Hemochromatosis
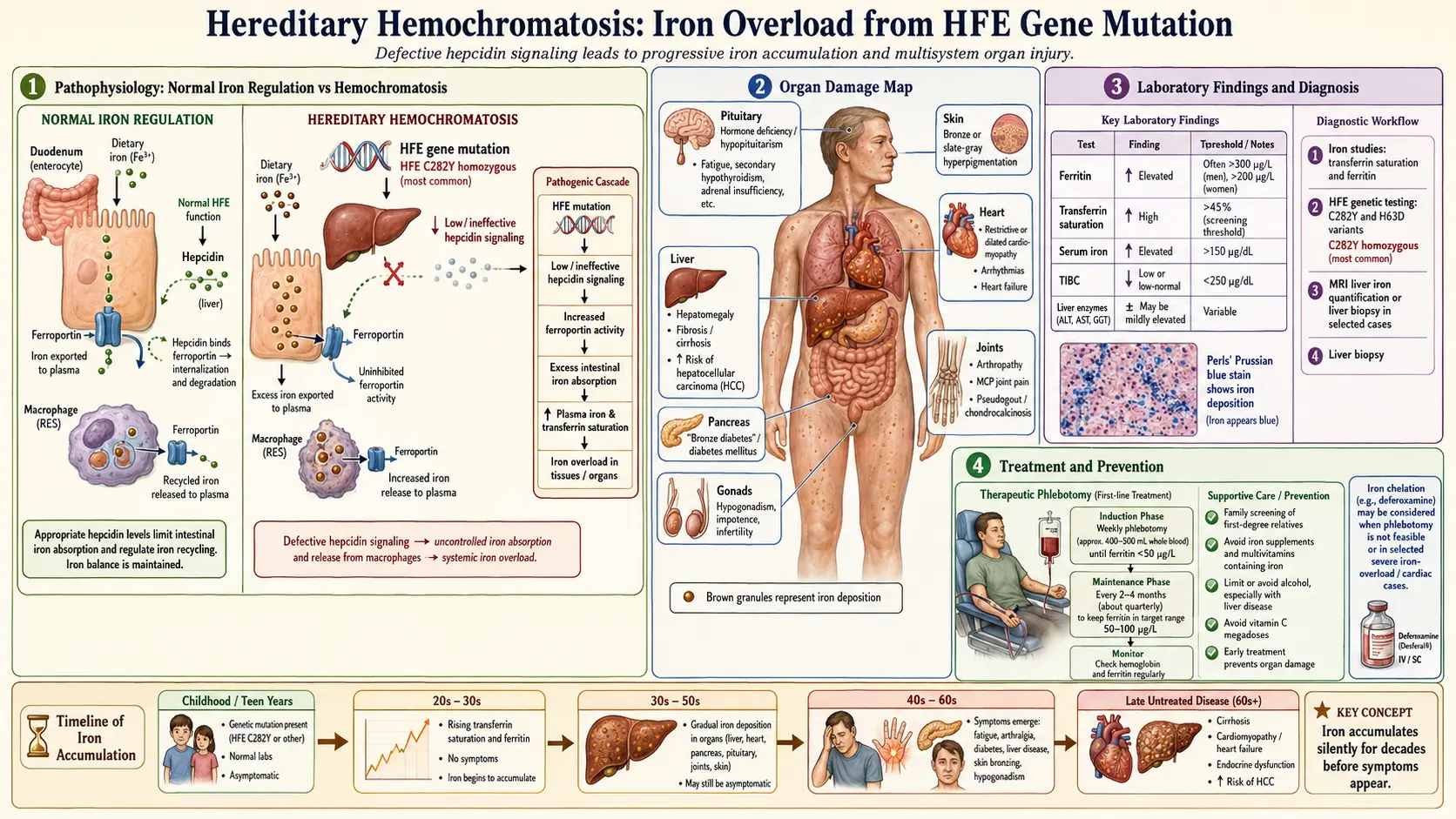
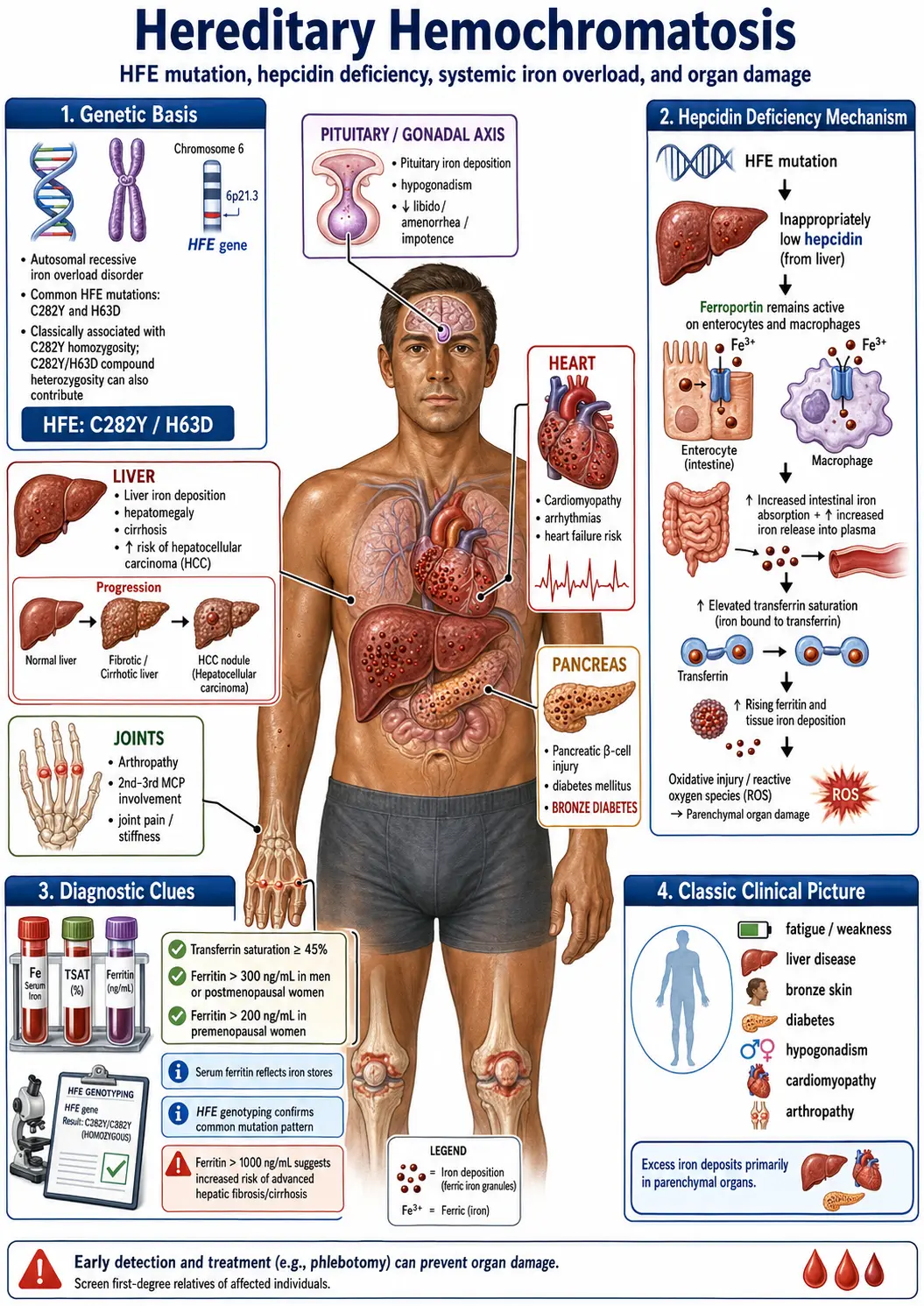
Table of Contents
- What Is Hemochromatosis?
- Hereditary vs Secondary Hemochromatosis
- How Iron Accumulates and Damages Organs
- Signs and Symptoms
- Diagnosis
- Conventional Treatment
- Natural Support Strategies
- Dietary Management
- Calcium, Tannins, and Iron Absorption Blockers
- Blood Donation as Maintenance
- Monitoring Schedule
- Important Cautions
- Research Papers
- Connections
- Featured Videos
What Is Hemochromatosis?
