Berberine and Iron Overload: Lowering Ferritin in Men with High Iron
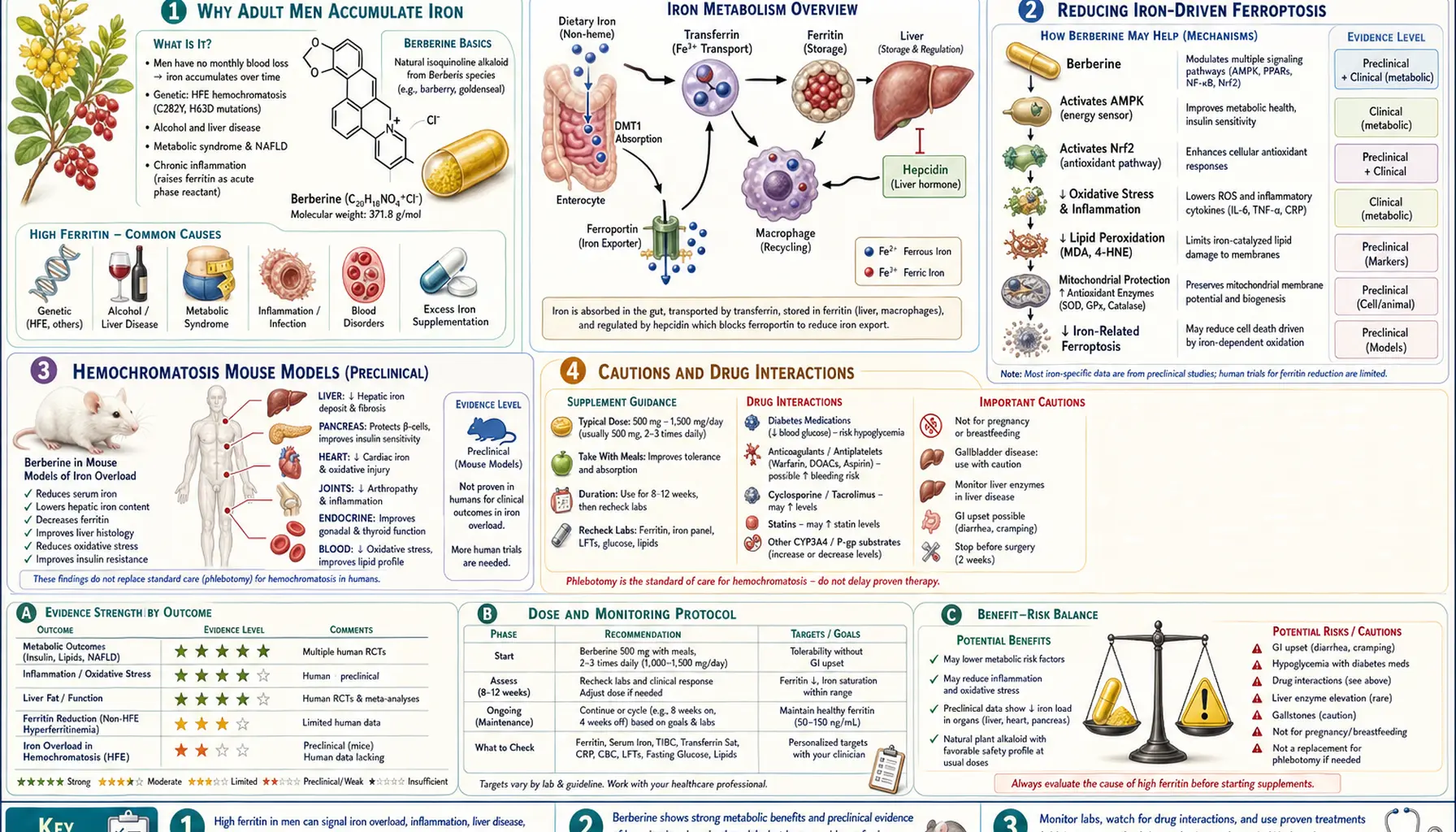
Table of Contents
- Why Adult Men Accumulate Iron
- Berberine Activates Hepcidin, the Master Iron Switch
- Direct Iron Chelation by Berberine
- Reducing Iron-Driven Ferroptosis
- Liver Iron, NAFLD, and Insulin Resistance
- Human Evidence: Ferritin Reduction in Metabolic Syndrome
- Hemochromatosis Mouse Models
- Practical Protocol for Men with High Ferritin
- Lab Markers to Track
- Cautions and Drug Interactions
- Research Papers and References
- Connections
- Featured Videos
Why Adult Men Accumulate Iron
Adult men have a unique iron-loading problem that women of reproductive age do not share: there is no monthly physiological route for offloading excess iron. The female menstrual cycle removes roughly 0.5-1.0 mg of iron per day on average across the cycle, a quiet but consistent excretion pathway. Adult men, by contrast, lose only the small amount of iron sloughed off in skin cells and intestinal turnover — about 1 mg per day — while a typical Western diet supplies 10-20 mg of iron daily.
