Folate and Mental Health
The relationship between folate (vitamin B9) and mental health has become an increasingly important area of research over the past three decades. Folate's role in one-carbon metabolism directly influences the synthesis of neurotransmitters implicated in mood disorders, and both epidemiological studies and clinical trials have established meaningful connections between folate status, depression, and cognitive decline. The approval of L-methylfolate as a medical food for depression management has brought this research into everyday clinical practice.
Table of Contents
- Key Benefits at a Glance
- One-Carbon Metabolism and SAMe Production
- Methylation and Neurotransmitter Synthesis
- Folate Deficiency and Depression
- MTHFR C677T Polymorphism
- L-Methylfolate as Adjunctive Antidepressant (Deplin)
- Homocysteine and Cognitive Decline
- Folate and Dementia Risk
- Clinical Evidence and Recommendations
- Research Papers
- Connections
- Featured Videos
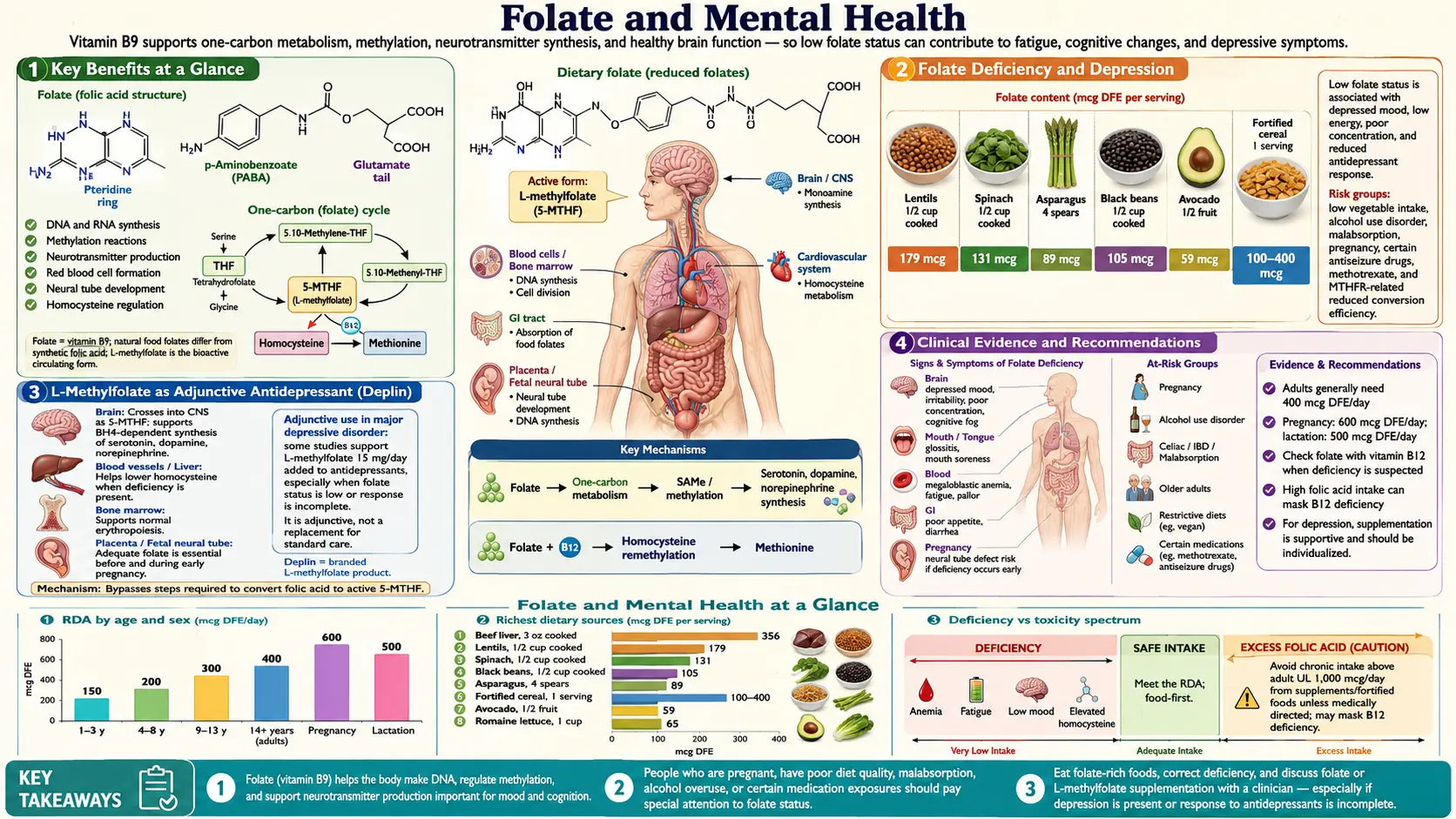
Key Benefits at a Glance
- Supports SAMe production — the universal methyl donor for over 200 CNS reactions.