Hemochromatosis is an iron overload disorder in which the body absorbs and stores too much iron from the diet. Unlike most nutrients where the body can regulate excretion, humans have no efficient mechanism for eliminating excess iron. Over time, this surplus iron deposits in organs and tissues, leading to oxidative damage and progressive organ dysfunction.
From a naturopathic perspective, hemochromatosis represents a fundamental disruption of mineral homeostasis. The condition is far more common than many realize, affecting approximately 1 in 200 people of Northern European descent. Early detection and management are essential, as organ damage can be prevented entirely if iron levels are controlled before complications develop.
Iron in excess acts as a potent pro-oxidant, generating free radicals through the Fenton reaction. This oxidative stress is the central mechanism by which hemochromatosis damages tissues throughout the body.
Hereditary vs Secondary Hemochromatosis
Hereditary Hemochromatosis (HH)
Hereditary hemochromatosis is the most common genetic disorder in people of Northern European descent. It is an autosomal recessive condition primarily caused by mutations in the HFE gene located on chromosome 6.
- C282Y mutation — The most clinically significant mutation. Individuals who are homozygous (two copies of C282Y) have the highest risk of developing clinical iron overload. Approximately 85-90% of clinically diagnosed hemochromatosis cases carry this genotype.
- H63D mutation — A less severe mutation that typically causes milder iron loading. Compound heterozygotes (one C282Y and one H63D) have a moderate risk, while H63D homozygotes rarely develop significant iron overload.
- S65C mutation — A rarer variant with minimal clinical impact unless present alongside C282Y.
It is important to note that not everyone with these genetic mutations will develop clinical disease. Penetrance is variable, meaning lifestyle, diet, and other genetic factors influence whether iron overload actually manifests.
Secondary Hemochromatosis
Secondary iron overload occurs from external causes rather than genetic predisposition:
- Chronic blood transfusions — Common in patients with thalassemia, sickle cell disease, or myelodysplastic syndrome
- Excessive dietary iron or supplementation — Prolonged high-dose iron supplementation without medical indication
- Chronic liver disease — Hepatitis C, alcoholic liver disease, and non-alcoholic fatty liver disease can impair iron regulation
- Ineffective erythropoiesis — Conditions where the bone marrow produces red blood cells inefficiently, signaling the body to absorb more iron
- African iron overload — Related to dietary practices and possible non-HFE genetic factors
How Iron Accumulates and Damages Organs
In hemochromatosis, the regulatory hormone hepcidin is produced at abnormally low levels. Hepcidin normally acts as the master regulator of iron absorption by blocking the iron transporter ferroportin on intestinal cells. When hepcidin is deficient, the intestinal lining absorbs far more iron than the body needs — sometimes 3 to 4 times the normal amount.
Excess iron is deposited in organs in the following pattern:
- Liver — The first and most commonly affected organ. Iron deposits cause hepatocyte damage, fibrosis, cirrhosis, and a significantly increased risk of hepatocellular carcinoma (liver cancer). The liver may enlarge and become tender.
- Heart — Iron deposition in cardiac muscle fibers leads to cardiomyopathy, arrhythmias, and congestive heart failure. Cardiac iron overload is the leading cause of death in untreated hemochromatosis.
- Pancreas — Iron damages insulin-producing beta cells, leading to "bronze diabetes," a form of secondary diabetes mellitus that may be partially or fully irreversible.
- Joints — Iron deposits in joint cartilage and synovial membranes cause a distinctive arthropathy, particularly affecting the second and third metacarpophalangeal joints (knuckles of the index and middle fingers).
- Skin — Iron and melanin deposits produce a characteristic bronze or slate-gray skin discoloration.
- Endocrine glands — Iron accumulation in the pituitary and gonads causes hypogonadism, loss of libido, impotence in men, and amenorrhea in women.
- Thyroid — Hypothyroidism may develop from iron infiltration of thyroid tissue.