The result is that serum ferritin in healthy American men rises steadily from age 20 onward, often crossing 200 ng/mL by the mid-30s and reaching 300-500 ng/mL in middle age even without hereditary hemochromatosis. Postmenopausal women catch up to men within a decade once monthly losses stop. High ferritin in the absence of acute inflammation is increasingly recognized as a marker of metabolic risk: it associates with insulin resistance, fatty liver disease, atherosclerosis, and accelerated biological aging.
Therapeutic phlebotomy (regular blood donation) is the most direct way to reduce body iron, but it is not always practical, accessible, or accepted by patients. Pharmacological iron chelators such as deferasirox and deferoxamine are reserved for transfusion-dependent disease because of cost and side-effect profile. This creates a gap in the middle: patients with mildly to moderately elevated ferritin, no anemia, and no obvious chelation indication, but a real cardiovascular and metabolic risk from excess body iron. Berberine has emerged as a credible dietary-supplement option in this space.
Berberine Activates Hepcidin, the Master Iron Switch
Hepcidin is the 25-amino-acid peptide hormone, produced primarily by the liver, that controls how much iron enters the bloodstream from the diet and from macrophage recycling. High hepcidin binds and degrades ferroportin, the cellular iron-export channel, locking iron inside enterocytes (intestinal cells) and macrophages. The net effect of elevated hepcidin is reduced dietary iron absorption and reduced release of stored iron into circulation.
A series of preclinical studies have demonstrated that berberine increases hepatic hepcidin expression. The mechanism appears to involve activation of the STAT3 signaling pathway downstream of IL-6 and BMP6, both physiological inducers of hepcidin transcription. By raising baseline hepcidin tone, berberine reduces the rate at which dietary iron crosses from the gut into the bloodstream — effectively dampening the daily iron intake at its first step.
This is a mechanistically attractive intervention for men with high ferritin and an iron-rich diet, because it works upstream: less iron is absorbed in the first place, which is more sustainable than trying to chelate iron that has already entered the body. Hepcidin upregulation is also why berberine should be used cautiously in patients with iron-deficiency anemia — the same mechanism that helps iron-overloaded men can interfere with iron repletion in deficient women.
Direct Iron Chelation by Berberine
Beyond its hepcidin effects, berberine itself acts as a mild iron-binding molecule. The isoquinoline ring system contains nitrogen donors that can coordinate ferrous (Fe2+) and ferric (Fe3+) ions, forming chelate complexes that have been characterized by spectroscopic and crystallographic studies. The binding affinity is lower than that of pharmaceutical chelators like deferoxamine, but it is sufficient to reduce the pool of catalytically active free iron available for participating in Fenton-reaction oxidative chemistry.
Fenton chemistry — the reaction of Fe2+ with hydrogen peroxide to generate the hydroxyl radical — is one of the principal sources of cellular oxidative damage. By tying up labile iron in less-reactive complexes, berberine reduces the rate at which free radicals are generated in tissues with high iron exposure (liver, heart, brain). This is part of why berberine displays antioxidant activity in cell-culture and animal models even when it is not directly scavenging reactive species.
The chelation activity is modest enough that berberine does not cause anemia in iron-replete individuals at typical supplement doses, but strong enough to be measurable in oxidative-stress markers (malondialdehyde, 4-hydroxynonenal, protein carbonyls) that decrease after berberine administration in animal iron-loading models.
Reducing Iron-Driven Ferroptosis
Ferroptosis is a form of regulated cell death distinct from apoptosis or necrosis, characterized by iron-dependent lipid peroxidation. It is now recognized as a major contributor to ischemia-reperfusion injury in the heart and brain, neurodegeneration (Parkinson's and Alzheimer's disease), and the toxicity of iron overload in the liver. The biochemical signature is depletion of the glutathione peroxidase 4 (GPX4) enzyme and accumulation of lipid hydroperoxides driven by free iron.
Berberine has been shown in multiple preclinical models to attenuate ferroptosis. Mechanisms documented in the literature include upregulation of GPX4 expression, increased glutathione synthesis through NRF2 pathway activation, suppression of ACSL4 (a lipid-remodeling enzyme that primes membranes for peroxidation), and reduction of the labile iron pool. The net effect is preservation of cells that would otherwise die from iron-mediated oxidative injury.
