Pantothenic Acid and Adrenal Function
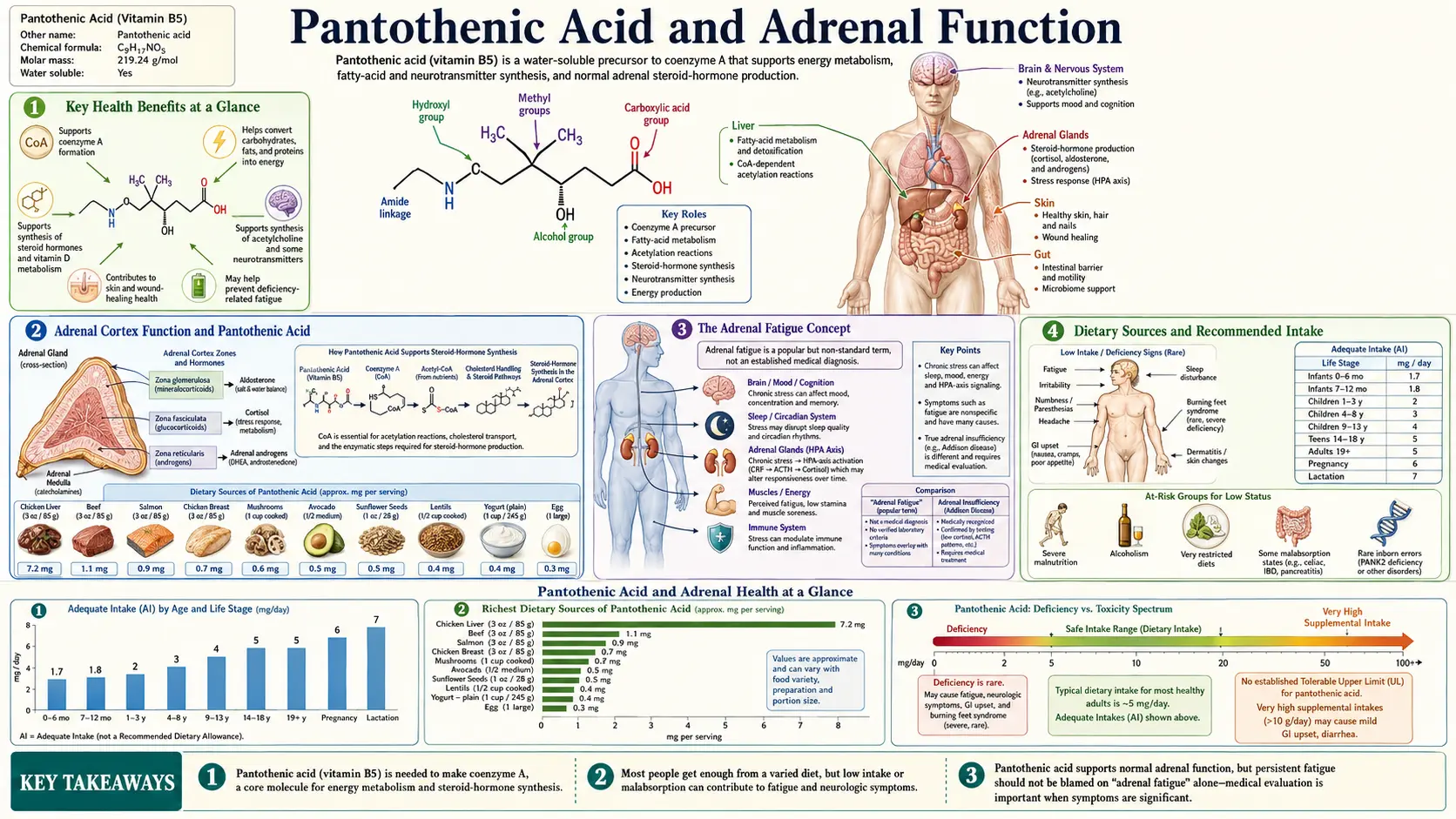
Pantothenic acid (vitamin B5) is an essential water-soluble vitamin whose name derives from the Greek word "pantos," meaning "everywhere," reflecting its widespread presence in virtually all foods. Despite its ubiquity in the diet, pantothenic acid plays a disproportionately critical role in adrenal gland function, steroid hormone synthesis, and the body's capacity to mount an appropriate stress response. The adrenal glands contain among the highest concentrations of pantothenic acid of any organ in the body, underscoring the intimate relationship between this vitamin and adrenal physiology.