- Low folate linked to higher depression risk in multiple population-based cohorts.
- L-methylfolate 15 mg/day has RCT evidence as an SSRI adjunct for treatment-resistant depression (Papakostas 2012).
- Supports monoamine synthesis (serotonin, dopamine, norepinephrine) via BH4 recycling.
- Lowers homocysteine — an independent risk factor for cognitive decline and Alzheimer's disease.
- VITACOG / OPTIMA trials showed slowed brain atrophy with B-vitamin therapy in MCI patients with elevated homocysteine.
- MTHFR 677TT genotype increases depression risk ~36%; adequate folate partially compensates.
- Epigenetic regulation of neurotransmitter-related genes via DNA methylation.
One-Carbon Metabolism and SAMe Production
Folate's influence on mental health stems primarily from its central role in one-carbon metabolism, a network of biochemical reactions that generates the universal methyl donor S-adenosylmethionine (SAMe).
- The folate cycle: Dietary folate is converted to tetrahydrofolate (THF), which accepts and donates single-carbon units in various oxidation states. The enzyme methylenetetrahydrofolate reductase (MTHFR) converts 5,10-methylenetetrahydrofolate to 5-methyltetrahydrofolate (5-MTHF), the predominant circulating form of folate and the methyl donor for the remethylation of homocysteine to methionine.
- Methionine and SAMe synthesis: The enzyme methionine synthase, using vitamin B12 as a cofactor, transfers the methyl group from 5-MTHF to homocysteine, regenerating methionine. Methionine is then activated by ATP to form S-adenosylmethionine (SAMe), the principal methyl donor for over 200 methyltransferase reactions in the body.
- SAMe and brain function: SAMe participates in critical methylation reactions in the central nervous system, including the methylation of DNA (epigenetic regulation), RNA, proteins, phospholipids (phosphatidylcholine synthesis for neuronal membranes), and small molecules including neurotransmitters. Adequate SAMe production is therefore essential for normal brain function.
- The methyl trap hypothesis: When folate or B12 is deficient, homocysteine accumulates and SAMe production declines. This creates a "methyl trap" in which methylation capacity throughout the brain is compromised, disrupting multiple neurotransmitter systems simultaneously. This mechanism provides a biochemical explanation for the psychiatric manifestations of folate deficiency.
Methylation and Neurotransmitter Synthesis
The neurotransmitters most directly affected by folate-dependent methylation are the monoamines: serotonin, dopamine, and norepinephrine, the same neurotransmitter systems targeted by modern antidepressant medications.
- Serotonin synthesis: The rate-limiting enzyme in serotonin synthesis, tryptophan hydroxylase 2 (TPH2), requires tetrahydrobiopterin (BH4) as a cofactor. BH4 regeneration depends on dihydrobiopterin reductase, and SAMe-dependent methylation is required for optimal BH4 recycling. Folate deficiency reduces SAMe availability, impairing BH4 regeneration and consequently reducing serotonin production in the raphe nuclei.
- Dopamine synthesis: Tyrosine hydroxylase, the rate-limiting enzyme in dopamine synthesis, also requires BH4 as a cofactor. Through the same BH4 recycling mechanism, folate deficiency can impair dopamine production in the ventral tegmental area and substantia nigra, potentially contributing to anhedonia, motivational deficits, and psychomotor retardation in depression.
