adults. The result has influenced clinical practice guidelines and public health recommendations.
Respiratory Infection Meta-Analysis (Martineau 2017)
- Study Overview – The individual participant data meta-analysis by Martineau et al., published in the BMJ in 2017, analyzed 25 randomized controlled trials including 11,321 participants aged 0 to 95 years. This rigorous methodology (using individual-level data rather than study-level summaries) provided the strongest available evidence on vitamin D and respiratory infections.
- Overall Results – Vitamin D supplementation reduced the risk of acute respiratory tract infections by 12 percent overall (adjusted odds ratio 0.88, 95% CI 0.81 to 0.96). The benefit was seen across all age groups.
- Subgroup with Greatest Benefit – The most striking finding was in participants with baseline 25(OH)D levels below 10 ng/mL (25 nmol/L), who experienced a 70 percent reduction in respiratory infection risk with daily or weekly supplementation (adjusted OR 0.30, 95% CI 0.17 to 0.53). This suggests that correcting frank deficiency has the most dramatic protective effect.
- Dosing Frequency – Daily or weekly dosing was protective, while intermittent bolus dosing (monthly or less frequently) showed no significant benefit. This finding is consistent with the hypothesis that maintaining stable circulating 25(OH)D levels is important for continuous immune readiness.
Tuberculosis Connection
- Historical Context – Before the antibiotic era, tuberculosis was treated with sunlight therapy (heliotherapy) and cod liver oil, both of which increase vitamin D levels. The Nobel Prize-winning use of UV light therapy for lupus vulgaris (cutaneous TB) by Niels Finsen in 1903 likely worked in part through vitamin D-mediated cathelicidin induction.
- Mechanistic Link – The TLR2/1-mediated vitamin D-cathelicidin pathway was discovered specifically in the context of macrophage defense against M. tuberculosis. Vitamin D-sufficient macrophages can kill intracellular M. tuberculosis through cathelicidin-mediated autophagy; vitamin D-deficient macrophages cannot.
- Epidemiological Evidence – Vitamin D deficiency is a consistent risk factor for tuberculosis in observational studies, and TB patients typically have lower 25(OH)D levels than matched controls. Immigrants from equatorial countries to northern latitudes show increased TB susceptibility as their vitamin D levels decline.
- Clinical Trials – Adjunctive vitamin D supplementation in TB treatment has shown mixed results in randomized trials. A trial by Martineau et al. found that high-dose vitamin D accelerated sputum culture conversion in patients with the tt genotype of the TaqI VDR polymorphism, suggesting pharmacogenomic factors influence treatment response.
Cancer Immunosurveillance
- VDR Expression in Immune and Epithelial Cells – The vitamin D receptor is expressed in nearly every cell of the immune system as well as in colonic, breast, and prostatic epithelium. Calcitriol signaling through VDR promotes cell-cycle arrest (upregulation of p21 and p27), differentiation, and apoptosis in premalignant cells, while simultaneously supporting the cytotoxic activity of natural killer cells and CD8+ T lymphocytes that eliminate transformed cells.
- Colorectal Cancer – Prospective cohort studies and meta-analyses of observational data consistently show an inverse association between circulating 25(OH)D and colorectal cancer incidence. Higher vitamin D status is also associated with improved survival after colorectal cancer diagnosis. Proposed mechanisms include Wnt/beta-catenin pathway suppression, enhanced mucosal immunity, and modulation of the colonic microbiome.
- VITAL Cancer Endpoints – The VITAL trial did not find a significant reduction in overall cancer incidence with 2,000 IU daily vitamin D3 over 5 years, but post-hoc analyses suggested reductions in cancer mortality and in advanced (metastatic or fatal) cancer, particularly among participants with a normal body mass index. These findings support the hypothesis that vitamin D primarily affects cancer progression rather than initiation.
- Clinical Implications – While vitamin D supplementation is not promoted as a cancer prevention strategy in isolation, maintaining 25(OH)D levels in the sufficient range (30 to 50 ng/mL) is consistent with both immune competence and a lower observed burden of several cancers in population studies.
Latitude Gradient and Seasonal Immunity
- Solar UVB and Vitamin D Synthesis – Cutaneous vitamin D3 production requires UVB radiation (wavelength 290 to 315 nm). At latitudes above approximately 35 degrees North (or South), UVB intensity is insufficient for vitamin D synthesis during winter months. In Boston (42 degrees North), there is essentially no cutaneous vitamin D production from November through February. In northern Scandinavia, the "vitamin D winter" extends from October through March.
- Seasonal Infection Patterns – The winter peak in respiratory infections (influenza, RSV, common cold) coincides with the nadir of population vitamin D levels. While indoor crowding and low humidity also contribute, the seasonal decline in vitamin D-dependent immune defenses (particularly cathelicidin and defensin production) may be a significant factor in seasonal infection susceptibility.