Table of Contents
- Key Health Benefits at a Glance
- Coenzyme A Synthesis Pathway
- Cortisol and Steroid Hormone Production
- Adrenal Cortex Function and Pantothenic Acid
- The Stress Response and the HPA Axis
- Pantothenic Acid Depletion During Chronic Stress
- The Adrenal Fatigue Concept
- Pantethine Supplementation
- Clinical Evidence for Stress Management
- Dietary Sources and Recommended Intake
- Research Papers and References
- Connections
- Featured Videos
Key Health Benefits at a Glance
The following summarizes the evidence-backed roles of adequate pantothenic acid (vitamin B5) status, with particular emphasis on adrenal physiology and the stress response. Each bullet is expanded in the sections below and supported by research in the Research Papers section.
- Coenzyme A (CoA) synthesis – Pantothenic acid is the obligate precursor to CoA, a cofactor for over 100 metabolic reactions spanning the citric acid cycle, fatty acid metabolism, and steroid hormone synthesis.
- Adrenal steroidogenesis – CoA-derived acetyl groups drive the conversion of cholesterol to pregnenolone and the downstream synthesis of cortisol, aldosterone, and DHEA.
- Among the highest tissue concentrations – The adrenal cortex maintains some of the highest CoA levels in the body, reflecting the extraordinary throughput of steroidogenic reactions.
- HPA-axis support – CoA-dependent energy metabolism underpins CRH release, ACTH processing, and adrenal cortisol output.
- Stress-induced depletion – Chronic psychological or physiological stress increases CoA demand and accelerates pantothenic acid turnover.
- Lipid-modifying effects of pantethine – 600–1,200 mg/day of pantethine reduces total cholesterol and triglycerides, an effect attributed to enhanced hepatic CoA availability.
- Glutathione synthesis support – Adequate CoA is required for the antioxidant defense pathways that protect adrenal tissue from oxidative damage during cortisol output.
- Outstanding safety profile – No tolerable upper intake level (UL) has been established; pantothenic acid and pantethine are non-toxic even at gram-level doses.
Coenzyme A Synthesis Pathway
The primary biochemical role of pantothenic acid is as a precursor to coenzyme A (CoA), one of the most important cofactors in all of metabolism. The biosynthesis of CoA from pantothenic acid proceeds through five enzymatic steps:
- Step 1: Phosphorylation — pantothenate kinase (PanK) phosphorylates pantothenic acid to 4'-phosphopantothenate. This is the rate-limiting step in CoA synthesis and is regulated by feedback inhibition from CoA and its thioesters.
- Step 2: Cysteine conjugation — phosphopantothenoylcysteine synthetase adds a cysteine residue to form 4'-phosphopantothenoylcysteine, incorporating the sulfhydryl group that will become the business end of CoA.
- Step 3: Decarboxylation — phosphopantothenoylcysteine decarboxylase removes the carboxyl group to yield 4'-phosphopantetheine.
- Step 4: Adenylylation — phosphopantetheine adenylyltransferase adds an AMP moiety to produce dephospho-CoA.
- Step 5: Final phosphorylation — dephospho-CoA kinase adds the final phosphate group to produce coenzyme A.
CoA participates in over 100 metabolic reactions, including the citric acid cycle (via acetyl-CoA), fatty acid synthesis and oxidation, amino acid metabolism, and critically for adrenal function, the synthesis of all steroid hormones from cholesterol.
