Vitamin B6 and Brain Health
Vitamin B6, in its active form pyridoxal 5'-phosphate (PLP), is arguably the most critical B vitamin for central nervous system function. It serves as a cofactor for over 140 enzymatic reactions, with a particularly high concentration of PLP-dependent enzymes involved in neurotransmitter synthesis, amino acid metabolism, and myelin formation. The brain maintains PLP concentrations 25 to 50 times higher than plasma levels, reflecting the extraordinary dependence of neural tissue on this cofactor.
Table of Contents
- Key Health Benefits at a Glance
- Neurotransmitter Synthesis
- PLP as a Versatile Cofactor
- Mood Regulation and Depression
- Cognitive Function in Aging
- Myelin Synthesis
- Homocysteine and Cerebrovascular Risk
- Pregnancy and Brain Development
- Vitamin B6 and ADHD Research
- Clinical Considerations and Safety
- Research Papers and References
- Connections
- Featured Videos
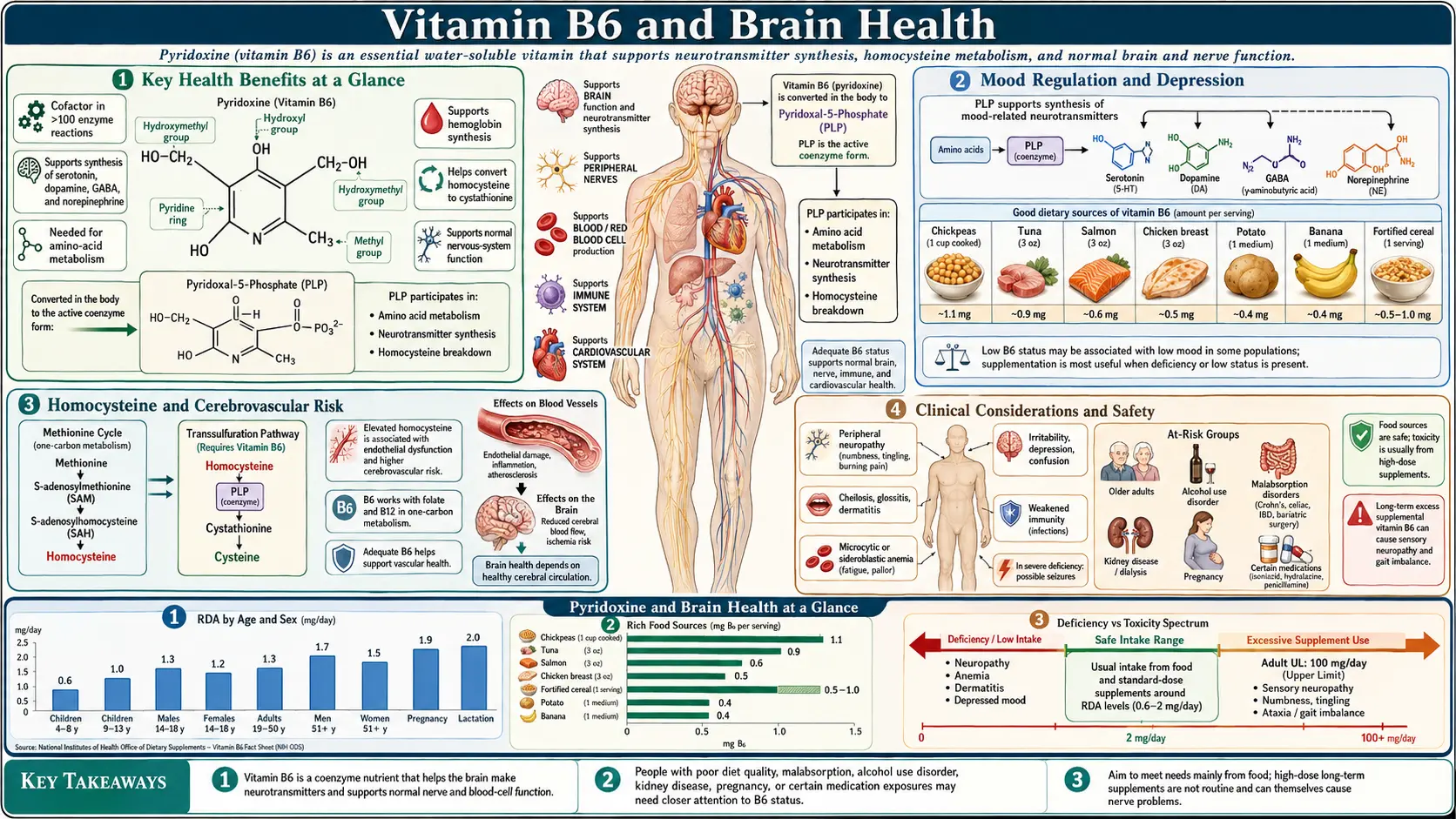
Key Health Benefits at a Glance
The following summarizes the evidence-backed neurological roles of adequate vitamin B6 (pyridoxal 5'-phosphate, PLP) status. Each is explored in more depth below, and supporting research is listed in the Research Papers section.