Tissues that have benefited from berberine's anti-ferroptotic action in animal models include cardiac muscle after ischemia-reperfusion, hepatocytes under iron loading, dopaminergic neurons in Parkinson's models, and renal tubular cells in acute kidney injury. The work is largely preclinical at this point, but the cellular biology is well-characterized and the pathway-level mechanisms map closely to the human conditions in which iron-driven oxidative damage is implicated.
Liver Iron, NAFLD, and Insulin Resistance
Non-alcoholic fatty liver disease (NAFLD) and its more advanced form, non-alcoholic steatohepatitis (NASH), frequently coincide with hepatic iron accumulation. The clinical entity called "dysmetabolic iron overload syndrome" describes patients with metabolic syndrome features (central obesity, insulin resistance, hypertension, dyslipidemia) and elevated ferritin without a hereditary hemochromatosis genotype. The combination is associated with faster progression of liver fibrosis and higher cardiovascular event rates.
Berberine's well-established benefits in NAFLD — reduction of hepatic steatosis on imaging, normalization of transaminases (ALT and AST), and improvement of insulin sensitivity through AMPK activation — overlap mechanistically with its iron-modulating effects. In dysmetabolic iron overload, both pathways pull in the same therapeutic direction: reduced fat deposition, reduced iron-driven oxidative damage to hepatocytes, and reduced systemic inflammation that further raises hepcidin and ferritin.
This makes berberine a particularly logical choice for the common patient profile of a middle-aged man with elevated fasting glucose, elevated ALT, elevated ferritin, and ultrasound or MRI evidence of fatty liver. The same daily dose addresses all three nodes of the syndrome simultaneously, which is a substantial practical advantage over combining separate interventions for each.
Human Evidence: Ferritin Reduction in Metabolic Syndrome
While dedicated human trials of berberine as an iron-lowering agent are limited, several clinical studies of berberine in metabolic syndrome and NAFLD have reported ferritin reduction as a secondary outcome. The pattern across these reports is consistent: 12-24 weeks of berberine at 500 mg two to three times daily produced statistically significant reductions in serum ferritin alongside the expected improvements in fasting glucose, HbA1c, triglycerides, and ALT.
The magnitude of ferritin reduction in these reports has typically been 15-30 percent from baseline. For a middle-aged man with a starting ferritin of 400 ng/mL, that translates to a reduction of 60-120 ng/mL over a single treatment course. The effect is smaller than therapeutic phlebotomy (which can remove 200-250 mg of iron per session) but is achieved with daily oral supplementation, no needles, and the additional metabolic benefits accruing in parallel.
Importantly, the iron-reducing effect in these trials did not produce anemia or worsen hemoglobin levels in the treated patients, who were iron-replete to overloaded at baseline. This safety pattern is consistent with the upstream-hepcidin mechanism: the body is being prevented from over-absorbing dietary iron rather than being acutely stripped of essential iron.
Hemochromatosis Mouse Models
The strongest preclinical evidence for berberine as an iron-overload intervention comes from studies in the HFE-knockout mouse, the standard animal model of hereditary hemochromatosis. These mice carry a deletion of the HFE gene that, in humans, is responsible for most cases of clinical hemochromatosis, and they progressively accumulate hepatic iron and develop fibrosis if untreated.
Published studies have shown that berberine treatment in HFE-knockout mice produces:
- Increased liver hepcidin mRNA and protein expression
- Reduced duodenal ferroportin protein, consistent with hepcidin-mediated degradation
- Lower non-heme iron content in liver and spleen on quantitative tissue measurement
- Reduced serum transferrin saturation and serum iron
- Attenuation of hepatic fibrosis markers and reduced inflammatory cytokine expression
These results are striking because the HFE-knockout mouse is, by design, partially resistant to physiological hepcidin induction — the entire premise of hemochromatosis is that the iron-sensing arm of hepcidin regulation is broken. Berberine appears to act downstream of the missing HFE signal, raising hepcidin through a pathway that is preserved in the disease state. This makes hereditary hemochromatosis carriers and patients with low-grade C282Y/H63D heterozygous variants a plausible target population for clinical investigation.
Practical Protocol for Men with High Ferritin
For an adult man with mildly to moderately elevated serum ferritin (200-500 ng/mL), absent active inflammation, and a metabolic-syndrome phenotype, a reasonable berberine-based approach is:
- Dose — 500 mg of standardized berberine HCl, taken two to three times daily with meals, total daily 1000-1500 mg
- Duration — minimum 12 weeks for measurable change in ferritin and hepatic iron markers; sustained use of 6-12 months may be appropriate in metabolic syndrome
- Diet alongside — reduce heme iron from red meat and iron-fortified cereal grains; pair iron-rich meals with tea, coffee, or calcium-rich foods to inhibit non-heme iron absorption
- Coordinate with blood donation — if eligible, donating whole blood every 8-12 weeks complements the absorption-reducing action of berberine; the two together can normalize ferritin substantially faster than either alone
- Consider dihydroberberine — the reduced form of berberine has roughly five-fold better oral bioavailability and may produce equivalent effects at lower doses with less gastrointestinal disturbance
This protocol is for adult men who are iron-replete to iron-overloaded. It is not appropriate for menstruating women with normal-to-low ferritin, where the same mechanism could deepen an iron-deficient state.