Cortisol and Steroid Hormone Production
The adrenal cortex synthesizes steroid hormones from cholesterol through a series of enzymatic reactions that are fundamentally dependent on CoA:
- Cholesterol mobilization — the initial and rate-limiting step in steroidogenesis involves the transport of cholesterol from the outer to the inner mitochondrial membrane by the steroidogenic acute regulatory protein (StAR). The cholesterol itself is delivered to the adrenal cortex via LDL receptor-mediated uptake and stored as cholesteryl esters, which are formed through CoA-dependent acyltransferase reactions.
- Pregnenolone synthesis — cholesterol is converted to pregnenolone by the cytochrome P450 side-chain cleavage enzyme (CYP11A1), the common precursor to all steroid hormones.
- Cortisol production — pregnenolone is converted to cortisol through a series of hydroxylation reactions in the zona fasciculata. Each step requires adequate cofactor availability, and the acetyl groups donated by acetyl-CoA are essential for the acetylation reactions that regulate enzyme activity and protein function throughout this cascade.
- Aldosterone synthesis — the zona glomerulosa produces aldosterone, the primary mineralocorticoid, through a parallel pathway from pregnenolone.
- DHEA and androgen production — the zona reticularis produces dehydroepiandrosterone (DHEA) and androstenedione, adrenal androgens that serve as precursors for peripheral sex hormone synthesis.
Adrenal Cortex Function and Pantothenic Acid
The adrenal glands have a uniquely high demand for pantothenic acid, reflected in several observations:
- Tissue concentration — the adrenal glands maintain CoA concentrations that are among the highest in the body, second only to the liver. This high concentration reflects the enormous throughput of acetyl-CoA required for steroid hormone synthesis.
- Animal studies — classic experiments in pantothenic acid deficiency in rats demonstrated adrenal cortical atrophy, hemorrhagic necrosis of the adrenal glands, and impaired corticosterone production. These animals showed reduced ability to mount a stress response and had shortened survival under experimental stress conditions.
- Adrenal ascorbic acid depletion — pantothenic acid-deficient animals show impaired release of ascorbic acid from the adrenal glands in response to ACTH stimulation, indicating disrupted adrenal signaling cascades.
- Human implications — while frank pantothenic acid deficiency is rare in humans due to its widespread dietary availability, subclinical insufficiency during periods of high demand may compromise optimal adrenal function.
The Stress Response and the HPA Axis
The hypothalamic-pituitary-adrenal (HPA) axis is the body's central stress response system, and pantothenic acid is integral to its function at every level:
- Hypothalamic activation — stress triggers the release of corticotropin-releasing hormone (CRH) from the paraventricular nucleus of the hypothalamus. CRH synthesis and release require adequate cellular energy metabolism, which depends on CoA-mediated pathways.
- Pituitary response — CRH stimulates the anterior pituitary to release adrenocorticotropic hormone (ACTH), which is derived from the precursor molecule proopiomelanocortin (POMC). The post-translational processing of POMC involves acetylation steps that are CoA-dependent.
- Adrenal cortisol output — ACTH stimulates the adrenal cortex to synthesize and release cortisol. As described above, this entire steroidogenic cascade requires adequate CoA availability derived from pantothenic acid.
- Negative feedback — cortisol feeds back to suppress CRH and ACTH release, completing the regulatory loop. Impaired cortisol production due to CoA insufficiency could theoretically result in loss of this negative feedback, leading to chronic HPA axis activation.
Pantothenic Acid Depletion During Chronic Stress
Chronic psychological, physical, or physiological stress places sustained demands on the adrenal glands that may deplete pantothenic acid reserves:
- Increased cortisol demand — chronic stress elevates cortisol production, increasing the throughput of CoA-dependent steroidogenic reactions and consuming pantothenic acid-derived CoA at an accelerated rate.
- Elevated energy expenditure — the heightened metabolic rate during stress increases demand for acetyl-CoA in the citric acid cycle and oxidative phosphorylation, competing with steroidogenic pathways for the available CoA pool.
- Nutrient depletion patterns — stressed individuals often adopt poor dietary habits, including increased consumption of refined foods that are low in B vitamins, creating a mismatch between increased demand and reduced intake.