- Cofactor for 140+ enzymes – PLP participates in neurotransmitter synthesis, transamination, sphingolipid biosynthesis, and one-carbon metabolism.
- Serotonin and dopamine synthesis – The PLP-dependent aromatic L-amino acid decarboxylase (AADC) converts 5-HTP to serotonin and L-DOPA to dopamine.
- GABA production – Glutamic acid decarboxylase (GAD) is PLP-dependent and generates the brain's primary inhibitory neurotransmitter.
- Mood regulation – Low plasma PLP is associated with roughly double the risk of depression; B6 reduces PMS mood symptoms.
- Treats nausea of pregnancy – First-line therapy (10–25 mg TID), often combined with doxylamine.
- Prevents pyridoxine-dependent seizures – Neonatal ALDH7A1 seizure disorder responds specifically to high-dose pyridoxine.
- Lowers homocysteine – PLP is the cofactor for cystathionine beta-synthase, reducing cerebrovascular risk.
- Slows brain atrophy – The VITACOG trial showed 30% slower atrophy in MCI patients on B vitamins including B6.
- Supports fetal brain development – Maternal PLP underpins fetal neurotransmitter synthesis and one-carbon metabolism during critical windows.
- Supports myelin integrity – Serine palmitoyltransferase (sphingolipid synthesis) is PLP-dependent.
Neurotransmitter Synthesis
PLP is an essential cofactor for the synthesis of several major neurotransmitters, making vitamin B6 status a direct determinant of brain chemistry:
Serotonin (5-Hydroxytryptamine)
- Biosynthetic pathway — tryptophan is first hydroxylated by tryptophan hydroxylase to 5-hydroxytryptophan (5-HTP), which is then decarboxylated by aromatic L-amino acid decarboxylase (AADC), a PLP-dependent enzyme, to produce serotonin.
- Functional significance — serotonin regulates mood, appetite, sleep, pain perception, and social behavior. Insufficient PLP availability directly reduces serotonin synthesis capacity, potentially contributing to depressive symptoms and sleep disturbances.
- Melatonin connection — serotonin is the precursor to melatonin via N-acetylation and O-methylation in the pineal gland. Reduced serotonin availability secondary to B6 deficiency may impair melatonin production and disrupt circadian rhythm regulation.
Dopamine
- Biosynthetic pathway — tyrosine is hydroxylated by tyrosine hydroxylase to L-DOPA, which is then decarboxylated by AADC (the same PLP-dependent enzyme involved in serotonin synthesis) to produce dopamine.
- Functional significance — dopamine is central to reward, motivation, motor control, and executive function. B6 deficiency can impair dopamine synthesis, potentially affecting motivation, attention, and movement coordination.
- Norepinephrine extension — dopamine is further converted to norepinephrine by dopamine beta-hydroxylase (a copper- and ascorbate-dependent enzyme), extending the impact of B6-dependent dopamine synthesis to the adrenergic system.
GABA (Gamma-Aminobutyric Acid)
- Biosynthetic pathway — glutamate is decarboxylated by glutamic acid decarboxylase (GAD), a PLP-dependent enzyme, to produce GABA, the brain's primary inhibitory neurotransmitter.
- Functional significance — GABA counterbalances excitatory neurotransmission, preventing neuronal over-excitation. GABA deficiency results in anxiety, insomnia, seizure susceptibility, and sensory hyperactivity.