Signs and Symptoms
Hemochromatosis is often called the "silent killer" because symptoms develop gradually over decades. Men typically present symptoms between ages 40-60, while women often present later due to the protective effect of menstrual blood loss.
Early Symptoms
- Chronic fatigue and unexplained weakness
- Joint pain and stiffness, particularly in the hands
- Abdominal pain, especially in the upper right quadrant
- Brain fog and difficulty concentrating
- Loss of libido and sexual dysfunction
Advanced Symptoms
- Bronze or gray skin discoloration
- Liver enlargement and signs of liver disease (jaundice, ascites)
- Diabetes mellitus — elevated blood sugar, increased thirst and urination
- Heart failure — shortness of breath, edema, irregular heartbeat
- Impotence in men, amenorrhea in women
- Hypothyroidism
- Osteoporosis
- Cirrhosis and liver cancer
Diagnosis
Diagnosis of hemochromatosis involves a combination of blood tests, genetic testing, and imaging studies:
Blood Tests
- Serum ferritin — Measures total body iron stores. Elevated ferritin (above 200 ng/mL in women, above 300 ng/mL in men) suggests iron overload, though ferritin can also rise with inflammation, infection, or liver disease.
- Transferrin saturation — The most sensitive early marker. A fasting transferrin saturation above 45% is a strong indicator of hemochromatosis and warrants further investigation.
- Serum iron and total iron-binding capacity (TIBC) — Used to calculate transferrin saturation.
- Liver function tests — ALT and AST may be elevated if liver damage is present.
Genetic Testing
- HFE gene analysis — Tests for C282Y, H63D, and S65C mutations. Recommended for all first-degree relatives of diagnosed individuals.
- A positive genetic test confirms hereditary hemochromatosis but does not predict severity of iron loading.
Imaging and Biopsy
- MRI (T2*) — Non-invasive measurement of liver and cardiac iron concentration. Increasingly preferred over biopsy.
- Liver biopsy — May be performed to assess the degree of fibrosis or cirrhosis, particularly when ferritin exceeds 1,000 ng/mL.
Conventional Treatment
Therapeutic Phlebotomy
Phlebotomy (blood removal) is the primary and most effective treatment for hereditary hemochromatosis. It works by depleting iron stores, as the body uses stored iron to produce new red blood cells.
- Induction phase — One unit of blood (approximately 500 mL, containing about 250 mg of iron) is removed weekly or biweekly until ferritin levels drop to 50-100 ng/mL.
- Maintenance phase — Phlebotomy every 2-4 months to keep ferritin between 50-100 ng/mL and transferrin saturation below 50%.
- Patients may require removal of 50-100 units of blood during the induction phase, which can take 1-2 years.
Iron Chelation Therapy
Chelation is reserved for patients who cannot tolerate phlebotomy (such as those with anemia or severe cardiac disease):
- Deferoxamine (Desferal) — Administered by subcutaneous infusion
- Deferasirox (Exjade/Jadenu) — Oral chelation agent
- Deferiprone (Ferriprox) — Oral agent particularly effective for cardiac iron removal
Natural Support Strategies
From a naturopathic perspective, several natural compounds may support iron management and protect organs from oxidative damage caused by excess iron. These are intended as adjuncts to phlebotomy, not replacements.
IP6 (Inositol Hexaphosphate)
IP6, also known as phytic acid, is a naturally occurring compound found in grains, legumes, and seeds. It is one of the most potent natural iron chelators known. IP6 binds iron in the digestive tract, reducing absorption, and may also chelate iron that has already been absorbed. Research suggests IP6 has additional antioxidant and anti-cancer properties, making it particularly relevant for hemochromatosis patients at elevated cancer risk.
Green Tea
Epigallocatechin gallate (EGCG), the primary polyphenol in green tea, is a powerful inhibitor of iron absorption. Drinking green tea with meals can reduce non-heme iron absorption by up to 60-70%. Green tea also provides antioxidant protection against iron-induced oxidative stress. Two to three cups daily with meals is commonly recommended.