- Inflammatory cytokine effects — chronic stress-induced inflammation may alter pantothenic acid metabolism and increase its catabolism, further reducing available stores.
- Clinical observations — early human studies found that subjects with pantothenic acid deficiency reported increased fatigue, irritability, and reduced tolerance to stress, symptoms that resolved with supplementation.
The Adrenal Fatigue Concept
The term "adrenal fatigue" has been popularized in integrative and functional medicine to describe a constellation of symptoms attributed to suboptimal adrenal function under chronic stress:
- Proposed mechanism — proponents suggest that prolonged stress exhausts the adrenal glands' capacity to produce adequate cortisol, leading to persistent fatigue, brain fog, sleep disturbance, salt and sugar cravings, and poor stress tolerance.
- Conventional medical perspective — major endocrine societies do not recognize "adrenal fatigue" as a medical diagnosis. The Endocrine Society has stated that there is no scientific evidence supporting the concept that chronic stress leads to "fatigued" adrenal glands with reduced cortisol output. True adrenal insufficiency (Addison's disease) is a well-defined medical condition with distinct diagnostic criteria.
- Pantothenic acid connection — practitioners who use the adrenal fatigue framework frequently recommend pantothenic acid supplementation as part of adrenal support protocols, based on the rationale that enhanced CoA availability supports optimal steroidogenesis. While the biochemistry is plausible, rigorous clinical evidence demonstrating that pantothenic acid supplementation improves adrenal function in otherwise healthy stressed individuals is limited.
- Balanced interpretation — regardless of whether "adrenal fatigue" exists as a distinct entity, the underlying premise that chronic stress increases demand for nutrients involved in cortisol synthesis, including pantothenic acid, is biochemically sound. Ensuring adequate nutritional status during periods of high stress is prudent general health advice.
Pantethine Supplementation
Pantethine is the disulfide form of pantetheine, the active metabolite of pantothenic acid and a direct precursor to CoA. It has attracted clinical interest for several reasons:
- Enhanced bioavailability — pantethine bypasses the rate-limiting pantothenate kinase step in CoA synthesis, potentially providing a more efficient route to increasing cellular CoA levels.
- Lipid-lowering effects — clinical trials have demonstrated that pantethine supplementation (600 to 1,200 mg daily) can reduce total cholesterol by 8 to 15 percent, LDL cholesterol by 10 to 20 percent, and triglycerides by 14 to 33 percent, while modestly increasing HDL cholesterol. These effects are attributed to enhanced CoA availability for hepatic fatty acid oxidation and reduced cholesterol synthesis.
- Adrenal support — by more efficiently increasing CoA availability, pantethine may provide superior support for steroidogenesis compared to pantothenic acid alone, though direct comparative studies in adrenal function are lacking.
- Safety profile — pantethine is generally well-tolerated, with gastrointestinal discomfort being the most commonly reported side effect. It does not share the flushing side effects associated with niacin supplementation.
Clinical Evidence for Stress Management
The evidence supporting pantothenic acid's role in stress management comes from several lines of investigation:
- Human deficiency studies — experimentally induced pantothenic acid deficiency in volunteers produced fatigue, headache, malaise, insomnia, and gastrointestinal disturbance, along with impaired antibody production and increased sensitivity to insulin. These symptoms closely overlap with those attributed to chronic stress.
- Supplementation studies — some small clinical trials have reported that B5 supplementation (up to 1,000 mg daily) improved subjective measures of stress tolerance and reduced self-reported fatigue in high-stress populations, though these studies were generally limited by small sample sizes and lack of blinding.
- Combination formulas — pantothenic acid is a common component of B-complex and adrenal support supplements. Studies of multi-nutrient formulas containing pantothenic acid alongside vitamin C, B6, and adaptogenic herbs have shown modest improvements in stress-related outcomes, though isolating the specific contribution of pantothenic acid is difficult.
- Animal evidence — rodent studies consistently demonstrate that pantothenic acid deficiency impairs adrenal function and stress tolerance, and that repletion restores these functions. However, animal studies using deficiency models may not directly translate to supplementation above adequate levels in humans.