- Clinical relevance of B6 and seizures — pyridoxine-dependent epilepsy is a rare genetic condition in which mutations in the ALDH7A1 gene (encoding antiquitin) lead to accumulation of alpha-aminoadipic semialdehyde, which inactivates PLP. These patients develop intractable seizures in the neonatal period that respond specifically to high-dose pyridoxine (50 to 200 mg IV), demonstrating the absolute requirement of PLP for adequate GABA synthesis.
Norepinephrine
- Pathway dependence — because norepinephrine is synthesized from dopamine, the PLP-dependent step in dopamine synthesis is rate-relevant for norepinephrine production as well.
- Functional significance — norepinephrine mediates alertness, arousal, attention, and the stress response. It also regulates blood pressure and heart rate through peripheral sympathetic nerve activity. Impaired synthesis can manifest as fatigue, poor concentration, and reduced stress tolerance.
PLP as a Versatile Cofactor
Beyond neurotransmitter synthesis, PLP participates in numerous brain-relevant biochemical reactions:
- Transamination reactions — PLP is the cofactor for all aminotransferases, enzymes that interconvert amino acids and keto acids. In the brain, these reactions are critical for maintaining the glutamate-glutamine cycle and for providing substrates for energy metabolism.
- Sphingolipid synthesis — serine palmitoyltransferase, the rate-limiting enzyme in sphingolipid biosynthesis, requires PLP. Sphingolipids are essential components of neuronal cell membranes and myelin, and disrupted sphingolipid metabolism has been implicated in neurodegenerative diseases.
- Histamine metabolism — histidine decarboxylase, which converts histidine to histamine, is PLP-dependent. Brain histamine regulates wakefulness, appetite, and neuroendocrine function.
- One-carbon metabolism — serine hydroxymethyltransferase (SHMT), a PLP-dependent enzyme, generates one-carbon units for nucleotide synthesis and methylation reactions, linking B6 to DNA repair and epigenetic regulation in the brain.
Mood Regulation and Depression
The relationship between vitamin B6 and mood disorders has been studied extensively:
- Epidemiological associations — large population studies, including data from NHANES, have consistently found that low plasma PLP levels are associated with increased risk of depression. Individuals in the lowest quartile of PLP status have approximately twice the risk of depressive symptoms compared to those in the highest quartile.
- Mechanistic basis — the link between B6 deficiency and depression is most directly explained by impaired serotonin and dopamine synthesis. Additionally, B6 deficiency elevates homocysteine levels, which may contribute to depression through oxidative stress, neuroinflammation, and impaired methylation of neurotransmitter-related genes.
- Premenstrual syndrome (PMS) — vitamin B6 supplementation (50 to 100 mg daily) has shown modest but consistent benefit in reducing PMS-related mood symptoms in multiple systematic reviews. The mechanism may involve enhanced serotonin synthesis during the luteal phase when estrogen-induced tryptophan catabolism increases B6 demand.
- Oral contraceptive-related mood changes — estrogen-containing oral contraceptives increase tryptophan catabolism through the kynurenine pathway, increasing PLP consumption and potentially reducing serotonin synthesis. B6 supplementation has been proposed to mitigate mood side effects of hormonal contraception.
- Supplementation evidence — while correcting frank B6 deficiency clearly improves mood symptoms, the evidence for high-dose B6 supplementation as a standalone treatment for clinical depression in B6-replete individuals is less convincing. B6 is more likely to be beneficial as part of a comprehensive approach that addresses overall nutritional status.
Cognitive Function in Aging
Age-related cognitive decline has been linked to vitamin B6 status through several pathways:
- Prevalence of B6 insufficiency in the elderly — plasma PLP levels decline with age due to reduced dietary intake, impaired absorption, increased catabolism, and medication interactions. Studies estimate that 20 to 30 percent of elderly individuals have marginal or deficient B6 status.
- Cognitive performance correlations — multiple observational studies have found positive associations between plasma PLP levels and performance on tests of memory, processing speed, and executive function in older adults. The Rotterdam Study, Framingham cohort, and other large prospective studies support this relationship.
- Brain atrophy — elevated homocysteine levels (a marker of inadequate B6, B12, and folate status) are associated with accelerated brain atrophy, particularly in the medial temporal lobe and hippocampus, regions critical for memory formation.
- VITACOG trial — this randomized controlled trial demonstrated that B vitamin supplementation (including B6, B12, and folic acid) slowed the rate of brain atrophy by 30 percent in elderly subjects with mild cognitive impairment and elevated homocysteine. The benefit was most pronounced in participants with the highest baseline homocysteine levels.