- Norepinephrine synthesis: Dopamine beta-hydroxylase converts dopamine to norepinephrine in the locus coeruleus and sympathetic neurons. Reduced dopamine availability due to impaired BH4 recycling cascades into decreased norepinephrine production, potentially contributing to fatigue, poor concentration, and cognitive symptoms of depression.
- Neurotransmitter degradation: Catechol-O-methyltransferase (COMT) and other methyltransferases involved in neurotransmitter degradation require SAMe as a methyl donor. Paradoxically, reduced SAMe availability may slow neurotransmitter degradation, but this compensatory effect is insufficient to overcome the reduction in synthesis.
- Epigenetic regulation of neurotransmitter genes: SAMe-dependent DNA methylation regulates the expression of genes encoding neurotransmitter receptors, transporters, and synthetic enzymes. Folate deficiency can alter the methylation status of promoter regions, producing sustained changes in neurotransmitter system function that persist beyond the period of deficiency.
Folate Deficiency and Depression
The association between low folate status and depression is among the most consistently replicated findings in nutritional psychiatry.
- Epidemiological evidence: Multiple large population-based studies have demonstrated an inverse relationship between serum or red blood cell folate levels and depression risk. The Framingham Heart Study offspring cohort found that individuals in the lowest quartile of serum folate had significantly higher rates of depression. Similar findings have been reported in the NHANES database and European cohort studies.
- Prevalence in psychiatric populations: Studies of hospitalized psychiatric patients have found folate deficiency in 15 to 38% of patients with major depressive disorder, compared to 5 to 10% of the general population. Whether low folate is a cause or consequence of depression (due to poor dietary intake) remains debated, though evidence supports a bidirectional relationship.
- Treatment resistance: Low folate status has been consistently associated with poorer response to antidepressant medications, particularly selective serotonin reuptake inhibitors (SSRIs). Patients with low serum or RBC folate levels are more likely to experience treatment-resistant depression, take longer to respond to pharmacotherapy, and have higher relapse rates.
- Dose-response relationship: Some studies suggest a dose-response gradient, with the lowest folate levels associated with the greatest depression risk and severity. However, the threshold below which folate deficiency clinically affects mood is not precisely defined and may vary by individual genetic background.
MTHFR C677T Polymorphism
The MTHFR C677T genetic variant has attracted particular attention in the context of folate-related mental health research.
- Functional impact: The homozygous TT genotype results in an enzyme with approximately 30% of normal activity at 37 degrees Celsius, leading to reduced production of 5-methyltetrahydrofolate and consequently diminished capacity for homocysteine remethylation and SAMe production. Individuals with this genotype have higher homocysteine levels and lower folate levels than those with the CC or CT genotype.
- Depression risk: A 2014 meta-analysis by Gilbody and colleagues, combining data from over 26,000 subjects, found that individuals with the MTHFR 677TT genotype had a statistically significant 36% increased risk of depression compared to those with the CC genotype (odds ratio 1.36, 95% CI 1.11-1.67).
- Gene-environment interaction: The impact of the MTHFR C677T polymorphism on depression risk is modulated by folate intake. The TT genotype primarily confers increased depression risk in the setting of low folate status; adequate folate intake may partially compensate for the reduced enzyme activity, underscoring the importance of nutritional optimization in genetically susceptible individuals.
- Clinical testing: While MTHFR genotyping is commercially available and increasingly requested by patients, routine testing is not recommended by major psychiatric organizations. The clinical utility of knowing MTHFR status for treatment decisions remains debated, and results should be interpreted in the context of actual folate levels and clinical presentation.
L-Methylfolate as Adjunctive Antidepressant (Deplin)
L-methylfolate (levomefolate calcium, marketed as Deplin) represents the most direct clinical application of folate-mental health research.
- Mechanism of action: L-methylfolate (5-MTHF) is the biologically active form of folate that crosses the blood-brain barrier via the folate receptor alpha and proton-coupled folate transporter. Once in the CNS, it directly supports SAMe production and consequently neurotransmitter synthesis without requiring MTHFR-mediated conversion.