- Latitude and Autoimmunity – Multiple sclerosis, type 1 diabetes, and inflammatory bowel disease all show increasing prevalence with increasing latitude, mirroring the gradient of declining UVB exposure and vitamin D synthesis. This pattern, observed across multiple continents and ethnic groups, is one of the strongest ecological arguments for a causal role of vitamin D in autoimmune disease prevention.
Clinical Dosing for Immune Health
- Minimum Effective Level – Most immune benefits of vitamin D appear to require 25(OH)D levels of at least 30 ng/mL (75 nmol/L). The Martineau meta-analysis suggests that correcting levels below 10 ng/mL provides the most dramatic immune benefit.
- Target Range – Many immunologists and functional medicine practitioners recommend targeting 25(OH)D levels of 40 to 60 ng/mL (100 to 150 nmol/L) for optimal immune function, based on the levels at which cathelicidin induction and immune cell function are maximized in laboratory studies.
- Daily Dosing – To achieve and maintain levels of 40 to 60 ng/mL, most adults require 2,000 to 5,000 IU of vitamin D3 daily, depending on baseline levels, body weight, skin pigmentation, sun exposure, and genetic factors affecting vitamin D metabolism.
- Loading Dose for Deficiency – For individuals with documented deficiency (below 20 ng/mL), a loading regimen of 5,000 to 10,000 IU daily for 8 to 12 weeks can rapidly restore levels, followed by a maintenance dose. Weekly doses of 50,000 IU for 8 weeks are an alternative approach commonly used in clinical practice.
- Cofactors – Optimal vitamin D metabolism requires adequate magnesium (a cofactor for both 25-hydroxylase and 1-alpha-hydroxylase), vitamin K2 (which works synergistically with vitamin D to direct calcium to bones rather than soft tissues), and adequate vitamin A to prevent VDR competition. These cofactors should be considered in comprehensive supplementation strategies.
- Monitoring – Regular 25(OH)D testing (every 3 to 6 months during dose adjustment, annually once stable) is recommended to guide supplementation and avoid both deficiency and excess. Testing is particularly important in obese individuals, those with malabsorption, and those taking medications that affect vitamin D metabolism (anticonvulsants, glucocorticoids, antiretrovirals).
This content is provided for informational purposes only and does not constitute medical advice. Consult a qualified healthcare provider before beginning any supplementation regimen, especially in the setting of granulomatous disease, hyperparathyroidism, or chronic kidney disease.
Research Papers and References
The following are landmark and frequently cited research papers underpinning the claims on this page. Links resolve to the publisher DOI or PubMed record.
Foundational Reviews
- Holick MF. Vitamin D deficiency. New England Journal of Medicine. 2007;357(3):266-281.
- Bikle DD. Vitamin D metabolism, mechanism of action, and clinical applications. Chemistry & Biology. 2014;21(3):319-329.
Respiratory Infection
- Martineau AR, Jolliffe DA, Hooper RL, et al. Vitamin D supplementation to prevent acute respiratory tract infections: systematic review and meta-analysis of individual participant data. BMJ. 2017;356:i6583.
- Liu PT et al. Toll-like receptor triggering of a vitamin D-mediated human antimicrobial response (PubMed search).
Autoimmune Disease
- Hahn J et al. VITAL ancillary study: Vitamin D and marine omega-3 fatty acid supplementation and incident autoimmune disease. BMJ. 2022 (PubMed search).
- Munger KL et al. Serum 25-hydroxyvitamin D levels and risk of multiple sclerosis (PubMed search).
- Hyppönen E et al. Intake of vitamin D and risk of type 1 diabetes: a birth-cohort study (PubMed search).
Overall Mortality and Broader Outcomes
- Chowdhury R, Kunutsor S, Vitezova A, et al. Vitamin D and risk of cause specific death: systematic review and meta-analysis. BMJ. 2014;348:g1903.
- Manson JE, Cook NR, Lee IM, et al. Vitamin D supplements and prevention of cancer and cardiovascular disease (VITAL). New England Journal of Medicine. 2019;380(1):33-44.
External Authoritative Resources
- NIH Office of Dietary Supplements — Vitamin D Fact Sheet for Health Professionals
- Linus Pauling Institute — Vitamin D
- Harvard T.H. Chan School of Public Health — The Nutrition Source: Vitamin D
Connections
- Vitamin D3
- Magnesium
- Vitamin A
- Vitamin D3 and Cancer Prevention
- Vitamin D3 and Bone Health
- Vitamin D Test
- Zinc
- Immune Boosting
- Selenium
- Vitamin C
- Vitamin E
- Vitamin K
- Cancer
- Tuberculosis
- Multiple Sclerosis
- Psoriasis