- Alzheimer's disease — low B6 status and elevated homocysteine have been associated with increased Alzheimer's disease risk in prospective studies, though whether B6 supplementation can prevent or delay Alzheimer's remains unproven.
Myelin Synthesis
Myelin, the insulating sheath surrounding nerve axons, depends on adequate B6 status for its synthesis and maintenance:
- Sphingolipid production — myelin is rich in sphingolipids, particularly sphingomyelin and cerebrosides. As noted above, the rate-limiting enzyme in sphingolipid synthesis (serine palmitoyltransferase) is PLP-dependent. B6 deficiency can impair sphingolipid availability for myelin formation and turnover.
- Peripheral neuropathy — both B6 deficiency and B6 excess can cause peripheral neuropathy. Deficiency produces a sensory neuropathy with numbness, tingling, and impaired proprioception due to demyelination. Paradoxically, chronic high-dose B6 supplementation (typically above 200 mg daily for extended periods) causes a sensory neuropathy through a different mechanism involving direct neuronal toxicity.
- Developmental myelination — adequate B6 status during infancy and childhood is critical for the progressive myelination of the developing nervous system, which continues into the third decade of life.
Homocysteine and Cerebrovascular Risk
Vitamin B6 is one of three B vitamins (along with B12 and folate) required for homocysteine metabolism, with direct implications for cerebrovascular health:
- Transsulfuration pathway — PLP is the essential cofactor for cystathionine beta-synthase (CBS), which converts homocysteine to cystathionine in the first step of the transsulfuration pathway. B6 deficiency impairs this conversion, leading to homocysteine accumulation.
- Cerebrovascular damage — elevated homocysteine damages cerebral blood vessel endothelium through oxidative stress, reduces nitric oxide bioavailability, promotes smooth muscle cell proliferation, and enhances thrombogenicity, collectively increasing the risk of stroke and vascular dementia.
- Blood-brain barrier integrity — homocysteine has been shown to increase blood-brain barrier permeability in experimental models, potentially exposing neural tissue to circulating toxins and inflammatory mediators.
- Clinical implications — maintaining adequate B6 status to keep homocysteine levels within normal range (below 12 micromoles per liter) is considered an important component of cerebrovascular risk reduction, particularly in individuals with other vascular risk factors.
Pregnancy and Brain Development
Vitamin B6 requirements increase during pregnancy, and adequate status is critical for fetal brain development:
- Increased demand — pregnancy increases B6 requirements due to fetal growth, placental metabolism, and expanded maternal blood volume. The RDA increases from 1.3 mg to 1.9 mg daily during pregnancy.
- Fetal neurotransmitter development — the fetal brain requires PLP for the same neurotransmitter synthetic pathways as the adult brain. Maternal B6 deficiency can impair fetal serotonin, dopamine, and GABA synthesis during critical periods of brain development.
- Neural tube development — while folate receives the most attention for neural tube defect prevention, B6 participates in one-carbon metabolism and homocysteine clearance, both of which are relevant to neural tube closure. Elevated maternal homocysteine is an independent risk factor for neural tube defects.
- Nausea of pregnancy — pyridoxine (10 to 25 mg three times daily) is a first-line treatment for nausea and vomiting of pregnancy, often combined with doxylamine. The mechanism may involve modulation of serotonin and dopamine signaling in the chemoreceptor trigger zone and gastrointestinal tract.
- Long-term neurodevelopmental outcomes — observational studies have associated maternal B6 status during pregnancy with offspring cognitive performance in early childhood, though confounding factors make causal inference difficult.
Vitamin B6 and ADHD Research
The relationship between vitamin B6 and attention deficit hyperactivity disorder (ADHD) has been explored based on the vitamin's role in dopamine and norepinephrine synthesis:
- Rationale — ADHD is characterized by dysfunction in dopaminergic and noradrenergic circuits in the prefrontal cortex and basal ganglia. Since B6 is required for the synthesis of both dopamine and norepinephrine, suboptimal B6 status could theoretically exacerbate ADHD symptoms.
- Nutritional status studies — some studies have found lower plasma PLP levels in children with ADHD compared to controls, though results have been inconsistent across populations and study designs.
- Supplementation trials — several small studies have tested B6 supplementation in ADHD children, with mixed results. A few have reported modest improvements in hyperactivity and attention, but methodological limitations (small sample sizes, lack of adequate controls, concurrent treatments) prevent firm conclusions.