- Key clinical trial: Papakostas et al. (2012) conducted a randomized, double-blind, placebo-controlled trial of L-methylfolate 15 mg daily as adjunctive therapy to SSRIs in patients with major depressive disorder who had inadequate response to antidepressant monotherapy. L-methylfolate demonstrated a statistically significant and clinically meaningful improvement in depression scores compared to placebo, with a number needed to treat (NNT) of approximately 6.
- Sequential parallel comparison design trials: Two additional trials using sequential parallel comparison design found that L-methylfolate 15 mg (but not 7.5 mg) significantly improved treatment response when added to ongoing SSRI therapy. The 15 mg dose was associated with a 32.3% response rate compared to 14.6% for placebo in the second study phase.
- Regulatory classification: Deplin is classified as a medical food (regulated under the Orphan Drug Act) rather than a pharmaceutical drug. This classification means it does not require an FDA-approved indication statement but must be used under medical supervision for a specific condition (in this case, depression with folate deficiency or suboptimal folate metabolism).
- Cost considerations: Brand-name L-methylfolate products such as Deplin are substantially more expensive than folic acid supplements. Generic L-methylfolate supplements are available at lower cost but are classified as dietary supplements with less regulatory oversight for purity and potency.
Homocysteine and Cognitive Decline
Elevated homocysteine levels, a direct consequence of inadequate folate status, have emerged as an independent risk factor for cognitive decline and neurodegenerative disease.
- Neurotoxicity mechanisms: Homocysteine exerts neurotoxic effects through multiple mechanisms: overstimulation of NMDA glutamate receptors (excitotoxicity), generation of reactive oxygen species (oxidative stress), promotion of endothelial dysfunction and cerebrovascular disease, and direct neurotoxicity to hippocampal neurons critical for memory formation.
- Epidemiological associations: The Framingham Heart Study demonstrated that plasma homocysteine levels above 14 micromol/L nearly doubled the risk of developing Alzheimer's disease over an 8-year follow-up period. Similar associations have been reported in the Hordaland Homocysteine Study and other large cohorts.
- Brain atrophy: Elevated homocysteine has been associated with accelerated brain atrophy on serial MRI, particularly in the medial temporal lobe including the hippocampus. The OPTIMA trial demonstrated that B vitamin supplementation (including folate) slowed brain atrophy by 30% in older adults with elevated homocysteine, with the greatest protection in the brain regions most vulnerable to Alzheimer's disease.
- Intervention trials: While B vitamin supplementation effectively lowers homocysteine levels, the results of randomized controlled trials examining cognitive outcomes have been mixed. Some trials have shown benefits, particularly in individuals with elevated baseline homocysteine, while others have been negative. Heterogeneity in study populations, intervention doses, outcome measures, and treatment duration complicates interpretation.
Folate and Dementia Risk
- Alzheimer's disease: Low folate status has been associated with increased risk of Alzheimer's disease in multiple prospective cohort studies. Proposed mechanisms include impaired DNA repair in neurons, altered methylation of amyloid precursor protein (APP) and presenilin genes, increased homocysteine-mediated vascular injury, and reduced SAMe-dependent phospholipid methylation affecting neuronal membrane integrity.
- Vascular dementia: Through its effects on homocysteine and endothelial function, folate status influences cerebrovascular health. Elevated homocysteine is an established risk factor for stroke and small vessel cerebrovascular disease, which are the primary pathological substrates of vascular dementia. Folate supplementation's homocysteine-lowering effect may have particular relevance for vascular dementia prevention.
- Mild cognitive impairment: The VITACOG trial (Oxford University) found that high-dose B vitamins (including 800 mcg folic acid) significantly slowed cognitive decline and brain atrophy in older adults with mild cognitive impairment, but only in those with elevated baseline homocysteine levels. This finding suggests that B vitamin intervention is most effective when targeting a specific metabolic abnormality rather than being applied to all elderly individuals.
- Timing of intervention: There is growing recognition that the window for effective intervention may be earlier than previously thought. By the time clinical dementia is diagnosed, neurodegeneration is advanced and irreversible. Maintaining adequate folate status throughout midlife may be more protective than initiating supplementation after cognitive decline is evident.