Lab Markers to Track
The recommended laboratory panel before starting and at 12-week intervals during berberine for iron management:
- Serum ferritin — the primary marker; goal is to reduce toward 100-150 ng/mL for adult men over months
- Transferrin saturation — the ratio of serum iron to total iron-binding capacity; elevated transferrin saturation (above 45 percent) is a more specific marker of iron overload than ferritin alone, which can rise with inflammation
- Complete blood count with differential — ensures hemoglobin and red cell indices stay in range; the iron-reducing effect should not produce anemia
- Liver enzymes (ALT, AST, GGT) — track concurrent improvement in dysmetabolic iron overload
- HbA1c and fasting insulin — berberine's metabolic effects, which usually accompany the iron-lowering response
- High-sensitivity C-reactive protein (hsCRP) — rule out inflammation-driven ferritin elevation, which berberine addresses through a different mechanism
A baseline genetic test for HFE C282Y and H63D variants is reasonable for any adult man with persistently elevated ferritin, especially with a family history of liver disease or unexplained joint and skin pigmentation findings.
Cautions and Drug Interactions
- Iron-deficiency anemia — the same hepcidin-raising mechanism that helps iron-overloaded men will worsen iron deficiency in menstruating women, pregnant patients, or anyone with low ferritin and low hemoglobin; check baseline ferritin and CBC before starting
- Pregnancy and breastfeeding — berberine should not be used; placental and neonatal effects are not fully characterized and the iron-reducing action is the opposite of what pregnancy requires
- CYP3A4 substrates — berberine inhibits CYP3A4 and can increase plasma levels of statins, calcium-channel blockers, immunosuppressants, and other CYP3A4-cleared drugs; review all chronic medications with a pharmacist before starting
- Hypoglycemic medications — berberine adds to the glucose-lowering action of metformin, sulfonylureas, and insulin; expect to titrate hypoglycemic medications downward as berberine takes effect, especially in patients with controlled type 2 diabetes
- Antihypertensives — berberine produces mild blood-pressure reduction that can be additive with prescribed antihypertensives
- Gastrointestinal sensitivity — nausea, cramping, and altered stools are common with berberine HCl, particularly at higher doses; dihydroberberine or divided dosing reduces this; take with food
- Liver disease — in active hepatitis or decompensated cirrhosis, do not initiate berberine without hepatology guidance; the metabolism of berberine relies on hepatic conjugation pathways
Research Papers and References
The following PubMed search links provide curated entry points into the published research on berberine, hepcidin, ferroptosis, and iron overload. Each link opens directly in PubMed at the National Library of Medicine.
- Berberine and hepcidin regulation — PubMed: berberine hepcidin
- Berberine and iron metabolism — PubMed: berberine iron metabolism
- Berberine iron chelation — PubMed: berberine iron chelation
- Berberine and ferroptosis — PubMed: berberine ferroptosis
- Berberine in NAFLD and dysmetabolic iron overload — PubMed: berberine NAFLD ferritin
- Berberine in HFE-knockout hemochromatosis models — PubMed: berberine HFE hemochromatosis
- Berberine ferritin reduction in clinical trials — PubMed: berberine ferritin clinical
- Berberine, STAT3, and hepcidin signaling — PubMed: berberine STAT3 hepcidin
- Dihydroberberine pharmacokinetics and bioavailability — PubMed: dihydroberberine bioavailability
- Iron overload, ferritin, and cardiovascular risk in men — PubMed: ferritin cardiovascular risk men
- Therapeutic phlebotomy in dysmetabolic iron overload — PubMed: phlebotomy dysmetabolic iron overload
- Hepcidin physiology and ferroportin regulation — PubMed: hepcidin ferroportin regulation
External Authoritative Resources
- NCCIH — Herbs at a Glance
- MedlinePlus — Herbs and Supplements
- PubMed — all research on berberine and iron
- NIDDK — Hemochromatosis
Connections
- Berberine Overview
- Berberine (Herb Profile)
- Hemochromatosis
- Iron Overload: Hidden Toxicity
- Copper-Iron Dysregulation
- Ceruloplasmin
- Iron Panel
- Heme vs Non-Heme Iron
- Iron Deficiency Anemia
- Metformin, Berberine, and GLP-1s
- Inflammatory Markers
- Hemoglobin A1C
- Iron
- Anemia
- Metabolic Syndrome
- Insulin Resistance
- Blood Sugar Support