Curcumin (Turmeric)
Curcumin acts as both an iron chelator and a potent anti-inflammatory and antioxidant. Studies demonstrate that curcumin can lower serum ferritin levels and reduce liver iron content. It also protects against iron-induced liver fibrosis. Bioavailability is enhanced when combined with black pepper (piperine) or taken in liposomal form.
Milk Thistle (Silybum marianum)
Silymarin, the active complex in milk thistle, is one of the most well-studied hepatoprotective botanicals. For hemochromatosis patients, milk thistle provides liver protection through multiple mechanisms: it acts as an antioxidant, stimulates hepatocyte regeneration, reduces inflammation, and may help prevent iron-induced liver fibrosis and cirrhosis.
Quercetin
Quercetin is a flavonoid with iron-chelating properties that also reduces oxidative stress. Found abundantly in onions, apples, and berries, supplemental quercetin may help reduce intestinal iron absorption and protect tissues from iron-mediated damage.
Dietary Management
Diet plays a critical role in managing hemochromatosis. While dietary changes alone cannot treat the condition, they can significantly slow iron accumulation and reduce the frequency of phlebotomy.
Foods and Substances to Avoid
- Vitamin C with meals — Vitamin C dramatically increases non-heme iron absorption (by 2-6 times). Avoid citrus fruits, juices, and vitamin C supplements at mealtimes. Vitamin C can be consumed between meals in moderate amounts.
- Iron-fortified foods — Avoid fortified cereals, breads, and processed foods that contain added iron. Read labels carefully.
- Red meat and organ meats — Contain heme iron, which is absorbed at a much higher rate (15-35%) than plant-based non-heme iron (2-20%). Limit consumption significantly or avoid entirely during the induction phase.
- Alcohol — Alcohol increases iron absorption, damages the liver, and dramatically increases the risk of cirrhosis in hemochromatosis patients. Complete abstinence is strongly recommended, especially if liver damage is present.
- Raw shellfish — Hemochromatosis patients are uniquely vulnerable to Vibrio vulnificus, a bacterium found in raw oysters and other shellfish. This organism thrives in iron-rich blood and can cause rapidly fatal septicemia. All shellfish must be thoroughly cooked.
- Iron supplements and multivitamins containing iron — Absolutely contraindicated.
Foods to Emphasize
- Whole grains, legumes, nuts, and seeds — Rich in phytates that naturally inhibit iron absorption
- Tea and coffee with meals — Tannins and polyphenols reduce iron absorption
- Dairy products with meals — Calcium inhibits both heme and non-heme iron absorption
- Eggs — Contain phosvitin, which binds iron and reduces its absorption
- Fruits and vegetables — Consume vitamin C-rich varieties between meals rather than with meals
Calcium, Tannins, and Iron Absorption Blockers
Several naturally occurring substances can reduce iron absorption when consumed with meals:
- Calcium — One of the few substances that inhibits both heme and non-heme iron absorption. Taking 300-600 mg of calcium with iron-rich meals can reduce absorption by 40-60%. Dairy products are a practical source.
- Tannins — Found in tea (especially black tea), coffee, and red wine (though alcohol is contraindicated). Black tea consumed with meals can reduce non-heme iron absorption by 60-80%.
- Phytates (phytic acid) — Present in whole grains, legumes, nuts, and seeds. Phytates form insoluble complexes with iron in the gut, preventing absorption.
- Polyphenols — Found in green tea, cocoa, and many fruits and vegetables. These compounds chelate iron in the digestive tract.
- Oxalates — Found in spinach, rhubarb, and beets. While spinach contains iron, its high oxalate content significantly reduces iron bioavailability.
- Soy protein — Contains compounds that reduce iron absorption independent of its phytate content.
Blood Donation as Maintenance
Regular blood donation serves as an excellent maintenance strategy for hemochromatosis patients who have achieved target ferritin levels through therapeutic phlebotomy. In many countries, hemochromatosis patients are eligible to donate blood, which benefits both the patient and the blood supply.
- Each whole blood donation removes approximately 250 mg of iron.
- Donation every 8-12 weeks can help maintain healthy iron levels.