- Magnesium and B6 combination — a French randomized trial found that supplementation with magnesium and B6 together improved ADHD symptoms in children with documented magnesium deficiency. The combination may be more effective than either nutrient alone due to their synergistic roles in neuronal function.
- Current status — B6 supplementation is not an established treatment for ADHD, and professional guidelines do not recommend it as an alternative to evidence-based pharmacological and behavioral interventions. However, ensuring adequate B6 intake as part of overall nutritional optimization is reasonable in the management of any child with ADHD.
Clinical Considerations and Safety
- Recommended Dietary Allowance — the RDA for adults is 1.3 mg per day (ages 19 to 50), increasing to 1.5 mg for women and 1.7 mg for men over age 50.
- Tolerable Upper Intake Level — the UL is 100 mg per day for adults. Chronic intake above 200 mg daily carries a risk of sensory peripheral neuropathy, which is usually reversible upon discontinuation but may take months to resolve.
- Dietary sources — poultry, fish (especially tuna and salmon), potatoes, chickpeas, bananas, fortified cereals, and organ meats are rich sources of B6.
- Drug interactions — levodopa (without carbidopa), phenytoin, phenobarbital, and isoniazid can all interfere with B6 status or metabolism, requiring monitoring and potential supplementation.
- Assessment — plasma PLP is the most widely used biomarker of B6 status. Levels below 20 nmol/L indicate deficiency, while levels of 20 to 30 nmol/L suggest marginal status.
This content is provided for informational purposes only and does not constitute medical advice. Pyridoxine-responsive seizures and homocystinuria require specialist management. Do not exceed 100 mg/day of supplemental B6 without medical supervision due to the risk of sensory neuropathy with chronic high-dose use.
Research Papers and References
The following are landmark and frequently cited research papers underpinning the claims on this page. Links resolve to the publisher DOI, PubMed record, or PubMed topic search.
B Vitamins, Homocysteine, and the Brain
- Reynolds E. Vitamin B12, folic acid, and the nervous system. Lancet Neurology. 2006;5(11):949-960.
- Smith AD, Smith SM, de Jager CA, et al. Homocysteine-lowering by B vitamins slows the rate of accelerated brain atrophy in mild cognitive impairment: a randomized controlled trial (VITACOG). PLOS ONE. 2010;5(9):e12244. (PubMed topic search)
- Douaud G, Refsum H, de Jager CA, et al. Preventing Alzheimer's disease-related gray matter atrophy by B-vitamin treatment. PNAS. 2013;110(23):9523-9528. (PubMed topic search)
Vitamin B6, Depression, and PMS
- Hvas AM, Juul S, Bech P, Nexø E. Vitamin B6 level is associated with symptoms of depression. Psychotherapy and Psychosomatics. 2004;73(6):340-343. (PubMed topic search)
- Wyatt KM, Dimmock PW, Jones PW, O'Brien PM. Efficacy of vitamin B-6 in the treatment of premenstrual syndrome: systematic review. BMJ. 1999;318(7195):1375-1381. (PubMed topic search)
PLP Biochemistry
- Percudani R, Peracchi A. A genomic overview of pyridoxal-phosphate-dependent enzymes. EMBO Reports. 2003;4(9):850-854. (PubMed topic search)
- Mills PB, Struys E, Jakobs C, et al. Mutations in antiquitin in individuals with pyridoxine-dependent seizures. Nature Medicine. 2006;12(3):307-309. (PubMed topic search)
Nausea of Pregnancy
B6 Toxicity and Safety
External Authoritative Resources
- NIH Office of Dietary Supplements — Vitamin B6 Fact Sheet for Health Professionals
- Linus Pauling Institute Micronutrient Information Center — Vitamin B6
- Harvard T.H. Chan School of Public Health — The Nutrition Source: Vitamin B6
- PubMed — Vitamin B6 and brain/neurotransmitter research
Connections
- Vitamin B6
- Vitamin B6 and Homocysteine
- Vitamin B12
- Vitamin B9
- Folate and Mental Health
- Vitamin D3
- Magnesium
- Zinc
- Tryptophan
- Tyrosine
- Depression
- Anxiety
- ADHD
- Alzheimer's Disease
- Peripheral Neuropathy
- Homocysteine
- Sleep Hygiene