Clinical Evidence and Recommendations
- Screening for folate deficiency in depression: Given the strong association between low folate status and treatment-resistant depression, serum folate and RBC folate levels should be measured in patients with depression, particularly those with inadequate response to standard antidepressant therapy. Homocysteine levels provide additional information about functional folate adequacy.
- Adjunctive L-methylfolate: Based on available clinical trial evidence, L-methylfolate 15 mg daily can be considered as adjunctive therapy for patients with major depressive disorder who have not achieved adequate response to SSRI or SNRI monotherapy. This recommendation is endorsed by several clinical practice guidelines, though it is not yet universally adopted.
- Folic acid supplementation: For patients with documented folate deficiency and depression, standard folic acid supplementation (1 to 5 mg daily) to correct the deficiency is a reasonable first step. Whether high-dose folic acid has antidepressant effects independent of correcting deficiency is less clear than for L-methylfolate.
- Dietary counseling: Patients with depression should be counseled to consume a diet rich in folate from natural food sources including dark leafy greens (spinach, kale, collard greens), legumes (lentils, chickpeas, black beans), asparagus, Brussels sprouts, avocados, and fortified grains. The Mediterranean diet, which is rich in these foods, has been independently associated with reduced depression risk.
- Interaction with antidepressants: L-methylfolate and folic acid are generally well tolerated when combined with antidepressant medications. No significant pharmacokinetic interactions have been reported with SSRIs, SNRIs, tricyclic antidepressants, or atypical antidepressants. However, patients should always inform their prescribing physician of all supplement use.
- Cognitive health in aging: For older adults, particularly those with elevated homocysteine levels, maintaining adequate folate status through diet and supplementation is a reasonable strategy for supporting cognitive health. A B-complex supplement providing 400 to 800 mcg of folic acid, along with adequate B12 and B6, can help maintain optimal one-carbon metabolism.
Summary
Folate plays a fundamental role in mental health through its essential contribution to one-carbon metabolism, SAMe production, and neurotransmitter synthesis. Low folate status is consistently associated with increased depression risk and poor treatment response. L-methylfolate at 15 mg daily has demonstrated efficacy as an adjunctive therapy for treatment-resistant depression. Elevated homocysteine, a marker of functional folate insufficiency, is associated with accelerated cognitive decline and increased dementia risk. Maintaining adequate folate status throughout life is an evidence-based strategy for supporting both mood and cognitive health.
Research Papers
- Papakostas GI, et al. L-methylfolate as adjunctive therapy for SSRI-resistant major depression: results of two randomized, double-blind, parallel-sequential trials. Am J Psychiatry. 2012;169(12):1267-1274.
- Gilbody S, et al. Methylenetetrahydrofolate reductase (MTHFR) genetic polymorphisms and psychiatric disorders: a HuGE review. (PubMed topic search)
- PubMed — Folate and depression meta-analyses (topic search)
- Smith AD, et al. Homocysteine-lowering by B vitamins slows the rate of accelerated brain atrophy in mild cognitive impairment (VITACOG). PLoS ONE. 2010;5(9):e12244.
- Seshadri S, et al. Plasma homocysteine as a risk factor for dementia and Alzheimer's disease. (PubMed topic search)
- PubMed — S-adenosylmethionine in depression (topic search)
- PubMed — Tetrahydrobiopterin (BH4) and monoamine synthesis (topic search)
- PubMed — Folate and cognitive decline (topic search)
- NIH Office of Dietary Supplements — Folate Fact Sheet for Health Professionals
- Linus Pauling Institute — Folate
- Harvard T.H. Chan School of Public Health — Folate (Folic Acid)
Connections
- Vitamin B9
- Depression
- Alzheimer's Disease
- Vitamin B12
- Folate and Pregnancy
- Vitamin B6
- Vitamin B6 and Homocysteine
- Magnesium
- Anxiety
- Homocysteine
- Dementia
- Methionine
- Tryptophan
- Fatigue
- B12 Deficiency Diagnosis
- Pyridoxine and Brain Health