- In the United States, the FDA permits blood from hemochromatosis patients to be used for transfusion if all standard eligibility criteria are met.
- Blood donation provides the psychological benefit of turning a medical necessity into an act of service.
- Some blood banks may require a physician's note or have specific protocols for hemochromatosis donors.
Monitoring Schedule
Regular monitoring is essential for lifelong management of hemochromatosis:
During Induction Phase
- Serum ferritin and transferrin saturation — Every 1-2 months
- Complete blood count (CBC) — Before each phlebotomy to ensure hemoglobin is adequate
- Liver function tests — Every 3-6 months
During Maintenance Phase
- Serum ferritin — Every 3-6 months (target: 50-100 ng/mL)
- Transferrin saturation — Every 3-6 months (target: below 50%)
- Liver function tests — Annually
- Fasting glucose or HbA1c — Annually to screen for diabetes
- Cardiac evaluation — Echocardiogram and ECG as indicated
- Liver imaging — Ultrasound or MRI annually if cirrhosis is present (hepatocellular carcinoma screening)
- Bone density scan (DEXA) — Every 2-3 years if osteoporosis risk factors are present
- Thyroid function tests — Annually
- Testosterone levels (men) — Annually if hypogonadism is suspected
Family Screening
All first-degree relatives (parents, siblings, children) of a diagnosed individual should undergo genetic testing and iron studies. Early identification allows for preventive management before organ damage occurs.
Important Cautions
- Do not self-diagnose or self-treat iron overload. Elevated ferritin can result from many conditions including inflammation, infection, metabolic syndrome, and liver disease. Proper diagnosis requires a physician.
- Never take iron supplements if you have hemochromatosis or carry HFE mutations. This includes multivitamins containing iron.
- Avoid high-dose vitamin C supplements (above 200 mg), as vitamin C mobilizes stored iron and can increase free iron in the blood, potentially worsening cardiac iron toxicity.
- Natural iron chelators (IP6, curcumin, quercetin) should complement, not replace, therapeutic phlebotomy. Their chelation capacity is modest compared to the iron removal achieved by phlebotomy.
- Alcohol avoidance is critical. Even moderate alcohol consumption synergistically increases liver damage risk in hemochromatosis patients.
- Never eat raw shellfish. Vibrio vulnificus infection in hemochromatosis patients carries a mortality rate exceeding 50%.
- Liver cancer screening is essential for patients who have developed cirrhosis, as hemochromatosis-related cirrhosis carries a 200-fold increased risk of hepatocellular carcinoma.
- Pregnancy considerations — Women with hemochromatosis should discuss iron management with their healthcare provider during pregnancy, as iron demands change significantly.
- Consult a qualified naturopathic doctor or integrative physician before beginning any natural supplement protocol, particularly if you are undergoing phlebotomy or taking medications.
Research Papers
The following PubMed topic searches return current peer-reviewed literature relevant to this condition. Each link opens a live PubMed query.
- Hereditary hemochromatosis HFE
- Hemochromatosis iron overload
- Therapeutic phlebotomy hemochromatosis
- Hemochromatosis ferritin transferrin saturation
- Hemochromatosis liver cirrhosis
- Hemochromatosis cardiomyopathy
- Hemochromatosis diabetes
- Hemochromatosis genetic testing
- Hemochromatosis screening
- Hemochromatosis guidelines
- Secondary iron overload transfusion
- Hepcidin hemochromatosis
Connections
- Iron
- Relationship Between Hemoglobin and Ceruloplasmin — why ceruloplasmin (the copper ferroxidase) is required to load iron into hemoglobin, and why "iron-deficiency anemia" is often functional copper deficiency.
- Thalassemia
- Anemia
- Cirrhosis
- Liver Disease
- Cardiomyopathy
- Heart Failure
- Diabetes
- Vitamin C
- Berberine and Iron Overload
- Milk Thistle
- Quercetin
- Complete Blood Count
- Hepatitis C
- Polycythemia Vera
- Sickle Cell Disease
- Non Alcoholic Fatty Liver Disease
- Beef