- Recommended approach — while high-dose pantothenic acid supplementation for "adrenal support" lacks strong clinical trial evidence, ensuring adequate intake (at least the AI of 5 mg daily, with many practitioners recommending 100 to 500 mg daily during periods of stress) is a reasonable and low-risk nutritional strategy.
Dietary Sources and Recommended Intake
- Rich dietary sources — liver and organ meats, egg yolks, sunflower seeds, mushrooms (especially shiitake), avocados, sweet potatoes, lentils, chicken breast, and yogurt.
- Adequate Intake (AI) — the AI for adults is 5 mg per day. No tolerable upper intake level (UL) has been established because toxicity from oral pantothenic acid has not been reported, even at doses of 10 to 20 grams per day, though gastrointestinal distress may occur at very high doses.
- Processing losses — pantothenic acid is sensitive to food processing, with losses of 20 to 70 percent during canning, freezing, and refining of grains. Diets high in processed foods may provide less pantothenic acid than expected.
This content is provided for informational purposes only and does not constitute medical advice. Consult a qualified healthcare provider before beginning any supplementation regimen, particularly if you have an underlying endocrine condition, are taking medications that affect adrenal or lipid metabolism, or are pregnant or breastfeeding.
Research Papers and References
The following are landmark and frequently cited research papers underpinning the claims on this page. Links resolve to the publisher DOI, PubMed record, or PubMed topic search.
Pantothenic Acid Biochemistry and CoA Synthesis
- Leonardi R, Zhang YM, Rock CO, Jackowski S. Coenzyme A: back in action. Progress in Lipid Research. 2005;44(2-3):125-153. (PubMed topic search)
- Jackowski S, Rock CO. Regulation of coenzyme A biosynthesis. (PubMed topic search)
Adrenal Steroidogenesis and CoA
- Miller WL, Auchus RJ. The molecular biology, biochemistry, and physiology of human steroidogenesis and its disorders. Endocrine Reviews. 2011;32(1):81-151. (PubMed topic search)
- Pantothenic acid deficiency and adrenal cortical function — classical rodent studies. (PubMed topic search)
Pantethine and Lipid Metabolism
- Evans M, Rumberger JA, Azumano I, Napolitano JJ, Citrolo D, Kamiya T. Pantethine, a derivative of vitamin B5, favorably alters total, LDL and non-HDL cholesterol in low to moderate cardiovascular risk subjects eligible for statin therapy: a triple-blinded placebo and diet-controlled investigation. Vascular Health and Risk Management. 2014;10:89-100. (PubMed topic search)
- McRae MP. Treatment of hyperlipoproteinemia with pantethine: a review and analysis of efficacy and tolerability. Nutrition Research. 2005;25(4):319-333. (PubMed topic search)
Human Deficiency and Stress
- Fry PC, Fox HM, Tao HG. Metabolic response to a pantothenic acid deficient diet in humans. Journal of Nutritional Science and Vitaminology. 1976;22(4):339-346. (PubMed topic search)
- Cadegiani FA, Kater CE. Adrenal fatigue does not exist: a systematic review. BMC Endocrine Disorders. 2016;16(1):48. (PubMed topic search)
External Authoritative Resources
- NIH Office of Dietary Supplements — Pantothenic Acid Fact Sheet for Health Professionals
- Linus Pauling Institute Micronutrient Information Center — Pantothenic Acid
- Harvard T.H. Chan School of Public Health — The Nutrition Source: Vitamin B5 (Pantothenic Acid)
- PubMed — Pantothenic acid and adrenal stress research
Connections
- Vitamin B5
- Pantothenic Acid and Wound Healing
- Vitamin B6
- Vitamin C
- Vitamin B12
- Magnesium
- Zinc
- Ashwagandha
- Rhodiola Rosea
- Stress Management
- Sleep Hygiene
- Anxiety
- Depression
- Addison's Disease
- Cushing's Syndrome
- Adrenal Fatigue
- Reverse T3 and Low T3 Syndrome